false
FY
0001872812
X0
00-0000000
21.63
0001872812
2023-01-01
2023-12-31
0001872812
TCBP:AmericanDepositarySharesEachRepresentingOneOrdinaryShareNominalValue0.0001PerShareMember
2023-01-01
2023-12-31
0001872812
TCBP:WarrantsEachWarrantRepresentingRightToPurchaseOneAmericanDepositaryShareMember
2023-01-01
2023-12-31
0001872812
2023-06-30
0001872812
2024-03-29
0001872812
2023-12-31
0001872812
2022-12-31
0001872812
2022-01-01
2022-12-31
0001872812
us-gaap:CommonStockMember
2021-12-31
0001872812
TCBP:DeferredStockMember
2021-12-31
0001872812
us-gaap:AdditionalPaidInCapitalMember
2021-12-31
0001872812
us-gaap:RetainedEarningsMember
2021-12-31
0001872812
2021-12-31
0001872812
us-gaap:CommonStockMember
2022-12-31
0001872812
TCBP:DeferredStockMember
2022-12-31
0001872812
us-gaap:AdditionalPaidInCapitalMember
2022-12-31
0001872812
us-gaap:RetainedEarningsMember
2022-12-31
0001872812
us-gaap:CommonStockMember
2022-01-01
2022-12-31
0001872812
TCBP:DeferredStockMember
2022-01-01
2022-12-31
0001872812
us-gaap:AdditionalPaidInCapitalMember
2022-01-01
2022-12-31
0001872812
us-gaap:RetainedEarningsMember
2022-01-01
2022-12-31
0001872812
us-gaap:CommonStockMember
2023-01-01
2023-12-31
0001872812
TCBP:DeferredStockMember
2023-01-01
2023-12-31
0001872812
us-gaap:AdditionalPaidInCapitalMember
2023-01-01
2023-12-31
0001872812
us-gaap:RetainedEarningsMember
2023-01-01
2023-12-31
0001872812
us-gaap:CommonStockMember
2023-12-31
0001872812
TCBP:DeferredStockMember
2023-12-31
0001872812
us-gaap:AdditionalPaidInCapitalMember
2023-12-31
0001872812
us-gaap:RetainedEarningsMember
2023-12-31
0001872812
2023-12-15
2023-12-15
0001872812
us-gaap:IPOMember
2022-02-10
2022-02-10
0001872812
us-gaap:CommonStockMember
us-gaap:IPOMember
2022-02-10
2022-02-10
0001872812
2022-02-10
2022-02-10
0001872812
us-gaap:CommonStockMember
2022-02-10
2022-02-10
0001872812
2022-02-10
0001872812
2022-06-07
2022-06-08
0001872812
2022-06-08
0001872812
TCBP:SecuritiesFirstPurchaseAgreementMember
2022-11-27
2022-11-27
0001872812
TCBP:SecuritiesFirstPurchaseAgreementMember
TCBP:PrefundedWarrantMember
2022-11-27
0001872812
TCBP:SecuritiesFirstPurchaseAgreementMember
TCBP:SeriesAOrdinaryWarrantMember
2022-11-27
0001872812
TCBP:SecuritiesFirstPurchaseAgreementMember
TCBP:SeriesBOrdinaryWarrantMember
2022-11-27
0001872812
TCBP:OrdinaryWarrantMember
2022-11-27
2022-11-27
0001872812
TCBP:SecuritiesFirstPurchaseAgreementMember
TCBP:OrdinaryWarrantMember
2022-11-27
0001872812
TCBP:SecuritiesSecondPurchaseAgreementMember
2023-03-27
2023-03-27
0001872812
TCBP:SecuritiesSecondPurchaseAgreementMember
TCBP:PrefundedWarrantMember
2023-03-27
0001872812
TCBP:SecuritiesSecondPurchaseAgreementMember
TCBP:SeriesCOrdinaryWarrantMember
2023-03-27
0001872812
TCBP:SecuritiesSecondPurchaseAgreementMember
TCBP:PlacementAgentWarrantMember
2023-03-27
0001872812
TCBP:SecuritiesSecondPurchaseAgreementMember
TCBP:OrdinaryWarrantMember
2023-03-27
2023-03-27
0001872812
TCBP:SecuritiesSecondPurchaseAgreementMember
TCBP:PrefundedWarrantsMember
2023-03-27
2023-03-27
0001872812
2023-03-27
0001872812
2023-03-27
2023-03-27
0001872812
TCBP:AgreementMember
TCBP:SeriesAWarrantHoldersMember
2023-08-30
0001872812
TCBP:AgreementMember
TCBP:SeriesBWarrantHoldersMember
2023-08-30
0001872812
TCBP:AgreementMember
TCBP:SeriesCWarrantHoldersMember
2023-08-30
0001872812
TCBP:AgreementMember
2023-01-01
2023-12-31
0001872812
TCBP:AgreementMember
TCBP:SeriesDWarrantsMember
2023-08-01
2023-08-30
0001872812
TCBP:AgreementMember
2023-08-30
0001872812
2023-08-30
2023-08-30
0001872812
TCBP:SecuritiesThirdPurchaseAgreementMember
2023-12-18
2023-12-18
0001872812
TCBP:SecuritiesThirdPurchaseAgreementMember
TCBP:PrefundedWarrantMember
2023-12-18
0001872812
TCBP:SecuritiesThirdPurchaseAgreementMember
us-gaap:WarrantMember
2023-12-18
2023-12-18
0001872812
TCBP:SecuritiesSecondPurchaseAgreementMember
TCBP:SeriesEPurchaseWarrantMember
2023-12-18
0001872812
TCBP:SecuritiesThirdPurchaseAgreementMember
us-gaap:WarrantMember
2023-12-18
0001872812
TCBP:SecuritiesThirdPurchaseAgreementMember
2023-12-18
0001872812
TCBP:SecuritiesThirdPurchaseAgreementMember
TCBP:OrdinaryWarrantMember
2023-12-18
2023-12-18
0001872812
TCBP:SecuritiesThirdPurchaseAgreementMember
2023-09-05
2023-09-05
0001872812
2023-09-05
2023-09-05
0001872812
2023-12-18
2023-12-18
0001872812
us-gaap:SubsequentEventMember
2024-03-22
0001872812
TCBP:FacilityAndScientificEquipmentMember
srt:MinimumMember
2023-12-31
0001872812
TCBP:FacilityAndScientificEquipmentMember
srt:MaximumMember
2023-12-31
0001872812
us-gaap:ComputerEquipmentMember
2023-12-31
0001872812
us-gaap:OfficeEquipmentMember
2023-12-31
0001872812
2022-11-18
2022-11-18
0001872812
srt:ScenarioPreviouslyReportedMember
2022-01-01
2022-12-31
0001872812
srt:RevisionOfPriorPeriodReclassificationAdjustmentMember
2022-01-01
2022-12-31
0001872812
srt:ScenarioPreviouslyReportedMember
2022-12-31
0001872812
srt:RevisionOfPriorPeriodReclassificationAdjustmentMember
2022-12-31
0001872812
TCBP:FacilitiesAndScientificEquipmentMember
2021-12-31
0001872812
us-gaap:ComputerEquipmentMember
2021-12-31
0001872812
us-gaap:OfficeEquipmentMember
2021-12-31
0001872812
TCBP:FacilitiesAndScientificEquipmentMember
2022-01-01
2022-12-31
0001872812
us-gaap:ComputerEquipmentMember
2022-01-01
2022-12-31
0001872812
us-gaap:OfficeEquipmentMember
2022-01-01
2022-12-31
0001872812
TCBP:FacilitiesAndScientificEquipmentMember
2022-12-31
0001872812
us-gaap:ComputerEquipmentMember
2022-12-31
0001872812
us-gaap:OfficeEquipmentMember
2022-12-31
0001872812
TCBP:FacilitiesAndScientificEquipmentMember
2023-01-01
2023-12-31
0001872812
us-gaap:ComputerEquipmentMember
2023-01-01
2023-12-31
0001872812
us-gaap:OfficeEquipmentMember
2023-01-01
2023-12-31
0001872812
TCBP:FacilitiesAndScientificEquipmentMember
2023-12-31
0001872812
TCBP:SoftwareMember
2021-12-31
0001872812
TCBP:PatentsAndLicensesMember
2021-12-31
0001872812
TCBP:SoftwareMember
2022-01-01
2022-12-31
0001872812
TCBP:PatentsAndLicensesMember
2022-01-01
2022-12-31
0001872812
TCBP:SoftwareMember
2022-12-31
0001872812
TCBP:PatentsAndLicensesMember
2022-12-31
0001872812
TCBP:SoftwareMember
2023-01-01
2023-12-31
0001872812
TCBP:PatentsAndLicensesMember
2023-01-01
2023-12-31
0001872812
TCBP:SoftwareMember
2023-12-31
0001872812
TCBP:PatentsAndLicensesMember
2023-12-31
0001872812
TCBP:ConvertibleLoanNoteMember
2022-08-09
2022-08-09
0001872812
TCBP:ConvertibleLoanNoteMember
2022-08-09
0001872812
TCBP:ConvertibleLoanNoteMember
2023-06-19
2023-06-19
0001872812
2021-04-30
0001872812
2022-08-09
2022-08-09
0001872812
2022-08-09
0001872812
2023-04-03
2023-04-03
0001872812
2023-04-03
0001872812
us-gaap:WarrantMember
2023-01-01
2023-12-31
0001872812
us-gaap:ConvertibleDebtMember
TCBP:ResidualLoanMember
2022-12-31
0001872812
us-gaap:ConvertibleDebtMember
TCBP:EmbeddedDerivativeMember
2022-12-31
0001872812
us-gaap:ConvertibleDebtMember
2022-12-31
0001872812
us-gaap:ConvertibleDebtMember
TCBP:ResidualLoanMember
2023-01-01
2023-12-31
0001872812
us-gaap:ConvertibleDebtMember
TCBP:EmbeddedDerivativeMember
2023-01-01
2023-12-31
0001872812
us-gaap:ConvertibleDebtMember
2023-01-01
2023-12-31
0001872812
us-gaap:ConvertibleDebtMember
TCBP:ResidualLoanMember
2023-12-31
0001872812
us-gaap:ConvertibleDebtMember
TCBP:EmbeddedDerivativeMember
2023-12-31
0001872812
us-gaap:ConvertibleDebtMember
2023-12-31
0001872812
us-gaap:MeasurementInputExercisePriceMember
us-gaap:FairValueInputsLevel3Member
2022-12-31
0001872812
us-gaap:MeasurementInputSharePriceMember
us-gaap:FairValueInputsLevel3Member
2022-12-31
0001872812
us-gaap:MeasurementInputExpectedTermMember
us-gaap:FairValueInputsLevel3Member
2022-01-01
2022-12-31
0001872812
us-gaap:MeasurementInputOptionVolatilityMember
us-gaap:FairValueInputsLevel3Member
2022-12-31
0001872812
us-gaap:MeasurementInputRiskFreeInterestRateMember
us-gaap:FairValueInputsLevel3Member
2022-12-31
0001872812
us-gaap:MeasurementInputExpectedDividendRateMember
us-gaap:FairValueInputsLevel3Member
2022-12-31
0001872812
us-gaap:MeasurementInputExercisePriceMember
2023-12-31
0001872812
us-gaap:MeasurementInputSharePriceMember
2023-12-31
0001872812
us-gaap:MeasurementInputExpectedTermMember
2023-12-31
0001872812
us-gaap:MeasurementInputOptionVolatilityMember
2023-12-31
0001872812
us-gaap:MeasurementInputRiskFreeInterestRateMember
2023-12-31
0001872812
us-gaap:MeasurementInputExpectedDividendRateMember
2023-12-31
0001872812
TCBP:DeferredSharesMember
2023-12-31
0001872812
us-gaap:CommonClassAMember
2023-12-31
0001872812
us-gaap:IPOMember
us-gaap:CommonClassAMember
2023-12-31
0001872812
us-gaap:IPOMember
us-gaap:CommonClassAMember
2023-01-01
2023-12-31
0001872812
TCBP:AmericanDepositarySharesMember
2022-02-10
0001872812
us-gaap:IPOMember
2022-02-10
0001872812
TCBP:AmericanDepositarySharesMember
2022-08-09
0001872812
TCBP:AmericanDepositarySharesMember
us-gaap:ConvertibleDebtMember
2022-08-09
0001872812
us-gaap:CommonStockMember
2022-11-15
2022-11-15
0001872812
us-gaap:CommonStockMember
2022-11-15
0001872812
us-gaap:CommonStockMember
2022-11-24
2022-11-24
0001872812
us-gaap:CommonStockMember
2022-11-24
0001872812
TCBP:SeriesABAndCWarrantMember
2023-08-30
2023-08-30
0001872812
TCBP:AgreementMember
TCBP:SeriesDWarrantsMember
2023-08-30
0001872812
TCBP:InducementLetterAgreementMember
2023-08-30
2023-08-30
0001872812
TCBP:SecuritiesThirdPurchaseAgreementMember
TCBP:PlacementAgentWarrantsMember
2023-12-18
0001872812
TCBP:SecuritiesThirdPurchaseAgreementMember
us-gaap:CommonStockMember
2023-12-18
2023-12-18
0001872812
2022-11-18
0001872812
2023-03-15
2023-03-15
0001872812
TCBP:EmployeesMember
2022-12-31
0001872812
TCBP:EmployeesMember
2023-01-01
2023-12-31
0001872812
TCBP:EmployeesMember
2023-12-31
0001872812
TCBP:EmployeesAndDirectorsMember
2022-12-31
0001872812
TCBP:EmployeesAndDirectorsMember
2023-01-01
2023-12-31
0001872812
TCBP:EmployeesAndDirectorsMember
2023-12-31
0001872812
TCBP:EmployeesAndDirectorsMember
2022-01-01
2022-12-31
0001872812
2020-08-25
0001872812
TCBP:FirstPurchaseAgreementMember
2022-11-27
0001872812
TCBP:FirstPurchaseAgreementMember
TCBP:PrefundedWarrantMember
2022-11-27
0001872812
TCBP:FirstPurchaseAgreementMember
TCBP:SeriesAWarrantMember
2022-11-27
0001872812
TCBP:FirstPurchaseAgreementMember
TCBP:SeriesBWarrantMember
2022-11-27
0001872812
TCBP:FirstPurchaseAgreementMember
TCBP:SeriesBWarrantMember
2022-11-27
2022-11-27
0001872812
TCBP:FirstPurchaseAgreementMember
TCBP:PlacementAgentWarrantsMember
2022-11-27
0001872812
TCBP:FirstPurchaseAgreementMember
TCBP:ADSAndOrdinaryWarrantsMember
2022-11-27
0001872812
TCBP:FirstPurchaseAgreementMember
TCBP:PrefundedWarrantAndOrdinaryWarrantsMember
2022-11-27
0001872812
TCBP:SecondSecuritiesPurchaseAgreementMember
2023-03-27
2023-03-27
0001872812
TCBP:SecondSecuritiesPurchaseAgreementMember
TCBP:PrefundedWarrantMember
2023-03-27
0001872812
TCBP:SecondSecuritiesPurchaseAgreementMember
TCBP:SeriesCWarrantMember
2023-03-27
0001872812
TCBP:SecondSecuritiesPurchaseAgreementMember
TCBP:PlacementAgentWarrantsMember
2023-03-27
0001872812
TCBP:SecondSecuritiesPurchaseAgreementMember
TCBP:ADSAndOrdinaryWarrantsMember
2023-03-27
0001872812
TCBP:SecondSecuritiesPurchaseAgreementMember
TCBP:PrefundedWarrantAndOrdinaryWarrantsMember
2023-03-27
0001872812
TCBP:NovemberTwoThousandTwentyTwoWarrantMember
2023-07-10
0001872812
TCBP:MarchTwoThousandTwentyThreeNovemberTwoThousandTwentyTwoAndExistingWarrantsMember
2023-07-10
0001872812
TCBP:MarchTwoThousandTwentyThreeNovemberTwoThousandTwentyTwoAndExistingWarrantsMember
2023-07-10
2023-07-10
0001872812
TCBP:SecondSecuritiesPurchaseAgreementMember
us-gaap:WarrantMember
2023-03-27
0001872812
TCBP:SeriesAWarrantMember
2023-08-30
0001872812
TCBP:SeriesBWarrantMember
2023-08-30
0001872812
TCBP:SeriesCWarrantMember
2023-08-30
0001872812
TCBP:SeriesDWarrantsMember
2023-08-30
0001872812
TCBP:PlacementAgentWarrantsMember
2023-08-30
0001872812
TCBP:SecuritiesPurchaseAgreementMember
2023-12-18
2023-12-18
0001872812
TCBP:SecuritiesPurchaseAgreementMember
2023-12-18
0001872812
TCBP:SecuritiesPurchaseAgreementMember
TCBP:SeriesCWarrantMember
2023-12-18
0001872812
TCBP:SecuritiesPurchaseAgreementMember
TCBP:PlacementAgentWarrantsMember
2023-12-18
0001872812
TCBP:SecuritiesPurchaseAgreementMember
TCBP:ADSAndOrdinaryWarrantsMember
2023-12-18
0001872812
TCBP:SecuritiesPurchaseAgreementMember
TCBP:PrefundedWarrantAndOrdinaryWarrantsMember
2023-12-18
0001872812
TCBP:SeriesAWarrantMember
2023-12-31
0001872812
TCBP:SeriesAWarrantMember
2022-12-31
0001872812
TCBP:SeriesBWarrantMember
2023-12-31
0001872812
TCBP:SeriesBWarrantMember
2022-12-31
0001872812
TCBP:SeriesABPlacementAgentWarrantsMember
2023-12-31
0001872812
TCBP:SeriesABPlacementAgentWarrantsMember
2022-12-31
0001872812
TCBP:SeriesCWarrantsMember
2023-03-30
0001872812
TCBP:SeriesCPlacementAgentWarrantsMember
2023-12-31
0001872812
TCBP:SeriesCPlacementAgentWarrantsMember
2023-03-30
0001872812
TCBP:SeriesDWarrantsMember
2023-08-30
0001872812
TCBP:SeriesDPlacementAgentWarrantsMember
2023-08-30
0001872812
TCBP:SeriesEWarrantsMember
2023-12-18
0001872812
TCBP:SeriesEPlacementAgentWarrantsMember
2023-12-18
0001872812
us-gaap:MeasurementInputExercisePriceMember
us-gaap:WarrantMember
2023-07-10
0001872812
us-gaap:MeasurementInputSharePriceMember
us-gaap:WarrantMember
2023-07-10
0001872812
us-gaap:MeasurementInputMaturityMember
us-gaap:WarrantMember
2023-07-10
0001872812
us-gaap:MeasurementInputPriceVolatilityMember
us-gaap:WarrantMember
2023-07-10
0001872812
us-gaap:MeasurementInputRiskFreeInterestRateMember
us-gaap:WarrantMember
2023-07-10
0001872812
TCBP:MeasurementInputDividendYieldMember
us-gaap:WarrantMember
2023-07-10
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputExercisePriceMember
TCBP:SeriesAWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputExercisePriceMember
TCBP:SeriesAWarrantsMember
2022-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputSharePriceMember
TCBP:SeriesAWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputSharePriceMember
TCBP:SeriesAWarrantsMember
2022-12-31
0001872812
us-gaap:MeasurementInputMaturityMember
us-gaap:FairValueInputsLevel2Member
TCBP:SeriesAWarrantsMember
2023-12-31
0001872812
us-gaap:MeasurementInputMaturityMember
us-gaap:FairValueInputsLevel2Member
TCBP:SeriesAWarrantsMember
2022-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputPriceVolatilityMember
TCBP:SeriesAWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputPriceVolatilityMember
TCBP:SeriesAWarrantsMember
2022-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputRiskFreeInterestRateMember
TCBP:SeriesAWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputRiskFreeInterestRateMember
TCBP:SeriesAWarrantsMember
2022-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
TCBP:MeasurementInputDividendYieldMember
TCBP:SeriesAWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
TCBP:MeasurementInputDividendYieldMember
TCBP:SeriesAWarrantsMember
2022-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputExercisePriceMember
TCBP:SeriesBWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputExercisePriceMember
TCBP:SeriesBWarrantsMember
2022-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputSharePriceMember
TCBP:SeriesBWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputSharePriceMember
TCBP:SeriesBWarrantsMember
2022-12-31
0001872812
us-gaap:MeasurementInputMaturityMember
us-gaap:FairValueInputsLevel2Member
TCBP:SeriesBWarrantsMember
2023-12-31
0001872812
us-gaap:MeasurementInputMaturityMember
us-gaap:FairValueInputsLevel2Member
TCBP:SeriesBWarrantsMember
2022-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputPriceVolatilityMember
TCBP:SeriesBWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputPriceVolatilityMember
TCBP:SeriesBWarrantsMember
2022-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputRiskFreeInterestRateMember
TCBP:SeriesBWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputRiskFreeInterestRateMember
TCBP:SeriesBWarrantsMember
2022-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
TCBP:MeasurementInputDividendYieldMember
TCBP:SeriesBWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
TCBP:MeasurementInputDividendYieldMember
TCBP:SeriesBWarrantsMember
2022-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputExercisePriceMember
TCBP:SeriesABPlacementAgentWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputExercisePriceMember
TCBP:SeriesABPlacementAgentWarrantsMember
2022-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputSharePriceMember
TCBP:SeriesABPlacementAgentWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputSharePriceMember
TCBP:SeriesABPlacementAgentWarrantsMember
2022-12-31
0001872812
us-gaap:MeasurementInputMaturityMember
us-gaap:FairValueInputsLevel2Member
TCBP:SeriesABPlacementAgentWarrantsMember
2023-12-31
0001872812
us-gaap:MeasurementInputMaturityMember
us-gaap:FairValueInputsLevel2Member
TCBP:SeriesABPlacementAgentWarrantsMember
2022-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputPriceVolatilityMember
TCBP:SeriesABPlacementAgentWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputPriceVolatilityMember
TCBP:SeriesABPlacementAgentWarrantsMember
2022-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputRiskFreeInterestRateMember
TCBP:SeriesABPlacementAgentWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputRiskFreeInterestRateMember
TCBP:SeriesABPlacementAgentWarrantsMember
2022-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
TCBP:MeasurementInputDividendYieldMember
TCBP:SeriesABPlacementAgentWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
TCBP:MeasurementInputDividendYieldMember
TCBP:SeriesABPlacementAgentWarrantsMember
2022-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputExercisePriceMember
TCBP:SeriesCWarrantsMember
2023-03-30
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputSharePriceMember
TCBP:SeriesCWarrantsMember
2023-03-30
0001872812
us-gaap:MeasurementInputMaturityMember
us-gaap:FairValueInputsLevel2Member
TCBP:SeriesCWarrantsMember
2023-03-30
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputPriceVolatilityMember
TCBP:SeriesCWarrantsMember
2023-03-30
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputRiskFreeInterestRateMember
TCBP:SeriesCWarrantsMember
2023-03-30
0001872812
us-gaap:FairValueInputsLevel2Member
TCBP:MeasurementInputDividendYieldMember
TCBP:SeriesCWarrantsMember
2023-03-30
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputExercisePriceMember
TCBP:SeriesCPlacementAgentWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputExercisePriceMember
TCBP:SeriesCPlacementAgentWarrantsMember
2023-03-30
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputSharePriceMember
TCBP:SeriesCPlacementAgentWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputSharePriceMember
TCBP:SeriesCPlacementAgentWarrantsMember
2023-03-30
0001872812
us-gaap:MeasurementInputMaturityMember
us-gaap:FairValueInputsLevel2Member
TCBP:SeriesCPlacementAgentWarrantsMember
2023-12-31
0001872812
us-gaap:MeasurementInputMaturityMember
us-gaap:FairValueInputsLevel2Member
TCBP:SeriesCPlacementAgentWarrantsMember
2023-03-30
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputPriceVolatilityMember
TCBP:SeriesCPlacementAgentWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputPriceVolatilityMember
TCBP:SeriesCPlacementAgentWarrantsMember
2023-03-30
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputRiskFreeInterestRateMember
TCBP:SeriesCPlacementAgentWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputRiskFreeInterestRateMember
TCBP:SeriesCPlacementAgentWarrantsMember
2023-03-30
0001872812
us-gaap:FairValueInputsLevel2Member
TCBP:MeasurementInputDividendYieldMember
TCBP:SeriesCPlacementAgentWarrantsMember
2023-12-31
0001872812
us-gaap:FairValueInputsLevel2Member
TCBP:MeasurementInputDividendYieldMember
TCBP:SeriesCPlacementAgentWarrantsMember
2023-03-30
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputExercisePriceMember
TCBP:SeriesDWarrantsMember
2023-08-30
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputSharePriceMember
TCBP:SeriesDWarrantsMember
2023-08-30
0001872812
us-gaap:MeasurementInputMaturityMember
us-gaap:FairValueInputsLevel2Member
TCBP:SeriesDWarrantsMember
2023-08-30
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputPriceVolatilityMember
TCBP:SeriesDWarrantsMember
2023-08-30
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputRiskFreeInterestRateMember
TCBP:SeriesDWarrantsMember
2023-08-30
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputExercisePriceMember
TCBP:SeriesDPlacementAgentWarrantsMember
2023-08-30
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputSharePriceMember
TCBP:SeriesDPlacementAgentWarrantsMember
2023-08-30
0001872812
us-gaap:MeasurementInputMaturityMember
us-gaap:FairValueInputsLevel2Member
TCBP:SeriesDPlacementAgentWarrantsMember
2023-08-30
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputPriceVolatilityMember
TCBP:SeriesDPlacementAgentWarrantsMember
2023-08-30
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputRiskFreeInterestRateMember
TCBP:SeriesBPlacementAgentWarrantsMember
2023-08-30
0001872812
us-gaap:FairValueInputsLevel2Member
TCBP:MeasurementInputDividendYieldMember
TCBP:SeriesDPlacementAgentWarrantsMember
2023-08-30
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputExercisePriceMember
TCBP:SeriesEWarrantsMember
2023-12-18
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputSharePriceMember
TCBP:SeriesEWarrantsMember
2023-12-18
0001872812
us-gaap:MeasurementInputMaturityMember
us-gaap:FairValueInputsLevel2Member
TCBP:SeriesEWarrantsMember
2023-12-18
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputPriceVolatilityMember
TCBP:SeriesEWarrantsMember
2023-12-18
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputRiskFreeInterestRateMember
TCBP:SeriesEWarrantsMember
2023-12-18
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputExercisePriceMember
TCBP:SeriesEPlacementAgentWarrantsMember
2023-12-18
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputSharePriceMember
TCBP:SeriesEPlacementAgentWarrantsMember
2023-12-18
0001872812
us-gaap:MeasurementInputMaturityMember
us-gaap:FairValueInputsLevel2Member
TCBP:SeriesEPlacementAgentWarrantsMember
2023-12-18
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputPriceVolatilityMember
TCBP:SeriesEPlacementAgentWarrantsMember
2023-12-18
0001872812
us-gaap:FairValueInputsLevel2Member
us-gaap:MeasurementInputRiskFreeInterestRateMember
TCBP:SeriesEPlacementAgentWarrantsMember
2023-12-18
0001872812
us-gaap:FairValueInputsLevel2Member
TCBP:MeasurementInputDividendYieldMember
TCBP:SeriesEPlacementAgentWarrantsMember
2023-12-18
0001872812
us-gaap:DomesticCountryMember
2023-12-31
0001872812
us-gaap:SubsequentEventMember
TCBP:PrefundedWarrantMember
2024-01-01
2024-01-31
0001872812
TCBP:NonEmployeeDirectorMember
us-gaap:SubsequentEventMember
TCBP:TwentyTwentyOneSharesOptionSchemeMember
2024-02-29
2024-02-29
0001872812
TCBP:NonEmployeeDirectorMember
us-gaap:SubsequentEventMember
TCBP:TwentyTwentyOneSharesOptionSchemeMember
2024-02-29
0001872812
srt:ChiefFinancialOfficerMember
us-gaap:SubsequentEventMember
TCBP:TwentyTwentyOneSharesOptionSchemeMember
2024-02-29
2024-02-29
0001872812
srt:ChiefFinancialOfficerMember
us-gaap:SubsequentEventMember
TCBP:TwentyTwentyOneSharesOptionSchemeMember
2024-02-29
0001872812
srt:ChiefExecutiveOfficerMember
us-gaap:SubsequentEventMember
TCBP:TwentyTwentyOneSharesOptionSchemeMember
2024-02-29
2024-02-29
0001872812
srt:ChiefExecutiveOfficerMember
us-gaap:SubsequentEventMember
TCBP:TwentyTwentyOneSharesOptionSchemeMember
2024-02-29
0001872812
us-gaap:SubsequentEventMember
TCBP:BryanKobelMember
2024-03-08
2024-03-08
0001872812
us-gaap:SubsequentEventMember
TCBP:BryanKobelMember
2024-03-08
0001872812
us-gaap:SubsequentEventMember
TCBP:BryanKobelMember
2024-03-07
2024-03-07
0001872812
us-gaap:SubsequentEventMember
TCBP:BryanKobelMember
2024-03-07
0001872812
us-gaap:SubsequentEventMember
2024-03-12
2024-03-12
0001872812
us-gaap:SubsequentEventMember
TCBP:SeriesDWarrantsMember
2024-03-12
0001872812
us-gaap:SubsequentEventMember
TCBP:SeriesDWarrantsMember
2024-03-12
2024-03-12
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
xbrli:pure
iso4217:EUR
iso4217:EUR
xbrli:shares
TCBP:Integer
iso4217:GBP
iso4217:GBP
xbrli:shares
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
FORM
10-K
| ☒ |
AnNUAL
REPORT pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 |
| |
| For
the fiscal year ended December 31, 2023 |
| |
| or |
| |
| ☐ |
Transition
report pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 |
| |
| For
the transaction period from _____________ to _____________ |
Commission
File No. 001-41231
TC
BIOPHARM (HOLDINGS) PLC
(Exact
Name of Registrant as Specified in Its Charter)
| Scotland |
|
N/A |
(State
or other jurisdiction of
incorporation or organization) |
|
(I.R.S.
Employer
Identification No.) |
Maxim
1, 2 Parklands Way
Holytown,
Motherwell, ML1 4WR
Scotland,
United Kingdom |
|
N/A |
| Address
of Principal Executive Offices |
|
Zip
Code |
+44
(0) 141 433 7557
(Registrant’s
telephone number, including area code)
Securities
registered pursuant to Section 12(b) of the Act:
| Title
of each class |
|
Trading
Symbol(s) |
|
Name
of each exchange on which registered |
| |
|
|
|
|
| American
Depositary Shares, each representing one Ordinary Share, nominal value £0.0001 per share |
|
TCBP |
|
The
Nasdaq Stock Market LLC |
| |
|
|
|
|
| Warrants,
each warrant representing the right to purchase one American Depositary Share |
|
TCBPW |
|
The
Nasdaq Stock Market LLC |
Securities
registered pursuant to Section 12(g) of the Act:
Indicate
by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐ No ☒
Indicate
by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Exchange Act. Yes ☐
No ☒
Indicate
by check mark whether the registrant: (1) filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act
of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has
been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate
by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule
405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant
was required to submit such files). Yes ☒ No ☐
Indicate
by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting
company or an emerging growth company. See definitions of “large accelerated filer,” “accelerated filer,” “smaller
reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large
Accelerated Filer ☐ |
Accelerated
Filer ☐ |
| Non-accelerated
Filer ☒ |
Smaller
Reporting Company ☒ |
| |
Emerging
Growth Company ☒ |
If
an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying
with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act.
Indicate
by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness
of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered
public accounting firm that prepared or issued its audit report. Yes ☐ No ☒
If
securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant
included in the filing reflect the correction of an error to previously issued financial statements. ☐
Indicate
by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation
received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate
by check mark whether the registrant is a shell company (as defined in Rule 12(b)-2 of the Exchange Act). Yes ☐ No ☒
The
aggregate market value of the voting and non-voting ordinary shares held by non-affiliates of the registrant, based upon $10.84, the
closing price of the registrant’s American Depositary Shares on the Nasdaq Capital Market on June 30, 2023 (the last business day
of the registrant’s most recently completed second fiscal quarter) was approximately $3.1 million.
As
of March 29, 2024, 63,902,641 shares of the registrant’s ordinary shares, £0.0001 par value per share representing 2,426,504
American Depository shares, were outstanding.
TC
BIOPHARM (HOLDINGS) PLC
2023
ANNUAL REPORT ON FORM 10-K
TABLE
OF CONTENTS
Unless
we state otherwise or the context otherwise requires, the terms “TC Biopharm,”, “TCB”, “we,” “us,”
“our” and the “Company” refer to TC Biopharm (Holdings) plc.
SPECIAL
NOTE REGARDING FORWARD-LOOKING STATEMENTS
This
Annual Report on Form 10-K, or Annual Report, contains forward-looking statements that involve risks and uncertainties. We make such
forward-looking statements pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995 and other federal
securities laws. All statements other than statements of historical facts contained in this Annual Report are forward-looking statements.
In some cases, you can identify forward-looking statements by terminology such as “may”, “will”, “should”,
“expects”, “intends”, “plans”, “anticipates”, “believes”, “estimates”,
“predicts”, “potential”, “continue” or the negative of these terms or other comparable terminology.
Forward-looking
statements are neither historical facts nor assurances of future performance, and are based only on our current beliefs, expectations
and assumptions regarding the future of our business, future plans and strategies, projections, anticipated events and trends, the economy
and other future conditions. Because forward-looking statements relate to the future, they are subject to inherent uncertainties, risks
and changes in circumstances that are difficult to predict and many of which are outside of our control. Therefore, you should not rely
on any of these forward-looking statements. Important factors that could cause our actual results and financial condition to differ materially
from those indicated in the forward-looking statements include, among others, the following:
| |
● |
the
ultimate impact of the coronavirus (COVID-19) pandemic, or any other health epidemic, on our business, results of operations,
cash flows, financial condition and liquidity, and the global economy as a whole; |
| |
● |
the
sufficiency of our existing cash and cash equivalents to meet our working capital and capital expenditure needs over the next 12
months and our need to raise additional capital; |
| |
● |
our
ability to generate revenue from products; |
| |
● |
our
limited operating history; |
| |
● |
our
ability to maintain proper and effective internal financial controls; |
| |
● |
our
ability to continue to operate as a going concern; |
| |
● |
changes
in laws, government regulations and policies and interpretations thereof; |
| |
● |
our
ability to obtain and maintain protection for our intellectual property; |
| |
● |
our
ability to attract and retain qualified employees and key personnel; |
| |
● |
our
ability to manage our rapid growth and organizational change effectively; |
| |
● |
the
possibility of security vulnerabilities, cyberattacks and network disruptions, including breaches of data security and privacy leaks,
data loss, and business interruptions; |
| |
● |
our
compliance with data privacy laws and regulations; |
| |
● |
our
ability to develop and maintain our brand cost-effectively; and |
| |
● |
the
other factors set forth in Part I, Item 1A, “Risk Factors” of this Form 10-K. |
These
forward-looking statements speak only as of the date of this Form 10-K and are subject to business and economic risks. We do not undertake
any obligation to update or revise the forward-looking statements to reflect events that occur or circumstances that exist after the
date on which such statements were made, except to the extent required by law.
PART
I
Overview
We
are a clinical-stage biopharmaceutical company focused on developing novel immunotherapy products based on our proprietary allogeneic
gamma delta T (GD-T) cell platform. Harnessing the innate ability of GD-Ts has enabled us to develop a range of clinical-stage cell therapies
designed to combat cancer and viral infection.
In-house
clinical studies have demonstrated that our unmodified allogeneic GD-T products are (i) well tolerated and (ii) show preliminary evidence
of disease modification in patients with the late-stage blood cancer, known as acute myeloid leukemia – AML. Based on clinical
data generated by us believe that unmodified GD-Ts have the potential to treat all blood cancers.
TCB
now is embarking on phase 2b-into-pivotal (phase 3) clinical studies with a view to launching its first oncology product for the treatment
of AML. Clinical results generated thus far have enabled us to obtain FDA orphan drug status for treatment of AML.
In
addition to unmodified allogenic GD-Ts for treatment of blood cancers, we are also developing an innovative range of genetically-modified
CAR-T products for treatment of solid cancers. We believe that solid cancers are more difficult to treat than blood cancers and may require
the addition of a CAR “chimeric antigen receptor” (i) to help therapeutic cells to “navigate” into diseased cancerous
tissue and (ii) to retain therapeutic cells in-situ at the lesion for maximal efficacy (increased persistence).
In
order to manufacture our portfolio of allogeneic products, we select the highest quality GD-T cells from healthy donors, activate the
cells and grow them in large numbers at our in-house GMP-compliant manufacturing facility before administration to a patient in order
to target and then destroy malignant or virally-infected tissues. We believe that we have introduced a step-change to our manufacturing
platform by implementing a freeze-thaw process that will allow product to be shipped from cleanroom to patient without any shelf-life
issue. Resulting products, we believe, will be more cost-effective and straightforward to ship form cleanroom to clinic.
History
and Development of the Company
We
are a public company with limited liability incorporated on October 25, 2021, pursuant to the laws of Scotland, under the name TC BioPharm
(Holdings) plc. We were incorporated with nominal share capital for the purpose of becoming the ultimate holding company of TC BioPharm
Limited, the company in which our principal clinical, research and development operations have been and are currently undertaken, and
for the purpose of consummating the corporate reorganization described herein. TC BioPharm (Holdings) plc will not conduct any operations
except those as the listed entity, as explained below.
TC
BioPharm Limited was incorporated on July 1, 2013, as a private company with limited liability pursuant to the laws of Scotland and conducts
our principal clinical. research and development operations. TC BioPharm Limited has two wholly owned subsidiaries:
| |
● |
TC
BioPharm BV, The Netherlands – incorporated March 2019 |
| |
● |
TC
BioPharm (North America) Inc. – incorporated June 2021 |
These
two subsidiaries have had limited operating activity since their incorporation. It is anticipated that TC BioPharm (North America) Inc.
will develop operations and a management presence in the United States, with a view to expanding our product offerings into that jurisdiction
in the future.
The
corporate reorganization took place in several steps as follows:
| |
● |
On
December 17, 2021, all shareholders in TC BioPharm Limited and holders of convertible loan notes in TC BioPharm Limited exchanged
their shares and convertible loan notes for the same number and classes of newly issued shares and/or convertible loan notes in TC
BioPharm (Holdings) Limited and, as a result, TC BioPharm Limited became a wholly owned subsidiary of TC BioPharm (Holdings) Limited. |
| |
|
|
| |
● |
On
December 17, 2021, TC BioPharm (Holdings) Limited carried out a 10 for 1 forward split of all classes of its share capital. |
| |
● |
On
December 30, 2021, holders of various options to subscribe for shares in TC BioPharm Limited exchanged their options for equivalent
options in TC BioPharm (Holdings) Limited. |
| |
|
|
| |
● |
On
January 10, 2022, TC BioPharm (Holdings) Limited re-registered under the laws of Scotland as a public limited company, with a change
of name to TC BioPharm (Holdings) plc. |
| |
|
|
| |
● |
Immediately
prior to the completion of the initial public offering, the different classes and nominal values of issued share capital of TC BioPharm
(Holdings) plc were reorganized into a single class of ordinary shares with the same nominal value. The ADSs represent a portion
of these ordinary shares. |
| |
|
|
| |
● |
On
February 10, 2022, TC BioPharm (Holdings) plc completed an initial public offering on the Nasdaq Capital Market. Our ADSs and warrants
are traded under the symbols TCBP and TCBPW respectively. Our ordinary shares are not listed. Our registered office in the United
Kingdom is located at Maxim 1, 2 Parklands Way, Holytown, Motherwell, ML1 4WR, Scotland, United Kingdom, and the telephone of our
registered office is +44 (0) 141 433 7557. |
On
November 18, 2022 the Company undertook a reverse share split such that fifty issued ordinary shares were exchanged for one new ordinary
share. As a result of the share split, all references in these consolidated financial statements and accompanying notes to units of ordinary
shares or per share amounts are reflective of the reverse share split for all periods presented. In addition, the exercise prices and
the numbers of ordinary shares issuable upon the exercise of any outstanding options to purchase ordinary shares were proportionally
adjusted pursuant to the respective anti-dilution terms of the share-based payment plans.
On
December 15, 2023 the Company changed its ratio of ADSs to one ADS representing one ordinary share to one ADS representing 20
ordinary shares. As a result of the ratio change, all references to ADSs in these consolidated financial statements and accompanying
notes to units of ADSs or per ADS amounts are reflective of the ratio change for all periods presented. In addition,
the exercise prices and the numbers of ADSs issuable upon the exercise of any outstanding options to purchase ADSs were proportionally adjusted pursuant to the respective anti-dilution terms of the share-based payment plans.
Our
agent for service of process in the United States is TC BioPharm (North America) Inc., c/o Business Filings, Inc., 108 West 13th Street,
Wilmington, Delaware 19801 and the telephone number is (800) 981-7183.
Our
capital expenditures for the years ended December 31, 2023 and 2022 were £0.1 million and £0.2 million, respectively. These
capital expenditures primarily consisted of property, plant and equipment, computer equipment and office equipment in the United Kingdom.
The
SEC maintains an Internet site that contains reports, proxy information statements and other information regarding issuers that file
electronically with the SEC. The address of that site is http://www.sec.gov. Our website address is www.tcbiopharm.com. Information contained
in, or that can be accessed through, our website is not a part of, and shall not be incorporated by reference into, this document. We
have included our website address in this document solely as an inactive textual reference.
Business
Strategy
We
have taken a step-wise approach to clinical development and commercialization. To achieve this, we have made the clinical transition
from autologous GD-Ts to allogeneic GD-Ts to CAR-modified allogeneic GD-Ts. Our commercialization strategy is to introduce products firstly
in blood cancers (AML initially) and then solid tumor indications.
Our
strategic objective is to build a global therapeutic business with an extensive portfolio of GD-T cell-based products with the potential
to significantly improve the outcomes of patients with cancer and infectious disease. In order to achieve our objective, we are focused
on delivering success in the following areas:
Progress
unmodified GD-T2s into Phase 2/3 clinical trials for the treatment of blood cancers
Having
generated meaningful clinical data showing our product is well-tolerated in late-stage AML patients with no remaining treatment options,
we commenced phase 2b-into pivotal (phase 3) clinical studies under the trial name ‘ACHIEVE’, with OmnImmune® during
2022 in AML patients who have failed to respond adequately to induction therapy. The aim is to provide a form of salvage therapy which
will either stabilize the patient, thereby preventing disease progression, or delay the requirement for human stem cell transplant. Our
initial trial centers are in the UK and we are currently dosing patients in this trial. Working on the premise that other blood cancers
should respond to GD-Ts in a similar manner to AML, TCB plans to conduct clinical studies for OmnImmune® in other hematological malignancies
in future.
OmnImmune®
clinical program
Our
OmnImmune® clinical program is an example of our stepwise approach to clinical development. The initial phase 1b/2a trials were
undertaken using fresh cell-based product under the program number TCB002. For ease of reference, when discussing that specific
trial, we refer the program as OmnImmune® (TCB002). The phase 2b-into pivotal (phase 3) clinical studies are using a frozen
cell-based product under the program number TCB008-001. When discussing that specific trial, we refer the program as OmnImmune®
(TCB008-001).
Unmodified
GD-T2s for use in the treatment of infectious disease
Gamma-delta
T cells are dysfunctional in patients with many severe viral diseases and TCB anticipates that its unmodified gamma delta T cell therapy
platform will be used in due course to treat viral infections as well as cancers under the name ImmuniStim®. For example, during
2022 TCB developed a clinical trial protocol to treat patients with COVID 19. Because of the progress of the disease and absence of appropriate
trial patients this trial is not currently being progressed, although we expect to continue our infectious disease program in future.
Progress
CAR-modified GD-Ts into Phase 1 clinical trials for treatment of solid tumors
TCB
aims to treat solid cancers using its patented co-stimulatory GD-T and is currently undergoing pre-clinical work to determine the most
appropriate CAR construct and target indication.
Grow
our business operations to support the increasing number of clinical-phase products in development
We
believe that our existing cell and gene manufacturing facility in the UK has the capacity to support our committed clinical development
plans. We plan to continue to build upon this to support expansion of our product pipelines to new assets and to grow our clinical team.
We also will work closely with vendors to embrace emerging technologies in our manufacturing operations that are appropriate and optimized
for our products to continually improve the quality and efficiency of our manufacturing systems. We believe that maintaining in-house
control of these activities is critical to effective and efficient progression and we will continue to seek to build integrated business
functions where possible.
Apply
our discovery engine to target further diseases and add additional functionality to our products
As
a platform technology, our co-stimulatory CAR-T GD-T cell system has a wealth of potential options to build added functionality into
our cell-based platform. We plan to continue to innovate and partner in the field to augment our drug products and introduce next generation
attributes. We also plan to continue to innovate our manufacturing and supply chains to efficiently scale our processes and simplify
the interface with patients and healthcare professionals, whilst continually seeking to reduce manufacturing costs to improve patient
access.
Expand
our intellectual property portfolio and acquire additional technologies to augment our strong IP position
We
intend to continue building on our technology platform, comprised of intellectual property, proprietary methods and know-how in the field
of GD-T cells. These assets form the foundation for our ability, not only to strengthen our product pipeline, but also to successfully
defend and expand our position as a leader in the field of GD-T based immune-oncology.
Our
Pipeline
What
are gamma delta T cells?
The
immune system plays an important role in targeting and destroying cancer cells. One component has evolved to scan the body for diseased
cells and eradicate them. In humans, GD-Ts arise as a number of different subtypes, defined by the sequence of the gamma and delta chains
of the T-cell receptor (TCR) on the cell surface. The gammadelta2 (GD-T2) subtype typically is the most abundant of these cells in healthy
humans, and its TCR- of anti-cancer immunity is GD-T cells – a type of white blood cell that express a variety of innate receptors,
which mediated signaling has been fully characterized by researchers.
Virally-infected
or cancerous cells become stressed and accumulate cell surface phosphoantigens (isopentenyl pyrophosphate – IPP’s) which
are recognized by GD-T2 cells. Our proprietary technology platform includes the manufacturing of unmodified and genetically modified
(CAR-T) GD-T cells as therapeutic candidates for use in clinical trials and commercialization. Almost all aspects of the value-chain
from product manufacture, quality systems, clinical and regulatory are operated in-house by TC BioPharm. We believe this is one of our
core competitive advantages, which we believe will contribute materially to our ability to overcome the challenging nature of developing
new products.
Human
lymphocytes comprise two groups of cells, B cells that generate antibodies for humoral immunity, and T cells that are responsible for
cellular immune responses. In healthy individuals, GD-T cells generally represent between 1% and 10% of peripheral blood T lymphocytes
and present one of the first lines of defense against a wide range of bacterial and viral pathogens, as well as surveillance for cancerous
cells. GD-T cells have the ability to regulate the initial immune response in several ways, including recruitment of other immune cells
such as neutrophils, dendritic cells and macrophages through production of various chemokines (Kirby et al., 2007). Depletion
of GD-T cells leads to impaired host defense to lung infections, for example (Moore et al., 2000; Lockhart et al., 2006).
The predominant subset of GD-T cells in the blood is the GD-T2, which mediates a variety of immune responses by direct cytolysis of cancer
cells and infected cells, development of memory phenotypes and modulation of other immune cells. The gammadelta1 (GD-T1) is a functionally
distinct subset of GD-T cells, which are a predominantly tissue resident population. GD-T1s are less well characterized, but their cytotoxic
function also has been described in different liquid and solid tumors (Siegers & Lamb, 2014).
Both
subsets of GD-T cells are thought to play a role in autoimmune disorders such as celiac disease, rheumatoid arthritis, autoimmune polyglandular
syndrome and sarcoidosis where such lymphocytes are seen to accumulate in high numbers.
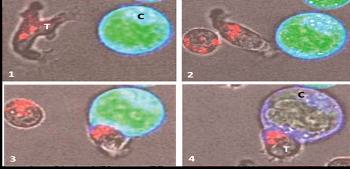 |
|
GD-T
cell killing a cancer cell.
(1)
A human GD-T (labelled ‘T’) identifies and scans (2) the surface of a cancer cell (labelled ‘C’). On contact
with the cancer cell (3) the GD-T releases perforin granules (stained red) into the cancer cell, rupturing its membrane (4) destroying
the cancer cell (adapted from – Enc Life Sci, Jul-2007). |
How
can GD-Ts be used to treat disease?
Cellular
immunotherapy is a form of treatment that harnesses the cells of the immune system to combat disease and is one of the most actively
pursued areas of research by biotechnology and pharmaceutical companies today. Interest in immunotherapy is largely driven by recent
compelling efficacy data in cancers and by the potential to achieve a cure or functional cure for some patients. While the field of immunotherapy
in cancer, in general, has achieved proof of concept and yielded significant durable responses in multiple tumor types, there remain
major tumor types such as colon, breast, and prostate cancers as well as patient groups within responsive tumors, that do not respond
to current immunotherapy treatments. One theory to explain this non-responsiveness is that certain tumors require direct immune stimulation.
T cell-based technologies seek to deliver activated T cells towards malignancies to initiate an immune response. The primary challenges
in the field have been to couple an acceptable efficacy and safety profile to successfully target solid tumors.
Adoptive
T cell transfer typically involves administration of autologous, allogeneic, or genetically-modified T cells (see footer below) into
a recipient host with the specific goal of boosting or transferring enhanced immunologic functionality. One of the most advanced cell-based
approaches – chimeric antigen receptor modified T cells (CAR-T) – has gained momentum. In a recent study, patients with refractory
B cell acute lymphoblastic leukemia were treated with autologous genetically-modified T cells, with almost 90% of patients showing a
marked improvement (Pan et al., 2017). Although the treatment is showing promise for specific tumor types, the safety profile
remains a concern, as serious adverse events have previously been reported following CAR-T therapy (Grigor et al., 2017). As a
consequence of safety issues related to this approach, regulatory approval may be more complex for this genetically modified T cell therapy
which effectively has two ‘starting materials’ – (i) the cellular component, and (ii) a lentiviral vector. The therapeutic
premise is well-established – T cells are transduced with a viral vector encoding a chimeric antigen receptor capable of recognizing
cancer-specific antigens, for example, CD19 which is commonly expressed on several tumors such as myeloma and B cell lymphomas. Transduction
is the process by which DNA is transferred from one cell to another by a virus; in this specific case DNA is introduced via a viral vector
(a tool commonly used by molecular biologists to deliver genetic material).
Following
transduction, the T cells are genetically primed to recognize and kill specific tumor cells expressing the target antigen. The process
involves extracting a patient’s T cells (or growing an allogeneic T cell bank), transfecting the cells with a gene for a chimeric-antigen-receptor
(CAR), and re-infusing transfected T cells into the patients. The use of cancer-specific cell therapies has gained momentum as several
companies demonstrated that genetically modified CAR-T cells are efficacious when directed against blood tumors. These breakthrough findings
have moved cell-based immunotherapy into the forefront of clinical oncology with two drugs now in the market.
T
lymphocytes have long been known to play an important role in cancer suppression and modulation of tumor growth and numerous experimental
studies have demonstrated the anti-cancer potential of GD-T lymphocytes. Indeed, GD-T cells can recognize a number of specific tumor-associated
molecules including non-peptidic antigens (IPP’s – isopentenyl pyrophosphate) and immune surveillance stress signals (such
as HSP60/70, MICA, MICB, and ULBP) present on the surface of transformed cells. The GD-T cell overexpresses IL-2 receptors and this cytokine
is necessary to activate them (Kjeldsen-Kragh, 1993). On recognizing a tumor cell, GD-T cells exert their anti-cancer properties via
release of both perforin and of granzyme, a serine protease which enters the target cell to trigger cell death (apoptosis). Our research
efforts are focused entirely on targeting tumors in ways that may result in an improved therapeutic index and that have potential applications
in solid tumors as well as hematological malignancies. In contrast to conventional AB CAR-T cells, our GD-T cell technology provides
greater specificity in targeting tumors through recognition of IPP-expressing cells, whilst avoiding on-target, off-tumor effects on
healthy tissue lacking in IPPs.
Liquid
cancers
For
cell therapies to be effective several parameters need to be addressed. These include (i) viability, (ii) homing to the tumor, (iii)
persistence at the tumor, and (iv) target-specificity.
Use
of unmodified GD-Ts to treat blood cancers addresses all the above factors. We believe that (i) we have demonstrated therapeutic cells
remain viable when injected into the bloodstream of cancer patients; (ii) our research shows GD-Ts injected into the bloodstream remain
in-situ; and (iii) they persist for up to 100 days after administration. Moreover, we believe we have demonstrated that certain late-stage
blood cancer patients treated with multiple GD-T doses have shown significantly positive responses. These findings lead TCB to believe
that all patients with similar blood cancers may respond to GD-T cell therapy in a positive manner.
Solid
cancers
We
believe that it may be necessary to use CAR-T technology (i) to maximize therapeutic cell homing into the solid tumor site, and (ii)
to increase GD-T cell persistence by ‘tethering’ the cell to antigens present on the cancer cell surface.
In
order to overcome toxicities seen with conventional CAR-T approaches, we believe that we have developed a ‘co-stimulatory’
GD-T CAR which will only attack and kill cancerous cells whilst leaving healthy cells unharmed. This is important as many of the current
conventional CAR-T therapies cannot distinguish target antigens expressed on healthy cells from those on cancerous cells, which results
in various pathologies, including cytokine release syndrome, that in some cases had led to patient death. Such targeting of health cells
with conventional CAR-T makes their use in solid cancers difficult, as too much healthy tissue is likely to be destroyed as ‘collateral’
damage in the treatment process.
The
diagram below illustrates how TCB’s approach works, using the innate receptors on the GD-T cell surface to act as a ‘safety
switch’ – such receptors are generally not triggered by healthy cells, only by disease markers (IPP’s) on the surface
of cancerous or virally infected cells.
| A |
B |
| |
|
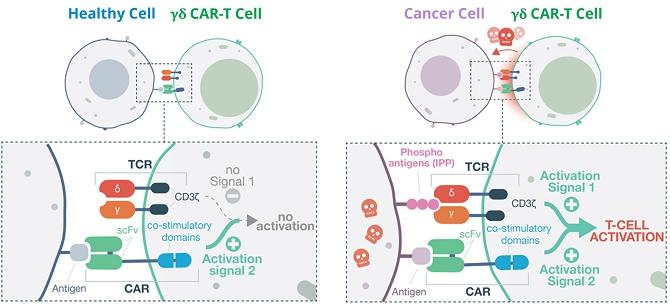 |
Co-stimulatory
CAR-T: A) No GD-T cell activation in healthy cell. B) GD-T activation and cell-killing in cancer cell.
Autologous
cells are derived from ‘self’, using patients own cells to treat their specific disease
Allogeneic
cells are derived from donor material, giving rise to cell banks able to treat numerous patients
Genetically-modified
cells are typically engineered with a ‘chimeric’ receptor to target specific cancer antigens
Commercialization
of conventional CAR-T cell therapy has taken decades of high-quality research in academia and industry, and it has provided transformational
results for a number of patients with B cell malignancies. However, as noted, there are numerous barriers to widespread adoption, including:
| |
● |
Severe
Toxicities. The significant risk of severe toxicities, especially cytokine release syndrome (CRS) and neurotoxicity occurring
up to 3 weeks from treatment. These toxicities have resulted in the need for implementing specific clinical pathways to certify staff
and facilities in the administration of the drugs and the management of the toxicities. |
| |
|
|
| |
● |
On-target,
off tumor toxicities. Conventional CAR-T products have no mechanism for discriminating between diseased and healthy cells. Activation
is governed solely by the expression of the target antigen, which can lead to toxicity when the target antigen is expressed on healthy
cells. In marketed products targeting CD19 (present in the vast majority of B cells), this can be tolerated as B-cell aplasia, albeit
with the need for regular long-term immunoglobulin replacement therapy. However, in experimental CAR-T products targeting other antigens
this has been shown to cause serious side-effects, up to and including fatality. |
| |
|
|
| |
● |
Complex
supply chains associated with autologous treatments. By definition, autologous treatments require the source cells to have been
collected from the patient. It therefore requires a personalized supply chain with multiple touch points and the manufacturing process
can only ever be performed on a single-patient batch size. This adds complexity to each treatment and has required the introduction
of completely new processes and infrastructure in able to commercialize the products. |
| |
|
|
| |
● |
Inherent
variability of the drug product. Each patient has a different cell population and so the starting material of each manufacturing
batch is always variable, leading to variable final product. This can be minimized during pre-screening, which eliminates some patients
from treatment, but there are still significant challenges in manufacturing to provide consistent batches of drug products and in
understanding which variables are critical to product quality. |
| |
|
|
| |
● |
High
list price of the products. The need for personalized manufacturing, new supply chain processes and management of acute and chronic
toxicities have all contributed to the high prices associated with the first CAR-T products reaching the market. In the USA, Kymriah®
has a list price of $475,000 for pediatric ALL, and Yescarta® lists at $373,000 for DLBCL patients. The associated
treatment costs and ongoing management can increase this price significantly. |
The
combination of the co-stimulatory CAR, with GD-T cells, provides TCB with a proprietary platform which we believe addresses the problems
with existing CAR-T products in the following ways:
| |
● |
Using
the natural T cell signaling of the GD-T cell will, we believe, result in less risk of hyperactivation and tonic signaling with an
overall reduction in the risk of CRS and less exhaustion of the cells. |
| |
|
|
| |
● |
The
requirement on cell activation remains on the endogenous GD-T cell TCR signal, which detects stress signals associated with cancerous
cells, so healthy cells are not targeted for destruction even if the target antigen is expressed and the CAR binds, thus off-tumor
toxicity is avoided. |
| |
|
|
| |
● |
Manufacturing
in batches of high dose numbers, without the complex patient collection of personalized supply chain steps, we believe will result
in a dramatic reduction in cost of goods. This will be reflected in a list price which is in line with current biologicals. With
the reduced likelihood of associated toxicities, the treatment and management costs should also be significantly lower, and the products
can be made available to many more patients as a result. |
| |
|
|
| |
● |
The
combination of a well-tolerated product and simplified supply chain (by virtue of our proprietary CryoTC freeze-thaw process), we
believe, will make the therapy suitable for administration in local oncology centers without patients having to locate in centralized
specialist centers of excellence, further reducing financial and logistic barriers to treatment. |
| |
● |
The
tolerance of “off tumor” antigen binding without associated toxicity allows for a complete change in the current target
identification paradigm. Instead of identifying targets that are exclusively expressed on tumor cells, we believe our co-stimulatory
CAR-T approach confers an advantage to select targets that can be highly expressed on tumors and at low levels on healthy tissue.
We select targets based on their relative therapeutic index increase in expression, their homogeneity in tumors and the antigen density.
This allows us to target significantly more tumor associated antigens and to significantly expand the therapeutic index into higher
doses or repeat administration. |
| |
|
|
| |
● |
GD-T
cells have multiple roles in humans, possessing both innate and adaptive functions. One role is a sentinel surveillance cell, and
they are biologically primed to travel through tissue searching for sites of cellular stress. This ability to penetrate tissue makes
them advantageous agents for treating solid tumors. We can add additional function to the GD-T cells by using one or more co-stimulatory
CAR-T constructs to add targeting to appropriate antigen(s) and to provide armor or strategies to overcome environmental and immune
suppression in the tumor microenvironment. Therefore, we believe that the platform offers a promising approach to target the full
spectrum of cancer diseases. |
Viral
infections
GD-Ts
are natural killers of virally infected cells, as well as cancerous cells. We believe that our unmodified GD-T therapy offers substantial
potential as a first line of attack against future viral pandemics. During the COVID-19 pandemic, we took the opportunity to develop
a trial protocol to treat patients with COVID-19, which was approved by the MHRA. We are currently not progressing this trial because
of the absence of available patients given the progression of the disease; however we would consider conducting a phase 1b/2a trial if
more severe/pathogenic variants emerge and we believe that there is considerable opportunity to deploy our GD-T therapy in the treatment
of viral infections, including rapid response treatment of future epidemics and pandemics and selected acute viral infections. Whilst
our current focus is to prioritize cancer treatment we will seek opportunities to develop viral treatments either on our own or in partnership
in future Numerous peer-reviewed publications have demonstrated that GD-T cells innate killers of cells which have become virally infected.
Using Epstein-Barr virus infected cells as an exemplar, TCB has conducted pre-clinical studies to demonstrate that our GMP-compliant
manufacturing process results in GD-T with potent anti-viral cytotoxicity
Autologous
versus allogeneic
Commercially
available cell therapies typically are either autologous or allogeneic. Autologous products are taken from one donor (the patient) and
used to treat that same donor (self-to-self), whilst allogeneic products are usually taken from a single donor (not a patient) and used
as the starting material to treat a large number of different individuals (patients). GD-T lymphocytes are known to exert their biological
effect in a non-MHC restricted manner. This means the potential for graft-versus-host mediated rejection is significantly reduced if
allogeneic (non-self) cells are used as a treatment compared with many other immune cell therapies. As many patients with late-stage
cancer or severe viral infections are also immunosuppressed, potential for host-mediated rejection of allogeneic cells is also reduced.
When compared with autologous variants, commercial benefits of allogenic treatment include the following:
| |
● |
significant
reduction in cost of goods; |
| |
|
|
| |
● |
product
can be campaign manufactured and stockpiled frozen; |
| |
|
|
| |
● |
increased
capacity to treat more patients; |
| |
|
|
| |
● |
logistics
of shipping product are simplified; |
| |
|
|
| |
● |
higher
doses of (reproducible) product are possible; and |
| |
|
|
| |
● |
product
is immediately available for acute disorders |
Our
strategy for developing an allogeneic solution for CAR-T is to select a pathway which will allow us to bring our products to patients
as quickly as possible. These concepts build upon decades of previous development in allogeneic cell therapies and have clear understanding
of development requirements in terms of manufacturing, clinical and regulatory execution.
Although
manufacture of allogeneic cell therapies allows product to be “pharmaceuticalized” by virtue of campaign manufacture and
storage, the approach is however not without technical and logistic challenges. To manufacture allogeneic banks, donor cells need to
be screened for numerous adventitious agents, including for example, HIV, hepatitis, CMV and syphilis. Additional tumorgenicity testing
is required, and assays conducted to ensure the cell bank is free from karyotypic aberrations. In order to overcome any potential for
rejection, TCB has developed allogeneic GD-T cell banks that are unlikely to elicit a graft-versus-host (GvH) or host-versus-graft (HvG)
immune response.
Donors
are screened and selected based on clinically-relevant history and then based on the proliferative capacity and phenotypic character
of their GD-Ts, based on a small volume blood draw and in-house assays. In this way, only good quality GD-T cells are selected for repeat
apheresis and banking. The banks are HLA-typed and become the starting material for all of the allogeneic CAR-T products. These banks
are cryopreserved in our facilities and can later be thawed, genetically engineered with the CAR, activated and expanded into final product,
before being frozen again as multiple individual doses of drug product.
Generation
of Gamma Delta T cells from IPSC cells
Identification
of appropriate donors whilst possible is challenging as only a limited number of batches can be created from a single donation. GD-T
cells can be routinely expanded from peripheral blood over 14 days. This provides a short window of opportunity for cell modification/engineering.
Induced
pluripotent stem cells (iPSCs) have the potential to overcome these issues because they are capable of unlimited proliferation and multidirectional
differentiation. In 2013, several research groups from Japan reported the successful reprogramming of αβT-cells, followed
by re-differentiation back to αβT cells (Vizcardo et al., 2013; Nishimura et al., 2013; Themeli et al.,
2013). While re-differentiated αβT cells-maintained antigen specificity, they were also characterized by higher proliferation
ability than an original T-cell clone.
We
hypothesized that GD-T derived iPSCs cells that carry the rearrangements at the TCRG and TCRD gene locus will be able to generate GD-T
but not αβT cells. Furthermore, iPSC cells will provide a vast opportunity for the gene-editing without any time constraints
of terminally differentiated cells.
Reprogramming
GD-T cells has proven to be a challenge, as these cells are not tolerant of cell sorting. Therefore, GD-T cells can be reprogrammed in
a bulk culture with the rest of peripheral blood cells or at the end of 14 days expansion, when the purity of GD-T is highest. After
several unsuccessful reprogramming attempts, we have optimized the conditions favoring GD-T cells reprogramming. In the last round of
reprogramming >50 clones were created. After extensive analysis of DNA rearrangements in δ- and γ-locus of 5 pre-selected
clones, it was confirmed that they are derived from GD-T cells with different TCR sequences.
IPSC
technology is an attractive approach for the limitless source of GD-T cells are successful progress in reprogramming has been demonstrated.
Further work is now required for the establishment of a GMP compatible T-cell differentiation protocol. Generation of GDT cells from
iPSC cells presents TCB with a vast opportunity for scaling without any time constraints of terminally differentiated cells.
Fresh
versus frozen product
Commercial
and clinical development of cellular therapy products will invariably require cryopreservation and frozen storage of cellular starting
materials, intermediates and/or final product.
Optimizing
cryopreservation is important to obtaining maximum yield and a consistent end-product. Suboptimal cryopreservation can lead not only
to batch-to-batch variation, lowered cellular functionality and reduced cell yield, but also to the potential selection of subpopulations
with genetic or epigenetic characteristics divergent from the original cell line.
Regulatory
requirements also impact on cryopreservation, requiring a robust and reproducible approach to freezing, storage and thawing of the product.
This requires attention to all aspects of the application of low temperatures; from the choice of freezing container and cryoprotectant,
the cooling rate employed and its mode of delivery, correct handling of the frozen material during storage and transportation, to eventual
thawing of the product by the end-user. Each of these elements influences all of the others to a greater or lesser extent and have been
taken into consideration as TCB moves from fresh to cryopreserved cell-based product.
In
a submission to UK regulators, we provided batch manufacture and supporting data, and TCB was granted approval to commence treatment
of cancer patients using frozen allogeneic product. This represents a significant milestone for TCB, as we pioneer use of cryopreserved-donated
cells to treat cancer. Obvious benefits include increased product reproducibility, ability to ship product globally on request and significant
economy of scale (through batch manufacture and storage).
Clinical
studies – unmodified GD-Ts in blood cancer
Management
of acute myeloid leukemia (AML) is based on intensive chemotherapy and/or stem cell transplant, but these therapies lead to high relapse
rates amongst treated patients. Particularly for the relapsed/refractory AML population or those who are not eligible for alloHSCT or
intensive chemotherapy, the therapy options are limited, and patients are often placed in experimental protocol therapies or palliative
care. As a result, there is a need for additional therapies, particularly for these cohorts.
GD-T
cells have emerged as a promising therapy due to their ability to specifically target cancer cells. Nonclinical studies performed in
AML cell lines suggest that GD-T cells specifically target AML tumor cells and lead to cell lysis in vitro (Kirk et al., 1993).
Additionally, in xenotransplantation animal models, GD-T cells obtained from healthy volunteers specifically target AML cells and result
in increased survival and diminished tumor burden in NOD mice (Gertner-Dardenne et al., 2012). Similarly, in vitro experiments
conducted by TCB further support such findings whilst providing evidence that OmnImmune® (TCB002) specifically targets stress induced
cells and effectively kills AML cells lines.
In
the clinic, allogeneic treatment in AML patients in the phase 1b/2a trial OmnImmune® (TCB002) has shown our product is well-tolerated
with some preliminary evidence of anticancer activity. Firstly, there were no signs of graft vs. host disease (GvHD) following therapy
and secondly, CR (complete response) and MLFS (morphologic leukemia free state) were observed. Earlier results with autologous product
demonstrated good tolerability. For the allogeneic product, OmnImmune® (TCB002), additional procedures were included to prevent GvHD
(e.g. AB T cell depletion). Literature reports were also supportive of the use of OmnImmune® (TCB002) in cancer patients. The phase
1b/2a trial tested OmnImmune® (TCB002) in active relapsed or refractory AML who were not eligible for or did not consent to high
dose salvage chemotherapy and/or allogeneic hematopoietic stem cell transplantation (alloHSCT). The trial was conducted to identify a
tolerable dose and better understand the safety of this therapy in the chosen indication as well as generate preliminary information
on potential clinical benefit. The primary, secondary and exploratory endpoints were as follows:
Primary
endpoints:
| |
● |
Assessment
of adverse events (Aes) graded by Common Terminology Criteria for Adverse Events (CTCAE) v5.0, vital signs and evaluation of laboratory
parameters |
| |
|
|
| |
● |
Incidence
of dose-limiting toxicities (DLTs) during the first 28 days after γδ T cell administration. |
| |
|
|
| |
● |
Establish
Maximum Tolerated Dose (MTD) of OmnImmune® |
Secondary
endpoints:
| |
● |
Complete
Remission (CR) rate |
| |
● |
Overall
survival (OS) |
| |
|
|
| |
● |
Quality
of life determined by EORTC QLQ-C30 questionnaire |
Exploratory
endpoints:
| |
● |
Changes
in γδ T cell count and phenotype before and after OmnImmune® infusion |
No
formal statistical analysis was planned. The incidence of DLTs were to be summarized descriptively by γδ T cells dose for
evaluable patients. The recommended dose would be determined as the greatest with an incidence of DLTs no greater than 1/3. All other
data including efficacy results were summarized descriptively by γδ T cells dose.
The
trial enrolled 8 patients and healthy donors aged >18 years.
Clinical
outcome
Seven
patients were treated with OmnImmune® (TCB002). The eighth patient could not be dosed because the study was terminated as a result
of the COVID-19 pandemic, which prevented the importation of investigational product from Scotland to the Czech Republic. No safety concerns
were raised during Safety Review Committee (SRC) meetings. No treatment related Serious Adverse Reactions (SARs) were reported in any
of the patients who were enrolled in the trial. No grade 3≥ OmnImmune® (TCB002) treatment related toxicities were noted in any
of the treated patients. No dose-limiting toxicities were observed and no emergency safety measures have occurred for any subjects receiving
OmnImmune® (TCB002). Two patients at 28 days post-treatment achieved a CR (one patient) or MLFS (one patient); another patient was
classified as attaining stable disease with > 50% reduction in bone marrow blast count; one additional patient exhibited reduction
in blast levels at 14 days; and one patient had disease progression (see table below). One patient (PRA1-5003) died 21 days after TCB002
due to bilateral pneumonia, determined unrelated to study medication. One patient (PRA1-5010) was withdrawn because of the COVD-19 pandemic
before bone marrow aspiration on day 28 post-treatment. These preliminary indications of anticancer activity were not expected given
the refractory profile of the enrolled patients.
The
EORTC QLQ-C30 questionnaire resulted in scoring from six of the seven patients dosed with OmnImmune® (TCB002) for varying periods
of time depending on their study duration. At 7 days post dosing, the average QoL score from six patients had decreased from 55.7 to
47.2 out of a possible maximum of 100. This negative impact on QoL reflects the well characterized side effects of preconditioning therapy
with cyclophosphamide and fludarabine given between 6 and 2 days prior to OmnImmune® (TCB002) administration. The score remained
lower in the four patients assessed at 28 days at a level of 50.0. In the two patients (one CR and one MLFS) who were assessed at the
end of the study (week 24), both had recovered to an improved QoL score, each of 67.0.
FDA
Orphan Drug Designation
About
60 million people living in the European Union (EU) and USA suffer from a rare disease. The European Medicines Agency (EMA) and FDA play
a central role in facilitating the development and authorization of medicines for rare diseases, which are termed ‘orphan medicines’
in the medical world. Developing medicines intended for small numbers of patients has little commercial incentive under normal market
conditions. Therefore, the EU and USA offer a range of incentives to encourage the development of designated orphan medicines.
The
general therapeutic strategy for the treatment of AML has not changed substantially over the past 30 years. Excluding APL (which should
be treated with trans-retinoic acid), AML management is based primarily on induction, incorporating an anthracycline and cytarabine,
and consolidation therapy, and/or allogeneic Hematopoietic Stem Cell Transplantation (alloHSCT). Induction/consolidation therapy leads
to high CRs rates in those who are eligible for treatment and present a favorable risk profile.
Several
novel agents are in various stages of development for the treatment of AML. Novel approaches include antibody-based immunotherapy and
adoptive cell therapy that aim to improve anti-leukemia T cell function, such as the therapies developed by TCB (OmnImmune®).
OmnImmune®
(TCB002) was initially studied in patients with active relapsed or refractory AML who are not eligible or do not consent to high dose
salvage chemotherapy and/or alloHSCT. In July 2019, OmnImmune® (TCB002) was granted ‘orphan medicine’ status from the
FDA for Acute Myeloid Leukemia (AML). TCB intends to conduct a further clinical phase 2/3 study (OmnImmune® (TCB008-001)) in 2021/2
aimed at treating earlier stage AML patients.

Summary
of TCB’s phase 1b/2a clinical trial in patients with fourth-line-of-treatment acute myeloid leukemia. Subsequent to the completion
of this study TCB commenced phase 2b into 3 (pivotal) patient treatment during H1, 2022.
Pipeline
and plan
Our
future pipeline is focused on treating liquid cancers with our unmodified GD-T therapies and the treatment of solid cancers with next-generation
allogeneic GD-T CAR-T therapies.
Our
unmodified cell therapy, used in the treatment of Acute Myeloid Leukemia, is supplied under the name OmnImmune.
OmnImmune®
is an allogeneic unmodified GD-T (GD-T2) cell product. Donor-derived GD-T cells for proliferative capacity, were activated and
expanded in our manufacturing facility before being infused into the patient as part of our OmnImmune® (TCB002) phase 1 trial.
This trial was completed in H1 2020 at the Institute of Hematology and Blood Transfusion in Prague, Czech Republic. Having generated
meaningful clinical data showing our product is well-tolerated in late-stage AML patients with no remaining treatment options, TCB
commenced a phase 2b-into pivotal (phase 3) clinical studies (with OmnImmune®) during 2022 in AML patients who have failed to
respond adequately to induction therapy. The aim is to provide a form of salvage therapy which will either stabilize the patient,
thereby preventing disease progression, or delay the requirement for human stem cell transplant. Our initial trial centers are
in the UK. Working on the premise that other blood cancers should respond
to GD-Ts in a similar manner to AML, TCB plans to conduct clinical studies for OmnImmune® in other hematological malignancies in
future. The initial phase 1b/2a trials were undertaken using fresh cell-based product under the program number TCB002. For ease of
reference, when discussing that specific trial, we refer the program as OmnImmune® (TCB002). The subsequent planned phase
2b-into pivotal (phase 3) clinical studies uses a frozen cell-based product under the program number TCB008-001. When discussing
that specific trial, we refer the program as OmnImmune®.
We
plan to develop a range of allogeneic co-stimulatory GD-T CAR pre-clinical drug candidates which will target antigens expressed on a
number of solid tumor types.
TCB
has generated in-vitro preclinical data as part of our CAR-T program which demonstrated that GD-Ts are very high purity and can be CAR-transduced
with high efficiency (see diagram below). Gamma delta cell purity and transduction efficiency have been measured using flow cytometry.
CAR positive cells were measured by a detection reagent labelled with the fluorophore Phycoerythrin (PE). Flow cytometry analysis used
the parameters of side scatter height (SSC-H) and PE area (PE-A) to define the cell populations. This is demonstrated in the figure below
comparing non-transduced (NTD) and transduction with a co-stimulatory CAR construct (co-stim CAR).
We
have also demonstrated that following transduction with different CAR constructs, GD-T’s can be effectively and reproducibly expanded
in-vitro whilst exhibiting increased cytotoxicity in a zoledronate-dependent manner (see diagrams below – zoledronate-dependency
reflects TCB’s proprietary process for commercial expansion of GD-T’s). The CAR constructs contained different endodomains
including DNAX-activating protein 10 (DAP-10) and the high affinity IgE receptor (FcR) with no endodomain (no-endo) and non-transduced
(NTD) as controls. These data outline the key preclinical parameters investigated in advance of progressing our CAR-T products into clinical
trials. TCB has engaged with UK regulators to discuss the design of GD-T CAR phase1b/2a clinical studies (specifically relating to patient
dosing and quality systems).
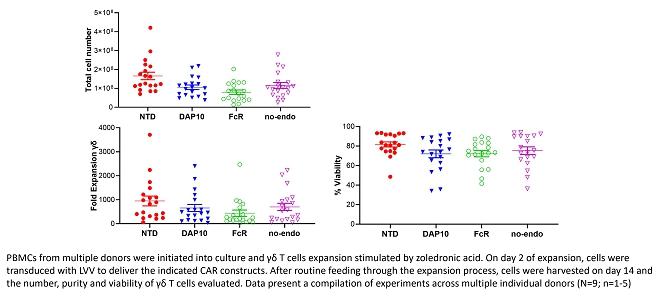
Peripheral
blood mononuclear cells (PBMCs) were initiated into culture and GD-T cells expansion stimulated by zoledronic acid. On day 2 of expansion,
cells were transduced with lentiviral vectors (LVV) to deliver the indicated CAR constructs. After routine feeding through the expansion
process, cells were harvested on day 14 and the total cell number, fold expansion and viability of GD-T cells evaluated. Data present
a compilation of experiments across multiple individual donors (N=9; n=1-5).
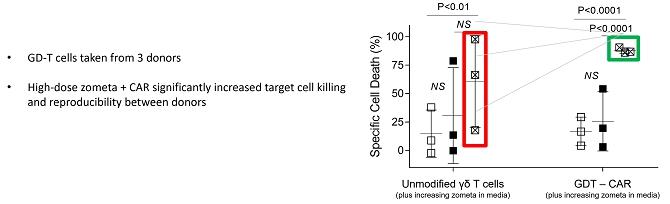
Manufacturing
Unlike
many pre-clinical and early clinical stage biotech companies that rely on outsourcing key manufacturing and development functions with
consequent complex and expensive supply chains and delays in delivery and execution, we have built a world-class fully integrated GMP
grade specialist GD-T manufacturing center in Glasgow, Scotland. This facility undertakes all key functions associated with our GD-T
cell development, testing, quality assurance, product manufacture, clinical trial recruitment, management design, support and interaction
with regulators. This has resulted in rapid, focused development; highly efficient cost control;, controlled supply chain; speed of development
and clinical delivery. We employ over 80 highly qualified people at our facility. The inspiration to create a fully integrated facility
came from our founders’ vision and considerable experience in cell therapy.
All
advanced therapy medicinal products in the UK must be manufactured by law under a manufacturer’s license granted by the MHRA.
TCB received its Manufacturer’s Authorisation for Investigational Medicinal Products MIA (IMP) from the MHRA in January 2015
(license number MIA (IMP) 42803). In April 2016, the MHRA granted the ‘Specials’ license to TCB as well as approving the
facility for ongoing GMP compliance, which permits the manufacture and release of Advanced Therapy Medicinal Products (ATMPs) for use
in clinical trials.
The
backbone of our company is TCB’s Quality Management System, which TCB based on the principles of the current GMP as described in
the ‘Rules and Guidance for Pharmaceutical Manufacturers and Distributors’ and EudraLex Volume 4 as revised. This is achieved
by the application of a Quality Management System based around the requirements of ICH Q10 and the EU GMP Guide, which address factors
affecting the desired quality, namely the personnel, facilities, equipment, materials, processes, procedures training, vendor selection
and approval, User Requirement Specification (URS) qualification and validation of assays and systems and the record keeping. All personnel
joining TCB undergo rigorous training on everything from GMP through to formalized systems for measuring and evaluating risk.

*MHRA
approved GMP compliant (last inspection December 2020 – observations only)
TCB’s
manufacturing facilities are equipped with two Class B clean rooms with space secured for future expansion for production of our products
as it progresses from phase I to phase III clinical trials. The facility is also equipped with Development and Quality control testing
laboratories together and ample stores for goods inwards and product release plus storage for intermediate and final product. Equipment
is controlled and monitored through a Management Information System with 24/7 monitoring. All laboratory equipment undergoes a formal
URS and once installed undergoes full qualification prior to it being put into routine use.
TCB’s
Quality Control team are responsible for the majority of release testing for our products. The Quality Control departments (analytical
and microbiology) are responsible for product characterization using bespoke phenotyping and potency assays, safety testing assays and
final release of the product to the clinic. In-house testing within TCB’s Quality Control laboratories eliminates the necessity
for third party involvement, resulting in reduced costs and gaining full control of scheduling. The Quality Control departments remit
also extends to the microbiological monitoring of the facility to measure, assess and control the exceptionally high levels of sterility
required within the aseptic manufacturing suites. Extensively equipped Quality Control microbial laboratories allow environmental monitoring
of the manufacturing cleanrooms to GMP standards. The laboratories house incubators, biological safety cabinets, centrifuges, fridges,
freezers, air and particle monitors.
Competition
The
biotechnology and pharmaceutical industries are characterized by rapidly advancing technologies, intense competition and a strong emphasis
on proprietary products. While we believe that our scientific knowledge, technology and development experience provide us with competitive
advantages, we face potential competition from many different sources, including major pharmaceutical, specialty pharmaceutical and biotechnology
companies, academic institutions, governmental agencies and public and private research institutions. Any GD-T cell therapeutic candidates
that we successfully develop and commercialize will compete with existing products and new products that may become available in the
future. We believe our advanced clinical products in allogeneic and unmodified GD-Ts provide us a first mover advantage in oncology and
viral indications. Our continued efforts in advancing our modified platform technologies, along with our efforts in banked GD-Ts, are
in direct competition with a number of public and private companies in the cell therapy space.
TC
BioPharm is part of a growing number of companies commercially active in the cellular immunotherapy space. Such companies developing
call-based products include AdicetBio who presented phase 1 data from patients with non-Hodgkin lymphoma using allogeneic
GDT-1’s modified with a CAR against CD20 – published initial results documented safety and CR/PR in some patients. Other
companies include Allogene who are using various CAR-modified allogeneic alpha-beta cells to treat non-Hodgkin lymphoma, multiple
myeloma, acute myeloid leukemia and renal cell carcinoma; Autolus who are conducting clinical studies using autologous alpha-beta T
cells using CAR’s directed against either CD19/22 or CD20, listed indications include acute lymphoblastic leukemia, B cell
non-Hodgkin lymphomas and chronic lymphoblastic leukemia; and In8Bio who have several ongoing phase 1 and 2 clinical programs
including an autologous DRI resistant to alkylating agents for treating glioblastoma and an allogeneic product for treating patients
with acute leukemia, In8Bio also have a bespoke GDT therapy for AML derived from hematopoietic stem cell transplantation. Apart from
TCB who manufacture cell-based product in-house, we believe the above companies contract-out GMP formulation of clinical product to
third-party organizations.
Commercial
leaders in the CAR-T space are Novartis AG (Basel, Switzerland) and Gilead (Foster City, CA). Both of these companies market autologous
CD19-targeted AB CAR-T products. Bristol Myers Squibb (New York, NY) has obtained approval for its CD19-directed CAR-T. Via third party collaborations, all three of the commercial leaders in autologous CAR-T, have accessed gene editing technology
with a view to creating allogeneic products. Novartis have partnered with Intellia (Cambridge, MA), Gilead have an agreement with Sangamo
Therapeutics (Richmond, CA) and Bristol Myers Squibb have an agreement with Editas Medicine (Cambridge, MA).
We
do not believe that any of these competitors will offer the same commercial proposition as our GD-T cell therapeutic candidates due to
our:
| |
● |
Ownership
of foundation IP of the co-stimulatory CAR technology within GD-Ts. |
| |
|
|
| |
● |
First-mover
advantage in the field of modified GD-Ts as therapeutics. |
| |
|
|
| |
● |
Ability
to GMP manufacture large numbers of modified GD-T cells to a high purity in a cost-effective manner. |
| |
|
|
| |
● |
Established
banks of allogeneic products which may be used in future (following appropriate regulatory approvals) to treat both cancer and severe
viral disease. |
| |
|
|
| |
● |
The
potential to create CAR-T therapies with significantly improved safety profile, suitable for widespread market adoption. |
| |
|
|
| |
● |
Experience
of, and in-house management of, our clinical trial programs. |
| |
|
|
| |
● |
Pipeline
development strategy and screening tools to develop a deep pipeline of platform products for a range of diseases. |
Our
Strengths
Our
clinical trials have provided very strong evidence of drug-toleration and some preliminary evidence of clinical benefit.
Our
clinical trial of TCB001 involved treatment of patients with autologous unmodified GD-Ts. In a phase 1b/2a dose-ranging safety study
(maximum total dose 30x109 cells) we saw no evidence of drug-related severe adverse events. A total of eight patients were
treated with escalating doses of TCB001, and no treatment-related toxicities were reported during the full six-week therapeutic course.
Data from OmnImmune® (TCB002) suggests an excellent tolerability, with no observed Host versus Graft Disease (HvGD) and some preliminary
indication of clinical benefit. OmnImmune® (TCB002) has been granted Orphan Drug Designation by the FDA.
Our
CAR-T platform is centered on development of safer and more widely applicable therapeutic candidates and associated process and manufacturing
capabilities.
Our
proprietary co-stimulatory CAR-T technology platform covers identification of target cancer antigens, successful design and engineering
of target sequences, preclinical safety testing and optimized manufacturing processes suitable for producing therapeutic candidates for
use in clinical trials and commercialization. We believe the platform will enable development of additional GD-T cell therapeutic candidates
targeting cancers that have previously been difficult to treat. We believe the products will be demonstrably safer than the current generation
of AB T cell CAR-T products because they will not attack healthy non-cancerous cells and augment the natural biological process rather
than bypassing it.
We
have identified a large and growing pool of cancer targets for which we can develop additional therapeutic candidates.
We
have identified over 20 antigens that are preferentially expressed in cancer cells and have established ongoing research programs to
develop several of these into our GD-T platform. Within the terms of our agreement, bluebird bio, we have first right of refusal on a
further three oncology targets. Each antigen target presents an opportunity to target many cancer types and therefore presents multiple
potential represents a development, collaboration and/or an out-licensing opportunity as each target could be used to target specific
cancer types. Growing the pipeline of products built on our co-stimulatory CAR-T and reaching patients is our priority.
We
have historically entered collaborative arrangements with partners (bluebird bio, Inc (now 2seventybio). (USA) and Nipro Corporation
(Japan), which involve funded or partly funded preclinical collaboration. It is uncertain at this time whether TCB will receive any significant
revenues from these collaborations.
We
retain control of key business elements, such as product manufacture and clinical research.
Whilst
many companies contract out product manufacture, quality systems and clinical trial management, we have elected to build these skills
in-house. TC BioPharm has a GMP (Good Manufacturing Practice) cleanroom facility where our products are manufactured. We also retain
all the quality support systems such as product testing and release of final product to the clinic. Keeping these systems in-house allows
the Company to control all aspects of the manufacturing process whilst significantly reducing costs of goods (CoGs). Further saving on
costs are accrued by in-house manufacture, as contract manufacturing organizations (CMOs) will typically charge several times more than
the actual costs to maintain their profit margins. Rather than fully outsource our clinical trial management, data management and pharmacovigilance,
we maintain an inhouse clinical team that partners with a contract clinical research organization (CRO) for data management and pharmacovigilance
services. The inhouse clinical team conducts and manages our own clinical trials in-house. In addition to significant cost savings, this
allows us to build a strong working relationship with physicians who are treating the cancer patients; we believe this is key to successful
product development as the physicians participating in our clinical studies will also be our future customers. We believe that retaining
control of key elements of our business such as GMP manufacture and clinical operations, has allowed TC BioPharm to move quickly and
efficiently since incorporation.
We
continue to file new patent applications from new in-house product development, and have a strong growing intellectual property portfolio
to protect our products and proprietary platform.
We
have a strong intellectual property portfolio covering the key aspects of our manufacturing processes and product platforms. Our in-house
product development team are dedicated to developing new therapeutic candidates and optimizing current
manufacturing processes. All of our patent families are currently in various stages of the patent approval process, and as leaders in
the path towards the commercialization of GD-Ts we hold significant first-mover advantage captured by trade secrets and know-how.
Our
policy of developing strategic alliances has and will provide additional support for product development and commercialization.
We
believe that strategic alliances, both historic and potential future alliances, have and will provide extensive experience in scale-up
and automation, culture media manufacture and post-authorization sales and marketing with regional expertise. Additionally, we expect
to use knowledge gained from our collaborations to improve development pathways for our unpartnered CAR-T therapeutic candidate programs.
We
have a highly knowledgeable and experienced management team with extensive industry experience and expertise in the United States and
in Europe.
Mr.
Kobel joined us as our Chief Executive Officer at the time of our IPO. Bryan brings a US presence to our executive team and over 15 years’
experience in Healthcare and Life Sciences capital markets. Martin Thorp, our Chief Financial Officer has over 40 years’ experience
in implementing capital strategies globally from seed investment to IPO. He was global CEO of Arthur Andersen Corporate Finance based
in New York.
Ability
to treat patients under the ‘Specials’ regulatory framework.
European
regulations (Regulation 167 of the Human Medicines Regulations 2012) set out the exemption from the requirement for a medicinal product,
placed on the market in the UK to hold a marketing authorization. This exemption flows from Article 5(1) of EU Directive 2001/83/EC,
which states that a member of the EU may, in accordance with legislation in force and to fulfil special needs, excludes from the provisions
of this Directive medicinal products supplied in response to a bona fide unsolicited order, formulated in accordance with the specifications
of an authorized healthcare professional and for use by an individual patient under his or her direct personal responsibility. Such an
unlicensed medicinal product may only be supplied in order to meet the special needs of an individual patient. An unlicensed medicinal
product should not be supplied where an equivalent licensed medicinal product can meet the special needs of the patient. Responsibility
for deciding whether an individual patient has “special needs” which a licensed product cannot meet should be a matter for
the doctor, dentist, nurse independent prescriber, pharmacist independent prescriber or supplementary prescriber responsible for the
patient’s care.
In
terms of time and cost, the ‘Specials’ scheme is an attractive strategy. We believe that accumulating evidence by this route
could lead to rapid and wider product uptake through ‘off-label’ use.
Intellectual
Property
We
have a strong portfolio of patents covering manufacture and commercialization of GD-T cell products and their modification via CAR-T
(summarized below). Our technology platform and clinical programs have enabled us to raise over $100 million in grant, equity and collaboration
funding since becoming operational in 2017. This financing has allowed us to enhance and expand our clinical and preclinical programs
as well as build our team of world-class scientists.
The
following table provides an overview of our core technology platforms, technology assets and competencies across the business. Additional
details of our intellectual property portfolio are provided below.
| ASSET
SUMMARY |
|
ATTRIBUTES |
| |
|
|
| GD-T
Vehicle |
|
● |
Readily
available and expanded to high numbers. |
| |
|
● |
Not
MHC-restricted, therefore no graft vs host disease – an allogeneic platform. |
| |
|
● |
Pre-programmed
tropism for infiltration of diseased tissue. |
| |
|
● |
Multiple
modes of innate cytotoxicity and coordinating a wider immune response. |
| |
|
● |
Clinical
tolerability of the allogeneic vehicle demonstrated at high dose level. |
| |
|
● |
Naturally
arising in different subtypes offering a menu of vehicles with unique properties. |
| Allogeneic
Cell Banks |
|
● |
Donor
GD-Ts selection based on highest therapeutic quality. |
| |
|
● |
Reproducible
product with low cost-of-goods compared with autologous (patient-bespoke) therapies, can be frozen-shipped, thawed at clinic. |
| |
|
● |
Well
understood clinical and regulatory pathway to commercialization. |
| |
|
|
|
| Co-stimulatory
CAR-T |
|
● |
Elimination
of off-tumor toxicity. |
| |
|
● |
Reduction
of cytokine release from killing healthy cells. |
| |
|
● |
Reliance
on natural T cell activation and no tonic signaling |
| |
|
● |
Antigen
expression on healthy tissue tolerated – greatly expanded range. |
| |
|
● |
Ability
to use multiple co-stimulatory receptors to add functionality. |
| |
|
|
|
| Integrated
Business Model |
|
● |
Full
control of critical stages of development projects, which increases speed and reliability of development and production, optimizes
operations to our specialized products and materially reduces our cost base |
| |
|
● |
No
pass-through or transaction costs form external service providers, which increases efficiency and speed of development and manufacturing
and materially reduces our cost base |
| |
|
● |
In-house
clinical management ensures best chance of clinical success and avoids use of very expensive clinical management in early-stage trials,
materially reducing our cost base. |
The
strength of our patents involves complex legal and scientific questions and can be uncertain.
We
actively seek to protect the intellectual property and proprietary technology that we believe is important to our business, including
seeking, maintaining, enforcing and defending patent rights for our therapeutics and processes, whether developed internally or licensed
from third parties. Our success will depend on our ability to obtain and maintain patent and other protection including data/market exclusivity
for our therapeutic products and platform technology, preserve the confidentiality of our know-how and operate without infringing the
valid and enforceable patents and proprietary rights of third parties.
Our
policy is to seek to protect our proprietary position generally by filing an initial priority filing at the U.K. Intellectual Property
Office, or UKIPO. This is followed by the filing of a patent application under the Patent Co-operation Treaty claiming priority from
the initial application(s) and then progressing to national applications in, for example, the United States, Europe, Japan, Australia,
New Zealand, China and Canada. In each case, we determine the strategy and territories required after discussion with our patent professionals
to ensure that we obtain relevant coverage in territories that are commercially important to us and our GD-T therapeutic candidates.
We will additionally rely on data exclusivity, market exclusivity and patent term extensions when available, including as relevant exclusivity
through orphan or pediatric drug designations. We also rely on trade secrets and know-how relating to our underlying platform technology
and therapeutic products. Prior to making any decision on filing any patent application, we consider, with our patent professionals,
whether patent protection is the most sensible strategy for protecting the invention concerned or whether the invention should be maintained
as confidential.
As
of March 22, 2024, we own 16 granted patents and 11 patent applications in 3 families, and have an exclusive license to an additional
1 family of 14 granted patents and 8 patent applications. Consistent with the filing strategy outlined above, all of our applications
are either UK applications, PCT applications or national phase applications derived from a corresponding PCT application. All sets of
national phase applications include a US application / patent. These patents and patent applications include claims directed to our therapeutic
products and platform technology or other manufacturing and process technology to further enable our therapeutic products and manufacturing
methods.
WO
2016/166544 (Modified Gamma Delta T Cells and uses thereof). International filing date April 14, 2016, earliest priority date April 15,
2015.
We
own patents and patent applications covering a method of treatment for cancer using GD-T cells that express chimeric antigen receptors
(CARs). The patent application claims are directed to GD-T cells expressing a co-stimulatory CAR, the advantages of the co-stimulatory
CAR by inhibiting on-target, off-tumor activation due to its design, to the method and process of modifying a GD-T cell to express the
co-stimulatory CAR, and to medical uses of the modified GD-T cell. A US patent application has issued as a granted patent in the US (US
10881688 B2, expires October 7, 2036 in view of USPTO calculated Patent Term Adjustment (PTA)). Applications have been granted in Australia
(AU 2016250211), China (ZL 201680034862.3) Israel (IL 255011), and Japan (JP 6995624) and at the Eurasian Patent Office. National applications
remain pending in: Australia, Hong Kong, and the US. A regional patent application is pending before the European Patent Office.
WO
2016/174461 (T cell which expresses a Gamma Delta T cell receptor and a Chimeric Antigen Receptor). International filing date April 29,
2016, earliest priority date April 30, 2015. Expiry date will be April 28, 2036 in most jurisdictions. Further patent term adjustments
may apply in the US.
We
are the exclusive licensee of patents and patent applications owned by UCL Business plc covering a method of treatment using a T cell
which expresses a Gamma Delta T cell receptor and a chimeric antigen receptor. The patent application claims are directed to GD-T cells
expressing a co-stimulatory CAR, the advantages of the co-stimulatory CAR by inhibiting on-target, off-tumor activation due to its design,
to the method and process of modifying a GD-T cell to express the co-stimulatory CAR, and to medical uses of the modified GD-T cell.
National applications have been granted in Australia (AU 2016255611, expires April 28, 2036), Canada (CA2982532), Japan (JP 6986449),
Israel (IL 255186), South Africa (ZA 2017/06923), and via the Eurasian regional patent (EA 041081) in Armenia, Azerbaijan, Belarus, Kyrgyzstan,
Kazakhstan, Russia, Tajikistan, Turkmenistan. Additional national applications are pending in: Brazil, China, Hong Kong, South Korea,
New Zealand, Singapore, and the US. A regional patent application is pending before the European Patent Office.
WO
2016/005752 (Gamma Delta T cells and uses thereof). International filing date July 8, 2015, earliest priority date July 9, 2014. Expiry
date for the patent granted in Israel will be July 7, 2035.
We
own an Israeli patent and Japanese patent applications covering the method of preparing and using GD-T cells in the allogeneic treatment
of subjects suffering from viral infection, fungal infection, protozoal infection or cancer. The patent application claims are directed
to the process of providing GD-T cells from a first subject to a second subject (allogeneic transfer).
WO
2019/064030 (Modified CAR-T). International filing date October 1, 2018, earliest priority date September 29, 2017.
We
own a US Patent (US11890302) and patent applications covering chimeric antigen receptors comprising an intracellular signaling domain
derived from GD-T cell surface receptors. When expressed in GD or natural killer (NK) cells, the resultant CAR-T cells exhibit improved
cytotoxicity. National applications are pending in: Hong Kong, Israel, Japan, and South Korea,. A regional patent application is pending
before the European Patent Offices.
Platform
technology patent applications – We have several other patent applications in the process of being drafted, improving T cell efficacy
by modulating PD1 expression, additional methods of expanding GD-T1 gamma-delta T cell populations and improvements to our GD CAR-T platform
Government
Regulation and Product Approval
As
a biopharmaceutical company, we are subject to extensive regulation. Our product candidates, if approved, will be regulated as biological
medicines. With this classification, commercial production of our products will need to occur in registered and licensed facilities in
compliance with current Good Manufacturing Practices, or cGMPs, for biologics.
Human
immunotherapy products are a new category of therapeutics. The FDA categorizes human cell- or tissue-based products as either minimally
manipulated or more than minimally manipulated and has determined that more than minimally manipulated products require clinical trials
to demonstrate product safety and efficacy and the submission of a Biologics License Application, or BLA, for marketing authorization.
Government
authorities in the United States (at the federal, state and local level) and in other countries and jurisdictions, including the European
Union, extensively regulate, among other things, the research, development, preclinical and clinical testing, manufacturing, quality
control, labeling, packaging, storage, record-keeping, promotion, advertising, sale, distribution, post-approval monitoring and reporting,
marketing and export and import of biopharmaceutical products such as those we are developing. Our product candidates must be approved
by the FDA before they may be legally marketed in the United States and by the appropriate foreign regulatory agency before they may
be legally marketed in foreign countries. Generally, our activities in other countries will be subject to regulation that is similar
in nature and scope as that imposed in the United States, although there can be important differences. Additionally, some significant
aspects of regulation in Europe are addressed in a centralized way, but country-specific regulation remains essential in many respects.
The process for obtaining regulatory marketing approvals and the subsequent compliance with applicable federal, state, local and foreign
statutes and regulations require the expenditure of substantial time and financial resources.
United
States Product Development Process
In
the United States, the FDA regulates biological products under the Public Health Service Act, or PHSA, and the Federal Food, Drug and
Cosmetic Act, or FDCA, and implementing regulations. Products are also subject to other federal, state and local statutes and regulations.
The process of obtaining regulatory approvals and the subsequent compliance with appropriate federal, state, local and foreign statutes
and regulations require the expenditure of substantial time and financial resources. Failure to comply with the applicable U.S. requirements
at any time during the product development process, approval process or after approval, may subject an applicant to administrative or
judicial sanctions. FDA sanctions could include, among other actions, refusal to approve pending applications, withdrawal of an approval,
a clinical hold, warning letters and similar public notice of alleged non-compliance with laws, product recalls or withdrawals from the
market, product seizures, total or partial suspension of production or distribution, fines, refusals of government contracts, restitution,
disgorgement of profits, or civil or criminal penalties. Any agency or judicial enforcement action could have a material adverse effect
on us. The process required by the FDA before a biological product may be approved for marketing in the United States generally involves
the following:
| |
● |
completion
of preclinical laboratory tests and animal studies according to Good Laboratory Practices, or GLPs, and applicable requirements for
the humane use of laboratory animals or other applicable regulations; |
| |
|
|
| |
● |
submission
to the FDA of an Investigational New Drug Application, or IND, which must become effective before human clinical trials may begin; |
| |
|
|
| |
● |
performance
of adequate and well-controlled human clinical trials according to the FDA’s regulations commonly referred to as Good Clinical
Practices, or GCPs, and any additional requirements for the protection of human research subjects and their health information, to
establish the safety and efficacy of the proposed biological product for its intended use; |
| |
|
|
| |
● |
preparation
and submission to the FDA of a Biologics License Application, or BLA, for marketing approval that includes substantive evidence of
safety, purity, and potency from results of nonclinical testing and clinical trials; |
| |
|
|
| |
● |
satisfactory
completion of one or more FDA inspections of the manufacturing facility or facilities where the biological product is produced to
assess compliance with cGMP to assure that the facilities, methods and controls used in product manufacture are adequate to preserve
the biological product’s identity, strength, quality and purity and, if applicable, the FDA’s current Good Tissue Practices,
or GTPs, for the use of human cellular and tissue products; |
| |
|
|
| |
● |
potential
FDA audit of the nonclinical study and clinical trial sites that generated the data in support of the BLA; |
| |
|
|
| |
● |
payment
of user fees for FDA review of the BLA; and |
| |
|
|
| |
● |
FDA
acceptance, review and approval, or licensure, of the BLA, which might include review by an advisory committee, a panel typically
consisting of independent clinicians and other experts who provide recommendations as to whether the application should be approved
and under what conditions. |
Before
testing any biological product candidate, including our product candidates, in humans, the product candidate must undergo rigorous preclinical
testing. Preclinical tests, also referred to as nonclinical studies, include laboratory evaluations as well as in vitro and animal studies
to assess the potential safety and efficacy of the product candidate. After sufficient preclinical testing has been conducted, the conduct
of the preclinical tests must comply with federal regulations and requirements including GLPs. The clinical trial sponsor must submit
an IND to the FDA before clinical testing can begin in the United States. An IND must contain the results of the preclinical tests, manufacturing
information, analytical data, any available clinical data or literature, a proposed clinical protocol, an investigator’s brochure,
a sample informed consent form, and other materials. Clinical trial protocols detail, among other things, the objectives of the clinical
trial, dosing procedures, subject selection and exclusion criteria, and the parameters to be used to monitor subject safety, including
stopping rules that assure a clinical trial will be stopped if certain adverse events should occur. Each protocol and any amendments
to the protocol must be submitted to the FDA as part of the IND. Some preclinical testing, such as toxicity studies, may continue even
after the IND is submitted.
The
IND automatically becomes effective 30 days after receipt by the FDA, unless the FDA raises concerns or questions regarding the proposed
clinical trials or places the trial on a clinical hold within that 30-day time period. In such a case, the IND sponsor and the FDA must
resolve any outstanding concerns before the clinical trial can begin. The FDA may also impose clinical holds on a biological product
candidate at any time before or during clinical trials due to safety concerns or non-compliance. If the FDA imposes a clinical hold,
trials may not recommence without FDA authorization and then only under terms authorized by the FDA. Accordingly, we cannot be sure that
submission of an IND will result in the FDA allowing clinical trials to begin, or that, once begun, issues will not arise that suspend
or terminate such trials.
Further,
each clinical trial must be reviewed and approved by an independent institutional review board, or IRB, at or servicing each institution
at which the clinical trial will be conducted. An IRB is charged with protecting the welfare and rights of trial participants and considers
such items as whether the risks to individuals participating in the clinical trials are minimized and are reasonable in relation to anticipated
benefits. The IRB also approves the form and content of the informed consent that must be signed by each clinical trial subject or his
or her legal representative and must monitor the clinical trial until completed. Clinical trials involving recombinant or synthetic nucleic
acid molecules also must be reviewed by an institutional biosafety committee, or IBC, a local institutional committee that reviews and
oversees basic and clinical research conducted at that institution. The IBC assesses the safety of the research and identifies any potential
risk to public health or the environment.
Clinical
trials involve the administration of the biological product candidate to healthy volunteers or patients under the supervision of qualified
investigators, generally physicians not employed by or under the trial sponsor’s control. Clinical trials must be conducted and
monitored in accordance with the FDA’s regulations comprising the GCP requirements, including the requirement that all research
patients provide informed consent.
Human
clinical trials are typically conducted in three sequential phases that may overlap or be combined:
| |
● |
Phase
1. The biological product is initially introduced into healthy human subjects and tested for safety. In the case of some products
for severe or life-threatening diseases, especially when the product may be too inherently toxic to ethically administer to healthy
volunteers, the initial human testing is often conducted in patients with the target disease or condition. |
| |
|
|
| |
● |
Phase
2. The biological product is evaluated in a limited patient population to identify possible adverse effects and safety risks,
to preliminarily evaluate the efficacy of the product for specific targeted diseases and to determine dosage tolerance, optimal dosage
and dosing schedule. |
| |
|
|
| |
● |
Phase
3. Clinical trials are undertaken to further evaluate dosage, clinical efficacy, potency, and safety in an expanded patient population,
generally at geographically dispersed clinical trial sites. These clinical trials are intended to generate enough data to statistically
evaluate the efficacy and safety of the product for approval, to establish the overall risk to benefit profile of the product and
to provide an adequate basis for product labeling. |
Phase
1, Phase 2 and Phase 3 clinical trials may not be completed successfully within any specified period, if at all.
Post-approval
clinical trials, sometimes referred to as Phase 4 clinical trials, may be conducted after initial marketing approval. These clinical
trials are used to gain additional experience from the treatment of patients in the intended therapeutic indication, particularly for
long-term safety follow-up.
During
all phases of clinical development, regulatory agencies require extensive monitoring and auditing of all clinical activities, clinical
data, and clinical trial investigators. Annual progress reports detailing the results of the clinical trials must be submitted to the
FDA. Written IND safety reports must be promptly submitted to the FDA, and the investigators for serious and unexpected adverse events,
any findings from other studies, tests in laboratory animals or in vitro testing that suggest a significant risk for human patients,
or any clinically important increase in the rate of a serious suspected adverse reaction over that listed in the protocol or investigator
brochure. The sponsor must submit an IND safety report within 15 calendar days after the sponsor determines that the information qualifies
for reporting. The sponsor also must notify the FDA of any unexpected fatal or life-threatening suspected adverse reaction within seven
calendar days after the sponsor’s initial receipt of the information. The FDA or the sponsor or its data safety monitoring board,
an independent group of experts that evaluates study data for safety and makes recommendations concerning continuation, modification,
or termination of clinical trials, may suspend or terminate a clinical trial at any time on various grounds, including a finding that
the research patients are being exposed to an unacceptable health risk, including risks inferred from other unrelated immunotherapy trials.
Similarly, an IRB can suspend or terminate approval of a clinical trial at its institution if the clinical trial is not being conducted
in accordance with the IRB’s requirements or if the biological product has been associated with unexpected serious harm to patients.
Because
this is a relatively new and expanding area of novel therapeutic interventions, there can be no assurance as to the length of the trial
period, the number of patients the FDA will require to be enrolled in the trials in order to establish the safety, efficacy, purity and
potency of immunotherapy products, or that the data generated in these trials will be acceptable to the FDA to support marketing approval.
Concurrently
with clinical trials, companies usually complete additional nonclinical studies and must also develop additional information about the
physical characteristics of the biological product as well as finalize a process for manufacturing the product in commercial quantities
in accordance with cGMP requirements. To help reduce the risk of the introduction of adventitious agents with use of biological products,
the PHSA emphasizes the importance of manufacturing control for products whose attributes cannot be precisely defined. The manufacturing
process must be capable of consistently producing quality batches of the product candidate and, among other things, the sponsor must
develop methods for testing the identity, strength, quality, potency and purity of the final biological product. Additionally, appropriate
packaging must be selected and tested and stability studies must be conducted to demonstrate that the biological product candidate does
not undergo unacceptable deterioration over its shelf life.
United
States Review and Approval Processes
After
the completion of clinical trials of a biological product, FDA approval of a BLA must be obtained before commercial marketing of the
biological product. The BLA must include results of product development, laboratory and animal studies, human trials, information on
the manufacture and composition of the product, proposed labeling and other relevant information. The testing and approval processes
require substantial time and effort and there can be no assurance that the FDA will accept the BLA for filing and, even if filed, that
any approval will be granted on a timely basis, if at all as the FDA has significant discretion to approve or reject the BLA and to require
additional preclinical or clinical studies.
Under
the Prescription Drug User Fee Act, or PDUFA, as amended, each BLA must be accompanied by a significant user fee. The FDA adjusts the
PDUFA user fees on an annual basis. PDUFA also imposes an annual program fee for approved biological products. Fee waivers or reductions
are available in certain circumstances, including a waiver of the application fee for the first application filed by a small business.
Additionally, no user fees are assessed on BLAs for products designated as orphan drugs, unless the product also includes a non-orphan
indication.
Within
60 days following submission of the application, the FDA reviews a BLA submitted to determine if it is substantially complete before
the agency accepts it for filing. The FDA may refuse to file any BLA that it deems incomplete or not properly reviewable at the time
of submission and may request additional information. In this event, the BLA must be resubmitted with the additional information. The
resubmitted application also is subject to review before the FDA accepts it for filing. Once the submission is accepted for filing, the
FDA begins an in-depth substantive review of the BLA. The FDA reviews the BLA to determine, among other things, whether the proposed
product is safe, potent, and/or effective for its intended use, and has an acceptable purity profile, and whether the product is being
manufactured in accordance with cGMP to assure and preserve the product’s identity, safety, strength, quality, potency and purity.
The FDA may refer applications for novel biological products or biological products that present difficult questions of safety or efficacy
to an advisory committee, typically a panel that includes clinicians and other experts, for review, evaluation and a recommendation as
to whether the application should be approved and under what conditions. The FDA is not bound by the recommendations of an advisory committee,
but it considers such recommendations carefully when making decisions. During the biological product approval process, the FDA also will
determine whether a Risk Evaluation and Mitigation Strategy, or REMS, is necessary to ensure that the benefits of the product outweigh
its risks and to assure the safe use of the biological product, which could include medication guides, physician communication plans,
or elements to assure safe use, such as restricted distribution methods, patient registries and other risk minimization tools. FDA determines
the requirement for a REMS, as well as the specific REMS provisions, on a case-by-case basis. If the FDA concludes a REMS is needed,
the sponsor of the BLA must submit a proposed REMS. The FDA will not approve a BLA without a REMS, if required.
Before
approving a BLA, the FDA will inspect the facilities at which the product is manufactured. The FDA will not approve the product unless
it determines that the manufacturing processes and facilities are in compliance with cGMP requirements and adequate to assure consistent
production of the product within required specifications. For immunotherapy products, the FDA also will not approve the product if the
manufacturer is not in compliance with the GTPs, to the extent applicable. These are FDA regulations and guidance documents that govern
the methods used in, and the facilities and controls used for, the manufacture of human cells, tissues, and cellular and tissue based
products, or HCT/Ps, which are human cells or tissue intended for implantation, transplant, infusion, or transfer into a human recipient.
The primary intent of the GTP requirements is to ensure that cell and tissue based products are manufactured in a manner designed to
prevent the introduction, transmission and spread of communicable disease. FDA GTP regulations also require tissue establishments to
register and list their HCT/Ps with the FDA and, when applicable, to evaluate donors through screening and testing. Additionally, before
approving a BLA, the FDA will typically inspect one or more clinical sites to assure that the clinical trials were conducted in compliance
with IND trial requirements and GCP requirements.
To
assure cGMP, GTP and GCP compliance, an applicant must incur significant expenditure of time, money and effort in the areas of training,
recordkeeping, production, and quality control.
Notwithstanding
the submission of relevant data and information, the FDA may ultimately decide that the BLA does not satisfy its regulatory criteria
for approval and deny approval. If the agency decides not to approve the BLA in its then current form, the FDA will issue a Complete
Response Letter, which generally outlines the specific deficiencies in the BLA identified by the FDA and may require additional clinical
or other data or impose other conditions that must be met in order to secure final approval of the application. The deficiencies identified
may be minor, for example, requiring labeling changes, or major, for example, requiring additional clinical trials. Even with the submission
of additional information, the FDA may ultimately decide that the application does not satisfy the regulatory criteria for approval.
If a Complete Response Letter is issued, the applicant may either resubmit the BLA, addressing all of the deficiencies identified in
the letter, or withdraw the application.
If
a product receives regulatory approval, the approval is limited to the conditions of use (e.g., patient population, indication)
described in the application.
Further,
the FDA may require that certain contraindications, warnings or precautions be included in the product labeling, or otherwise limit the
scope of any approval. In addition, the FDA may require post marketing clinical trials, sometimes referred to as Phase 4 clinical trials,
designed to further assess a biological product’s safety and effectiveness, and testing and surveillance programs to monitor the
safety of approved products that have been commercialized. After approval, many types of changes to the approved product, such as adding
new indications, manufacturing changes and additional labeling claims, are subject to further testing requirements and FDA review and
approval.
In
addition, under the Pediatric Research Equity Act, or PREA, a BLA or supplement to a BLA must contain data to assess the safety and effectiveness
of the product for the claimed indications in all relevant pediatric subpopulations and to support dosing and administration for each
pediatric subpopulation for which the product is safe and effective. The FDA may grant deferrals for submission of data or full or partial
waivers.
United
States Post-Approval Requirements
Any
products for which we receive FDA approvals are subject to continuing regulation by the FDA, including, among other things, record-keeping
requirements, reporting of adverse experiences with the product, providing the FDA with updated safety and efficacy information, product
sampling and distribution requirements, and complying with FDA promotion and advertising requirements, which include, among others, standards
for direct-to-consumer advertising, restrictions on promoting products for uses or in patient populations that are not described in the
product’s approved uses (known as “off-label use”), limitations on industry-sponsored scientific and educational activities,
and requirements that important safety information and material facts related to the product be disclosed. Although physicians may prescribe
legally available products for off-label uses, if the physicians deem to be appropriate in their professional medical judgment, manufacturers
may not market or promote such off-label uses. The FDA and other agencies actively enforce the laws and regulations prohibiting the promotion
of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant civil, criminal
and administrative liability.
In
addition, quality control and manufacturing procedures must continue to conform to applicable manufacturing requirements after approval
to ensure the long-term stability of the product. We currently are making clinical trial product in our own facilities in Scotland, United
Kingdom. In the future, however, we expect to rely, on third parties for the production of commercial quantities of our products in accordance
with cGMP regulations. cGMP regulations require among other things, quality control and quality assurance as well as the corresponding
maintenance of records and documentation and the obligation to investigate and correct any deviations from cGMP. Manufacturers and other
entities involved in the manufacture and distribution of approved products are required to register their establishments with the FDA
and certain state agencies, and are subject to periodic unannounced inspections by the FDA and certain state agencies for compliance
with cGMP and other laws. Accordingly, manufacturers must continue to expend time, money, and effort in the area of production and quality
control to maintain cGMP compliance. Discovery of problems with a product after approval may result in restrictions on a product, manufacturer,
or holder of an approved BLA, including, among other things, recall or withdrawal of the product from the market.
The
FDA also may require post-marketing testing, known as Phase 4 testing, and surveillance to monitor the effects of an approved product.
Discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, with manufacturing
processes, or the failure to comply with applicable FDA requirements can have negative consequences, including adverse publicity, judicial
or administrative enforcement, complete withdrawal from the market, product recalls, warning letters from the FDA, mandated corrective
advertising or communications with doctors, product seizure or detention, injunctions, and civil or criminal penalties, among others.
Newly discovered or developed safety or effectiveness data may require changes to a product’s approved labeling, including the
addition of new warnings and contraindications, and also may require the implementation of other risk management measures. Also, new
government requirements, including those resulting from new legislation, may be established, or the FDA’s policies may change,
which could delay or prevent regulatory approval of our products under development.
United
States Marketing Exclusivity
The
Biologics Price Competition and Innovation Act amended the PHSA to authorize the FDA to approve similar versions of innovative biologics,
commonly known as biosimilars. Biosimilars are approved pursuant to an abbreviated pathway whereby applicants need not submit the full
slate of preclinical and clinical data, and approval is based in part on the FDA’s findings of safety, purity, and potency for
the original biologic (i.e., the reference product). Original BLAs are eligible to receive 12 years of exclusivity from the time of first
licensure of the product, which prevents the FDA from approving any biosimilars to the reference product through the abbreviated pathway,
but does not prevent approval of BLAs that are accompanied by a full data package and that do not rely on the reference product. A biosimilar
may be approved if the product is highly similar to the reference product notwithstanding minor differences in clinically inactive components
and there are no clinically meaningful differences with the reference product in terms of the safety, purity, and potency.
Pediatric
exclusivity is another type of regulatory market exclusivity in the United States. Pediatric exclusivity, if granted, adds six months
to existing exclusivity periods and patent terms. This six-month exclusivity, which runs from the end of other exclusivity protection
or patent term, may be granted based on the voluntary completion of a pediatric trial in accordance with an FDA-issued “Written
Request” for such a trial.
United
States Coverage, Pricing and Reimbursement
Significant
uncertainty exists as to the coverage and reimbursement status of any product candidates for which we obtain regulatory approval.
In the United States and markets in other countries, sales of any products for which we receive regulatory approval for commercial sale
will depend, in significant part, on the extent to which third-party payors provide coverage, and establish adequate reimbursement levels
for such products. In the United States, third-party payers include federal and state healthcare programs, private managed care organizations,
health insurers and other organizations. The process for determining whether a third-party payer will provide coverage for a product
may be separate from the process of establishing the reimbursement rate that such a payor will pay for the product. Third-party payors
may limit coverage to specific products on an approved list, also known as a formulary, which might not include all of the FDA-approved
products for a particular indication. Third-party payors are increasingly challenging the price, examining the medical necessity of and
reviewing the cost-effectiveness of medical products, therapies and services, in addition to questioning their safety and efficacy.
Reimbursement
may impact the demand for, and/or the price of, any product candidate which obtains marketing approval. Even if coverage and reimbursement
is obtained for a given product candidate by a third-party payor, the resulting reimbursement payment rates may not be adequate or may
require co-payments that patients find unacceptably high. Patients who are prescribed medications for the treatment of their conditions,
and their prescribing physicians, generally rely on third-party payors to reimburse all or part of the costs associated with those medications.
Patients are unlikely to use a product, and physicians may be less likely to prescribe a product, unless coverage is provided and reimbursement
is adequate to cover all or a significant portion of the cost of the product. Therefore, coverage and adequate reimbursement is critical
to new drug product acceptance.
Different
pricing and reimbursement schemes exist in other countries. In the EU, governments influence the price of pharmaceutical products through
their pricing and reimbursement rules and control of national healthcare systems that fund a large part of the cost of those products
to consumers. Some jurisdictions operate positive and negative list systems under which products may only be marketed once a reimbursement
price has been agreed. To obtain reimbursement or pricing approval, some of these countries may require the completion of additional
clinical trials that compare the cost-effectiveness of a particular product candidate to currently available therapies. Other member
states allow companies to fix their own prices for medicines, but monitor and control company profits. In addition, in some countries,
cross-border imports from low-priced markets exert a commercial pressure on pricing within a country.
The
downward pressure on healthcare costs in general, particularly prescription drugs and biologics, has become very intense. Governments
have shown significant interest in implementing cost-containment programs, including price controls, restrictions on reimbursement and
requirements for substitution of generic products. As a result, increasingly high barriers are being erected to the entry of new products.
The marketability of any product candidates for which we receive regulatory approval for commercial sale may suffer if the government
and third-party payors fail to provide coverage and adequate reimbursement. In addition, emphasis on managed care in the United States
has increased and we expect will continue to increase the pressure on healthcare pricing. Coverage policies and third-party reimbursement
rates may change at any time. Even if favorable coverage and reimbursement status is attained for one or more products for which we receive
regulatory approval, less favorable coverage policies and reimbursement rates may be implemented in the future.
United
States Healthcare Laws Governing Interactions with Healthcare Providers
In
addition to FDA restrictions on marketing of pharmaceutical products, several other types of state and federal laws restrict our business
activities, including certain marketing practices. These laws include, without limitation, anti-kickback laws, false claims laws, data
privacy and security laws, as well as transparency laws regarding payments or other items of value provided to healthcare providers.
The
U.S. federal Anti-Kickback Statute prohibits any person or entity from, among other things, knowingly and willfully offering, paying,
soliciting or receiving remuneration, directly or indirectly, in cash or in kind, to induce or reward, or in return for, purchasing,
leasing, ordering or arranging for the purchase, lease or order of any healthcare item, good, facility or service reimbursable, in whole
or in part, under Medicare, Medicaid or other federal healthcare programs. The term “remuneration” has been broadly interpreted
to include anything of value. This statute has been interpreted to apply to arrangements between pharmaceutical manufacturers on the
one hand and prescribers, purchasers and formulary managers on the other hand. Although there are a number of statutory exceptions and
regulatory safe harbors protecting certain common activities from prosecution or other regulatory sanctions, the exceptions and safe
harbors are drawn narrowly, and practices that involve remuneration that are alleged to be intended to induce prescribing, purchases
or recommendations may be subject to scrutiny if they do not qualify for an exception or safe harbor. Failure to meet all of the requirements
of a particular applicable statutory exception or regulatory safe harbor does not make the conduct per se illegal under the U.S. federal
Anti-Kickback Statute. Instead, the legality of the arrangement will be evaluated on a case-by-case basis based on a cumulative review
of all its facts and circumstances. Several courts have interpreted the statute’s intent requirement to mean that if any one purpose
of an arrangement involving remuneration is to induce referrals of federal healthcare covered business, the U.S. federal Anti-Kickback
Statute has been violated. Additionally, the intent standard under the U.S. federal Anti-Kickback Statute was amended by the Patient
Protection and Affordable Care Act of 2010, as amended by the Health Care and Education Reconciliation Act of 2010, collectively the
Affordable Care Act, or ACA, to a stricter standard such that a person or entity no longer needs to have actual knowledge of the statute
or specific intent to violate it in order to have committed a violation. In addition, the ACA codified case law that a claim including
items or services resulting from a violation of the U.S. federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes
of the U.S. federal False Claims Act.
Federal
civil and criminal false claims laws and civil monetary penalties laws, including the U.S. federal False Claims Act, which can be enforced
through civil whistleblower or qui tam actions, prohibit any person or entity from, among other things, knowingly presenting, or causing
to be presented, a false claim for payment to the federal government, or knowingly making, or causing to be made, a false statement to
have a false claim paid. Pharmaceutical and other healthcare companies have been prosecuted under these laws for, among other things,
allegedly inflating drug prices they report to pricing services, which in turn were used by the government to set Medicare and Medicaid
reimbursement rates, and for allegedly providing free product to customers with the expectation that the customers would bill federal
programs for the product. In addition, certain marketing practices, including off-label promotion, may also violate false claims laws.
Further, pharmaceutical manufacturers can be held liable under the U.S. federal False Claims Act even when they do not submit claims
directly to government payors if they are deemed to “cause” the submission of false or fraudulent claims.
The
U.S. federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, created new federal criminal statutes that prohibit
among other actions, knowingly and willfully executing, or attempting to execute, a scheme to defraud any healthcare benefit program,
including private third- party payors, knowingly and willfully embezzling or stealing from a healthcare benefit program, willfully obstructing
a criminal investigation of a healthcare offense, and knowingly and willfully falsifying, concealing or covering up a material fact or
making any materially false, fictitious or fraudulent statement in connection with the delivery of or payment for healthcare benefits,
items or services. Like the U.S. federal Anti-Kickback Statute, the ACA amended the intent standard for certain healthcare fraud under
HIPAA such that a person or entity no longer needs to have actual knowledge of the statute or specific intent to violate it in order
to have committed a violation.
HIPAA,
as amended by the Health Information Technology for Economic and Clinical Health Act, or HITECH, and their implementing regulations,
impose certain requirements on “covered entities,” including certain healthcare providers, health plans and healthcare clearinghouses,
as well as their respective “business associates” that create, receive, maintain or transmit individually identifiable health
information for or on behalf of a covered entity, and their covered subcontractors, relating to the privacy, security, transmission and
breach of individually identifiable health information. Further, HITECH also created four new tiers of civil monetary penalties, amended
HIPAA to make civil and criminal penalties directly applicable to business associates, and gave state attorneys general new authority
to file civil actions for damages or injunctions in U.S. federal courts to enforce HIPAA and seek attorneys’ fees and costs associated
with pursuing federal civil actions.
Additionally,
the federal Physician Payments Sunshine Act, created under the ACA, and its implementing regulations, require certain manufacturers of
drugs, devices, biologics and medical supplies for which payment is available under Medicare, Medicaid or the Children’s Health
Insurance Program (with certain exceptions) to annually report to the Centers for Medicare and Medicaid Services, or CMS, information
related to certain payments or other transfers of value provided to physicians (defined to include doctors, dentists, optometrists, podiatrists
and chiropractors) and teaching hospitals, or to entities or individuals at the request of, or designated on behalf of, the physicians
and teaching hospitals as well as certain ownership and investment interests held by physicians and their immediate family members. Beginning
in 2022, applicable manufacturers also will be required to report information regarding payments and transfers of value provided to physician
assistants, nurse practitioners, clinical nurse specialists, certified nurse anesthetists, anesthesiologist assistants, and certified
nurse-midwives.
Additionally,
similar healthcare laws and regulations in the European Union and other jurisdictions, including reporting requirements detailing interactions
with and payments to healthcare providers and laws governing the privacy and security of certain protected information, such as GDPR,
which imposes obligations and restrictions on the collection and use of personal data relating to individuals located in the European
Union (including health data).
Finally,
the majority of states also have statutes or regulations similar to the aforementioned federal laws, some of which are broader in scope
and apply to items and services reimbursed under Medicaid and other state programs, or, in several states, apply regardless of the payor.
Some state laws require pharmaceutical companies to comply with the pharmaceutical industry’s voluntary compliance guidelines and
the relevant compliance guidance promulgated by the federal government in addition to requiring drug manufacturers to report information
related to payments to clinicians and other healthcare providers or marketing expenditures. Some states and local jurisdictions require
the registration of pharmaceutical sales representatives. State and foreign laws also govern the privacy and security of health information
in some circumstances, many of which differ from each other in significant ways and often are not preempted by HIPAA, thus complicating
compliance efforts.
Because
of the breadth of these laws and the narrowness of their exceptions and safe harbors, it is possible that business activities can be
subject to challenge under one or more of such laws. The scope and enforcement of each of these laws is uncertain and subject to rapid
change in the current environment of healthcare reform, especially in light of the lack of applicable precedent and regulations. Federal
and state enforcement bodies have recently increased their scrutiny of interactions between healthcare companies and healthcare providers,
which has led to a number of investigations, prosecutions, convictions and settlements in the healthcare industry.
Ensuring
that business arrangements with third parties comply with applicable healthcare laws and regulations is costly and time consuming. If
business operations are found to be in violation of any of the laws described above or any other applicable governmental regulations
a pharmaceutical manufacturer may be subject to penalties, including civil, criminal and administrative penalties, damages, fines, disgorgement,
individual imprisonment, exclusion from governmental funded healthcare programs, such as Medicare and Medicaid, contractual damages,
reputational harm, diminished profits and future earnings, additional reporting obligations and oversight if subject to a corporate integrity
agreement or other agreement to resolve allegations of non-compliance with these laws, and curtailment or restructuring of operations,
any of which could adversely affect a pharmaceutical manufacturer’s ability to operate its business and the results of its operations.
United
States Healthcare Reform Efforts
A
primary trend in the United States healthcare industry and elsewhere is cost containment. Over the last several years, there have been
federal and state proposals and legislation enacted regarding the pricing of pharmaceutical and biopharmaceutical products, limiting
coverage and reimbursement for drugs and other medical products, and making changes to healthcare financing and the delivery of care
in the United States.
There
continues to be heightened Congressional scrutiny in the United States of pharmaceutical pricing practices designed to, among other things,
bring more transparency in product pricing, review the relationship between pricing and manufacturer patient programs, and reform government
program reimbursement methodologies for products. At the state level, legislatures have increasingly enacted legislation and implemented
regulations designed to control pharmaceutical and biological product pricing, including price or patient reimbursement constraints,
discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed
to encourage importation from other countries and bulk purchasing. In addition, regional healthcare authorities and individual hospitals
are increasingly using bidding procedures to determine what pharmaceutical products and which suppliers will be included in their prescription
drug and other healthcare programs.
In
addition, other federal health reform measures have been proposed and adopted in the United States that could impact cell therapy. Most
notably, there has been political support for rules related to value-based payment alternatives in the Medicaid program. Medicaid is
a jointly run federal and state program that provides health benefits coverage for low-income residents and children. In exchange for
broad coverage in Medicaid, drug manufacturers are required to sign a Medicare Drug Rebate agreement which requires them to offer Medicaid
programs the “best price” available for a particular product. This “best price” takes into consideration any
rebates or concessions manufacturers offer, with some exceptions. The final rule would exempt value-based or outcomes-based payment arrangements
from the definition of “best price” which provides manufacturers more flexibility to work with commercial payers and states
on innovate payment mechanisms for high-cost cell and gene therapies. While Medicaid is not a significant driver of cell therapy sales
it is a bellwether program and one we watch closely.
United
States FCPA, the Bribery Act and Other Laws
The
FCPA, prohibits any U.S. individual or business from paying, offering, or authorizing payment or offering of anything of value, directly
or indirectly, to any foreign official, political party or candidate for the purpose of influencing any act or decision of the foreign
entity in order to assist the individual or business in obtaining or retaining business. The FCPA also obligates companies whose securities
are listed in the United States to comply with accounting provisions requiring the company to maintain books and records that accurately
and fairly reflect all transactions of the corporation, including international subsidiaries, and to devise and maintain an adequate
system of internal accounting controls for international operations. Activities that violate the FCPA, even if they occur wholly outside
the United States, can result in criminal and civil fines, imprisonment, disgorgement, oversight, and debarment from government contracts.
Our
operations are also subject to non-U.S. anti-corruption laws such as the Bribery Act. As with the FCPA, these laws generally prohibit
us and our employees and intermediaries from authorizing, promising, offering, or providing, directly or indirectly, improper or prohibited
payments, or anything else of value, to government officials or other persons to obtain or retain business or gain some other business
advantage. Under the Bribery Act, we may also be liable for failing to prevent a person associated with us from committing a bribery
offense.
We
are also subject to other laws and regulations governing our international operations, including regulations administered by the governments
of the United Kingdom and the United States and authorities in the European Union, including applicable export control regulations, economic
sanctions and embargoes on certain countries and persons, anti-money laundering laws, import and customs requirements and currency exchange
regulations, collectively referred to as trade control laws.
Failure
to comply with the Bribery Act, the FCPA and other anti-corruption laws and trade control laws could subject us to criminal and civil
penalties, disgorgement and other sanctions and remedial measures, and legal expenses.
Review
and Approval of New Drug Products in the European Union
In
the European Union, medicinal products, including advanced therapy medicinal products, or ATMPs, are subject to extensive pre- and post-market
regulation by regulatory authorities at both the European Union and national levels. ATMPs comprise gene therapy products, somatic-cell
therapy products and tissue engineered products, which are cells or tissues that have undergone substantial manipulation and that are
administered to human beings in order to regenerate, repair or replace a human tissue. We anticipate that our therapy products will be
regulated as ATMPs in the European Union. There is legislation at a European Union level relating to the standards of quality and safety
for the collection and testing of human blood and blood components for use in cell-based therapies, which could apply to our products.
Additionally, there may be local legislation in various European Union Member States, which may be more restrictive than the European
Union legislation, and we would need to comply with such legislation to the extent it applies.
EU
Clinical Trials
Clinical
trials of medicinal products in the European Union must be conducted in accordance with European Union and national regulations and the
International Conference on Harmonization, or ICH, guidelines on Good Clinical Practices, or GCP. Additional GCP guidelines from the
European Commission, focusing in particular on traceability, apply to clinical trials of ATMPs. The sponsor must take out a clinical
trial insurance policy, and in most European Union countries, the sponsor is liable to provide “no fault” compensation to
any study subject injured in the clinical trial.
Prior
to commencing a clinical trial, the sponsor must obtain a clinical trial authorization from the competent authority, and a positive opinion
from an independent ethics committee. The application for a clinical trial authorization must include, among other things, a copy of
the trial protocol and an investigational medicinal product dossier containing information about the manufacture and quality of the medicinal
product under investigation. Currently, clinical trial authorization applications must be submitted to the competent authority in each
EU Member State in which the trial will be conducted. Under the new Regulation on Clinical Trials, which is to take effect in December
2021, there will be a centralized application procedure where one national authority takes the lead in reviewing the application and
the other national authorities have only a limited involvement. Any substantial changes to the trial protocol or other information submitted
with the clinical trial applications must be notified to or approved by the relevant competent authorities and ethics committees. Medicines
used in clinical trials must be manufactured in accordance with cGMP. Other national and European Union-wide regulatory requirements
also apply.
During
the development of a medicinal product, the EMA and national medicines regulators within the European Union provide the opportunity for
dialogue and guidance on the development program. At the EMA level, this is usually done in the form of scientific advice, which is given
by the Scientific Advice Working Party of the Committee for Medicinal Products for Human Use, or CHMP. A fee is incurred with each scientific
advice procedure. Advice from the EMA is typically provided based on questions concerning, for example, quality (chemistry, manufacturing
and controls testing), nonclinical testing and clinical studies, and pharmacovigilance plans and risk-management programs. In accordance
with the EMA’s policy, scientific advice will not be legally binding with regard to any future marketing authorization application
of the product concerned.
EU
Marketing Authorizations
In
order to market a new medicinal product in the European Union, a company must submit and obtain approval from regulators of a marketing
authorization application, or MAA. The process for doing this depends, among other things, on the nature of the medicinal product.
The
centralized procedure results in a single marketing authorization, or MA, granted by the European Commission that is valid across the
EEA (i.e., the European Union as well as Iceland, Liechtenstein and Norway). The centralized procedure is compulsory for human drugs
that are: (i) derived from biotechnology processes, such as genetic engineering, (ii) contain a new active substance indicated for the
treatment of certain diseases, such as HIV/AIDS, cancer, diabetes, neurodegenerative diseases, autoimmune and other immune dysfunctions
and viral diseases, (iii) officially designated orphan medicines and (iv) advanced-therapy medicines, such as gene therapy, somatic cell
therapy or tissue-engineered medicines. The centralized procedure may at the request of the applicant also be used in certain other cases.
Therefore, the centralized procedure would be mandatory for the products we are developing.
The
Committee for Advanced Therapies, or CAT, is responsible in conjunction with the CHMP for the evaluation of ATMPs. The CAT is primarily
responsible for the scientific evaluation of ATMPs and prepares a draft opinion on the quality, safety and efficacy of each ATMP for
which a marketing authorization application is submitted. The CAT’s opinion is then taken into account by the CHMP when giving
its final recommendation regarding the authorization of a product in view of the balance of benefits and risks identified. Although the
CAT’s draft opinion is submitted to the CHMP for final approval, the CHMP may depart from the draft opinion, if it provides detailed
scientific justification. The CHMP and CAT are also responsible for providing guidelines on ATMPs and have published numerous guidelines,
including specific guidelines on gene therapies and cell therapies. These guidelines provide additional guidance on the factors that
the EMA will consider in relation to the development and evaluation of ATMPs and include, among other things, the preclinical studies
required to characterize ATMPs; the manufacturing and control information that should be submitted in a marketing authorization application;
and post-approval measures required to monitor patients and evaluate the long-term efficacy and potential adverse reactions of ATMPs.
Although these guidelines are not legally binding, we believe that our compliance with them is likely necessary to gain and maintain
approval for any of our product candidates.
Under
the centralized procedure in the European Union, the maximum timeframe for the evaluation of an MAA by the EMA is 210 days. This excludes
so-called clock stops, during which additional written or oral information is to be provided by the applicant in response to questions
asked by the CHMP. At the end of the review period, the CHMP provides an opinion to the European Commission. If this is opinion favorable,
the Commission may then adopt a decision to grant an MA. In exceptional cases, the CHMP might perform an accelerated review of an MAA
in no more than 150 days. This is usually when the product is of major interest from the point of view of public health and, in particular,
from the viewpoint of therapeutic innovation.
The
European Commission may grant a so-called “marketing authorization under exceptional circumstances”. Such authorization is
intended for products for which the applicant can demonstrate that it is unable to provide comprehensive data on the efficacy and safety
under normal conditions of use, because the indications for which the product in question is intended are encountered so rarely that
the applicant cannot reasonably be expected to provide comprehensive evidence, or in the present state of scientific knowledge, comprehensive
information cannot be provided, or it would be contrary to generally accepted principles of medical ethics to collect such information.
Consequently, marketing authorization under exceptional circumstances may be granted subject to certain specific obligations, which may
include the following:
| |
● |
the
applicant must complete an identified program of studies within a time period specified by the competent authority, the results of
which form the basis of a reassessment of the benefit/risk profile; |
| |
|
|
| |
● |
the
medicinal product in question may be supplied on medical prescription only and may in certain cases be administered only under strict
medical supervision, possibly in a hospital and in the case of a radiopharmaceutical, by an authorized person; and |
| |
|
|
| |
● |
the
package leaflet and any medical information must draw the attention of the medical practitioner to the fact that the particulars
available concerning the medicinal product in question are as yet inadequate in certain specified respects. |
A
marketing authorization under exceptional circumstances is subject to annual review to reassess the risk-benefit balance in an annual
reassessment procedure. Continuation of the authorization is linked to the annual reassessment and a negative assessment could potentially
result in the marketing authorization being suspended or revoked. The renewal of a marketing authorization of a medicinal product under
exceptional circumstances, however, follows the same rules as a “normal” marketing authorization. Thus, a marketing authorization
under exceptional circumstances is granted for an initial five years, after which the authorization will become valid indefinitely, unless
the EMA decides that safety grounds merit one additional five-year renewal.
The
European Commission may also grant a so-called “conditional marketing authorization” prior to obtaining the comprehensive
clinical data required for an application for a full marketing authorization. Such conditional marketing authorizations may be granted
for product candidates (including medicines designated as orphan medicinal products), if (i) the risk-benefit balance of the product
candidate is positive, (ii) it is likely that the applicant will be in a position to provide the required comprehensive clinical trial
data, (iii) the product fulfills an unmet medical need and (iv) the benefit to public health of the immediate availability on the market
of the medicinal product concerned outweighs the risk inherent in the fact that additional data are still required. A conditional marketing
authorization may contain specific obligations to be fulfilled by the marketing authorization holder, including obligations with respect
to the completion of ongoing or new studies, and with respect to the collection of pharmacovigilance data. Conditional marketing authorizations
are valid for one year, and may be renewed annually, if the risk-benefit balance remains positive, and after an assessment of the need
for additional or modified conditions and/or specific obligations. The timelines for the centralized procedure described above also apply
with respect to the review by the CHMP of applications for a conditional marketing authorization.
EU
Data Exclusivity
Marketing
authorization applications for generic medicinal products do not need to include the results of preclinical and clinical trials, but
instead can refer to the data included in the marketing authorization of a reference product for which regulatory data exclusivity has
expired. If a marketing authorization is granted for a medicinal product containing a new active substance, that product benefits from
eight years of data exclusivity, during which generic marketing authorization applications referring to the data of that product may
not be accepted by the regulatory authorities, and a further two years of market exclusivity, during which such generic products may
not be placed on the market. The two-year period may be extended to three years if during the first eight years a new therapeutic indication
with significant clinical benefit over existing therapies is approved.
There
is a special regime for biosimilars, or biological medicinal products that are similar to a reference medicinal product but that do not
meet the definition of a generic medicinal product, for example, because of differences in raw materials or manufacturing processes.
For such products, the results of appropriate preclinical or clinical trials must be provided, and guidelines from the EMA detail the
type of quantity of supplementary data to be provided for different types of biological product. There are no such guidelines for complex
biological products, such as gene or cell therapy medicinal products, and so it is unlikely that biosimilars of those products will currently
be approved in the European Union. However, guidance from the EMA states that they will be considered in the future in light of the scientific
knowledge and regulatory experience gained at the time.
EU
Pediatric Development
In
the European Union, companies developing a new medicinal product must agree to a Pediatric Investigation Plan, or PIP, with the EMA and
must conduct pediatric clinical trials in accordance with that PIP, unless a deferral or waiver applies, (e.g., because the relevant
disease or condition occurs only in adults). The marketing authorization application for the product must include the results of pediatric
clinical trials conducted in accordance with the PIP, unless a waiver applies, or a deferral has been granted, in which case the pediatric
clinical trials must be completed at a later date. Products that are granted a marketing authorization on the basis of the pediatric
clinical trials conducted in accordance with the PIP are eligible for a six-month extension of the protection under a supplementary protection
certificate (if any is in effect at the time of approval) or, in the case of orphan medicinal products, a two-year extension of the orphan
market exclusivity. This pediatric reward is subject to specific conditions and is not automatically available when data in compliance
with the PIP are developed and submitted.
EU
Post-Approval Controls
The
holder of a marketing authorization must establish and maintain a pharmacovigilance system and appoint an individual qualified person
for pharmacovigilance, or QPPV, who is responsible for oversight of that system. Key obligations include expedited reporting of suspected
serious adverse reactions and submission of periodic safety update reports, or PSURs.
All
new marketing authorization applications must include a risk management plan, or RMP, describing the risk management system that the
company will put in place and documenting measures to prevent or minimize the risks associated with the product. The regulatory authorities
may also impose specific obligations as a condition of the marketing authorization. Such risk-minimization measures or post-authorization
obligations may include additional safety monitoring, more frequent submission of PSURs, or the conduct of additional clinical trials
or post-authorization safety studies. RMPs and PSURs are routinely available to third parties requesting access, subject to limited redactions.
All advertising and promotional activities for the product must be consistent with the approved summary of product characteristics, and
therefore all off-label promotion is prohibited. Direct-to-consumer advertising of prescription medicines is also prohibited in the European
Union. Although general requirements for advertising and promotion of medicinal products are established under EU directives, the details
are governed by regulations in each EU Member State and can differ from one country to another.
EU
Pricing and Reimbursement in the European Union
Governments
influence the price of medicinal products in the European Union through their pricing and reimbursement rules and control of national
healthcare systems that fund a large part of the cost of those products to consumers. Some jurisdictions operate positive and negative
list systems under which products may only be marketed once a reimbursement price has been agreed. To obtain reimbursement or pricing
approval, some of these countries may require the completion of clinical trials that compare the cost-effectiveness of a particular product
candidate to currently available therapies. Other EU Member States allow companies to fix their own prices for medicines, but monitor
and control company profits. The downward pressure on healthcare costs in general, particularly prescription medicines, has become very
intense. As a result, increasingly high barriers are being erected to the entry of new products.
Brexit
and the Regulatory Framework in the United Kingdom
The
United Kingdom officially withdrew from the European Union on January 31, 2020 (“Brexit”). Pursuant to the formal withdrawal
arrangements agreed between the United Kingdom and the European Union, the United Kingdom was subject to a transition period until December
31, 2020, or the Transition Period, during which European Union rules continued to apply. A trade and cooperation agreement, or the Trade
and Cooperation Agreement, that outlines the future trading relationship between the United Kingdom and the European Union was agreed
in December 2020.
Brexit
may influence the attractiveness of the United Kingdom as a place to conduct clinical trials. The European Union’s regulatory environment
for clinical trials is being harmonized as part of the Clinical Trial Regulations, which come into full effect at the end of 2021, but
it is currently unclear as to what extent the United Kingdom will seek to align its regulations with the European Union. Failure of the
United Kingdom to closely align its regulations with the EU may have an effect on the cost of conducting clinical trials in the United
Kingdom as opposed to other countries and/or make it harder to seek a marketing authorization for our product candidates on the basis
of clinical trials conducted in the United Kingdom.
In
the short term there will be few changes to clinical trials that only have sites in the United Kingdom. The MHRA have confirmed that
the sponsor of a clinical trial can be based in the EEA for an initial period following Brexit. Further investigational medicinal products
can be supplied directly from the EU/EEA to a trial site in the United Kingdom without further oversight until January 1, 2022, and to
Northern Ireland beyond such date. The United Kingdom is now a “third country” for the purpose of clinical trials that have
sites in the EEA. For such trials the sponsor/legal representative must be based in the EEA, and the trial must be registered on the
EU Clinical Trials Register (including data on sites outside of the EEA).
The
data exclusivity periods in the United Kingdom are currently in line with those in the European Union, but the Trade and Cooperation
Agreement provides that the periods for both data and market exclusivity are to be determined by domestic law, and so there could be
divergence in the future. It is currently unclear whether the MHRA in the United Kingdom is sufficiently prepared to handle the increased
volume of marketing authorization applications that it is likely to receive.
Orphan
designation in the United Kingdom following Brexit is based on the prevalence of the condition in the United Kingdom as opposed to the
current position where prevalence in the European Union is the determinant. It is therefore possible that conditions that are currently
designated as orphan conditions in the United Kingdom will no longer be and that conditions that are not currently designated as orphan
conditions in the European Union will be designated as such in the United Kingdom.
EU
Orphan Drug Designation
The
European Medicines Agency (EMA) and FDA play a central role in facilitating the development and authorization of medicines for rare diseases,
which are termed ‘orphan medicines’ in the medical world. In the EU, sponsors who obtain orphan designation benefit from
protocol assistance, a type of scientific advice specific for designated orphan medicines, and market exclusivity once the medicine is
on the market. Fee reductions are also available depending on the status of the sponsor and the type of service required. When planning
the development of their medicinal product, sponsors should consult the relevant scientific guidelines.
The
general therapeutic strategy for the treatment of AML has not changed substantially over the past 30 years. Excluding APL (which should
be treated with all trans-retinoic acid), AML management is based primarily on induction, incorporating an anthracycline and cytarabine,
and consolidation therapy, and/or allogeneic Hematopoietic Stem Cell Transplantation (alloHSCT). Induction/consolidation therapy leads
to high CRs rates in those who are eligible for treatment and present a favorable risk profile.
Several
novel agents are in various stages of development for the treatment of AML. Novel approaches include antibody-based immunotherapy and
adoptive cell therapy that aim to improve anti-leukaemia T cell function, such as the therapies developed by TCB (OmnImmune®). OmnImmune®
(TCB002) was initially studied in patients with active relapsed or refractory AML who are not eligible or do not consent to high dose
salvage chemotherapy and/or alloHSCT. In July 2019, OmnImmune® (TCB002) was granted ‘orphan medicine’ status from the
FDA for Acute Myeloid Leukaemia (AML). As a follow on to OmnImmune® (TCB002), TCB intends to conduct phase 2b/3 studies to treat
earlier stage AML and expects to commence treating patients in these trials (as OmnImmune® (TCB008-001)) in H1 2022.
Legal
Proceedings
From
time to time, we may be party to litigation that arises in the ordinary course of our business.
In
accordance with the terms of a Convertible Loan Note (‘Note’) on August 9, 2022 (the Conversion Date) the Company issued
183,820 Ordinary Shares and 367,640 listed warrants to the Note holder in full satisfaction of the Note in the aggregate amount of $781,233.
The holder filed a claim in the English courts on June 19, 2023 asserting that notice was provided such that the Company should have
paid it the value of the Note in cash, rather than by settling it through the issuance of Ordinary Shares and listed warrants. The holder
is demanding payment of the face value of the Note, together with interest, (approximately $860,000). The litigation process is in its
early stages and is not expected to conclude until late 2024 or later. The Company has retained English solicitors and is contesting the claim
in its entirety. The Company believes that it acted correctly under the terms of the Note and has accounted for the transaction on that
basis, and that no further amounts are payable to the holder.
Segment
Information
We
operate in a single operating segment and a single reporting segment. Operating segments are defined as components of an enterprise about
which separate financial information is regularly evaluated by the chief operating decision maker function (which is fulfilled by our
chief executive officer) in deciding how to allocate resources and in assessing performance. Our chief executive officer allocates resources
and assesses performance based upon financial information at the level. Since we operate in one operating segment, all required financial
segment information is presented in the financial statements.
Corporate
Information
Our
corporate headquarters and most of our operations, including our research and manufacturing facilities, are located at Maxim 1, 2 Parklands
Way, Holytown, Motherwell, ML1 4WR, United Kingdom. The lease for this space expires February 28, 2029 and covers a total leasable area
of approximately 26,300 square feet. We believe that our office facilities and the production and research facilities in the United Kingdom
are sufficient to meet our current needs.
We
are an “emerging growth company” as defined in the Jumpstart Our Business Startups Act of 2012. We will remain an emerging
growth company until the earlier of: (i) the last day of the fiscal year (a) following the fifth anniversary of the completion of our
IPO, (b) in which we have total annual gross revenue of at least $1.07 billion, or (c) in which we are deemed to be a large accelerated
filer, which means the market value of our common stock that is held by non-affiliates exceeds $700.0 million as of the prior June 30th,
and (ii) the date on which we have issued more than $1.0 billion in non-convertible debt during the prior three-year period.
Available
Information
We
maintain a public website at https://tcbiopharm.com and use our website as a routine channel of distribution of company information,
including press releases, analyst presentations, and supplemental financial information, as a means of disclosing material non-public
information and for complying with our disclosure obligations under Regulation FD. Our website includes an investors’ section through
which we make available, free of charge, our Annual Reports on Form 10-K (for the year ended December 31, 2023), our Annual Reports on
20-F (for periods prior to December 31, 2023), Reports on Form 6-K, as well as any amendments to those reports filed or furnished pursuant
to the Exchange Act as soon as reasonably practicable after we electronically file such material with, or furnish it to, the SEC. Accordingly,
investors should monitor our website in addition to following press releases, filings with the SEC, and public conference calls and webcasts.
Summary
of Risk Factors
Our
business is subject to a number of risks and uncertainties, including those risks discussed at length in the section below titled “Risk
Factors.” These risks include among others the following:
| |
● |
We
have generated operating losses since inception and expect to continue to generate losses. We may never achieve or maintain profitability.
We will continue to require financing to continue to implement our business plan and sustain operations. |
| |
|
|
| |
● |
We,
as well as our independent registered public accounting firm, in relation to our financial position, have expressed substantial doubt
about our ability to continue as a going concern. |
| |
|
|
| |
● |
Our
lack of any approved products and our limited operating history may make it difficult for an investor to evaluate the success of
our business to date and to assess our future viability. |
| |
|
|
| |
● |
GD-T
cell therapies are a novel approach to treating cancers and infectious diseases, which have development risks and will require us
to obtain regulatory approvals for development, testing, commercialization, manufacturing and distribution. We may not achieve all
the required regulatory approvals or approvals may not be obtained as timely as needed. |
| |
● |
Because
GD-T cell therapies are a novel approach, potential side effects, and long-term efficacy, regulatory approval will require considerable
time for trials, data collection, regulatory submissions and funding for the process. |
| |
|
|
| |
● |
Enrolling
patients in clinical trials may be difficult for many reasons, including high screen failure, GD-T cell proliferation capacity, timing,
proximity and availability of clinical sites, perceived risks, and publicity about the success or lack of success in the methods
of treatment. |
| |
|
|
| |
● |
Because
GD-T cell therapies are novel, our research and development and clinical trial results may not support our products intended purposes
and regulatory approval. We are heavily dependent on the success of our lead product candidate (OmnImmune®), and intend to seek
breakthrough therapy designation for some or all of our other therapeutic candidates in due course. |
| |
|
|
| |
● |
Market
opportunities for certain of our product candidates may be limited to those patients who are ineligible for or have failed prior
treatments. This class of patient may be limited in number, difficult to locate and service, require special governmental approval,
and unable to pay or obtain reimbursement. |
| |
|
|
| |
● |
We
rely on many third parties for aspects of our product development and commercialization, such as raw material supply, clinical trials,
obtaining approvals, aspects of manufacturing, development of additional product candidates and distribution. We may not be able
to control these parties and their business practices, such as compliance with good manufacturing requirements or their ability to
supply or service us timely, which will likely disrupt our business. |
| |
|
|
| |
● |
We
face substantial competition: others may discover, develop and/or commercialize competing products before or more successfully than
TCB. |
| |
● |
Even
if we are able to commercialize any product candidates, such drugs may become subject to unfavorable pricing regulations or third-party
coverage and reimbursement policies. Commercialized products may not be adopted by the medical profession. |
| |
|
|
| |
● |
Because
we operate internationally, we are subject to a wide array of regulation of the United Kingdom, European Union and United States.
In addition to regulation surrounding new drug development and their manufacture, distribution and use, we will be subject, for example
to data protection rules relating to medical records, medical and general privacy laws, environmental laws regarding medical waste,
and bribery and corrupt practices law, in addition to all the drug related approval, manufacturing and distribution rules. |
| |
● |
Product
liability claims are frequent in drug development of novel therapies and insurance is mandatory and expensive. The inability to obtain
insurance may prevent product development and claims may surpass our ability to pay and call into question the efficacy of a product
with resulting reputational damage. |
| |
|
|
| |
● |
Protecting
our intellectual property is paramount in our ability to be able to commercialize our products and generate revenues and investment
return for our stockholders. We may not be able to obtain the intellectual property protection we seek due to its cost, requirement
to pursue it in many jurisdictions, challenges by others and patent office rejection. |
| |
|
|
| |
● |
Obtaining
and maintaining our patent protection depends on compliance with various procedural, document submission, fee payment and other requirements
imposed by governmental patent agencies acting in multiple jurisdictions, and our patent protection could be reduced or eliminated
for non-compliance with these requirements. |
| |
|
|
| |
● |
As
part of product development, we may need to license aspects of our research and products from third parties or if our IP is challenged,
we may have to seek license accommodation, any of which may be expensive, limited in scope, or unavailable. |
| |
|
|
| |
● |
We
currently have a limited number of employees, and our future success depends on our ability to retain key executives and to attract,
retain and motivate qualified personnel at all levels. |
| |
● |
We
will need to grow the size and capabilities of our organization, and we may experience difficulties in managing this growth including,
but not limited to, operating as a public company and taking a therapeutic through to market approval and acceptance. |
| |
|
|
| |
● |
We
expect to expand our development and regulatory capabilities and potentially implement sales, marketing and distribution capabilities,
and as a result, we may encounter difficulties in achieving and managing our growth, which could disrupt our operations. We expect
to require further funding for these expansions of activity. |
| |
|
|
| |
● |
We
incur substantial costs as a result of operating as a public company in the United States, and our management is required to devote
substantial time to required SEC compliance and corporate governance practices. |
| |
|
|
| |
● |
If
we fail to maintain proper and effective internal controls, our ability to produce accurate financial statements on a timely basis
could be impaired, which would adversely affect our business and our stock price. |
| |
|
|
| |
● |
Certain
of our existing shareholders, members of our board of directors and senior management maintain the ability to exercise significant
control over us. The interests of investors may conflict with the interests of these other stockholders. |
| |
|
|
| |
● |
Our
ADSs provide rights that are different from directly holding our ordinary shares. The outstanding warrants do not have the rights
of shareholders until exercised. Our warrants form a substantial part of our capitalization, and they have substantial protective
provisions, which may limit our ability to raise capital. |
| |
|
|
| |
● |
Future
sales, or the possibility of future sales, of a substantial number of our ordinary shares, through the additional deposit of ordinary
shares for ADSs, issuances and/or exercises of our warrants, could adversely affect the price of our ADSs or warrants in the market.
After any lock up period, a substantial number of our issued and outstanding ordinary shares will be eligible for trading on the
public securities market by their being deposited with the depositary for ADSs. |
| |
|
|
| |
● |
Shareholder
rights and recourse will be governed by and ultimately determined by Scottish and United Kingdom law and judicial process, which
in many ways are more limited than United States law and practice. Most of our assets are located in the United Kingdom. |
| |
|
|
| |
● |
If
we fail to meet the requirements for continued listing on Nasdaq, our ADSs could be delisted from trading, which would decrease the
liquidity of our ADSs and our ability to raise additional capital. |
Risks
Related to Our Financial Position and Need for Additional Capital
We
have incurred net losses every year since our inception and expect to continue to incur net losses in the future and may never achieve
profitability.
We
have generated losses since our inception in 2013. Since then, we have devoted substantially all of our resources to research and development
efforts relating to our genetically unmodified and genetically engineered GD-T cell candidates, including engaging in activities to manufacture
and supply our GD-T cell candidates for clinical trials, conducting initial clinical trials of our lead candidates, general and administrative
support for these operations, and protecting our intellectual property. Based on our current plans, we do not expect to generate product
or royalty revenues until we obtain marketing approval for, and commercialize, any of our GD-T cell-based candidates.
For
the fiscal years ended December 31, 2023 and 2022, we incurred net losses of £5.9 million and £1.3 million, respectively.
As of December 31, 2023, we had an accumulated deficit of £38.8 million. We expect to continue incurring significant losses as
we continue with our research and development and clinical programs and to incur general and administrative costs associated with our
operations. The extent of funding required to develop our product candidates is difficult to estimate given the novel nature of our GD-T
cell-based cell therapy candidates and their un-proven route to market. Ultimately, our profitability is dependent upon the successful
development, approval, and commercialization of our GD-T cell-based therapeutic candidates and achieving a level of revenues adequate
to support our cost structure. We may never achieve profitability and until we do, we will continue to need to raise additional cash.
Our
ability to generate revenue from sales of our therapeutic candidates and become profitable depends significantly on our success in a
number of factors.
We
continue to focus on development activities for our technologies and implementation of the early parts of our business plan. We may never
achieve commercial success and continue to operate in the research and development stage, without commercially launching any products
at this time. We have limited historical financial data upon which we may base our projected revenue and base our planned operating expenses.
Our limited operating history makes it difficult for potential investors to evaluate our potential product candidates, drug therapies
or prospective operations and business prospects. As a development stage company, we are subject to all the risks inherent in the initial
organization, business development, financing, unexpected expenditures, and complications and delays that often occur in a new business.
Investors should evaluate an investment in us in light of the uncertainties encountered by developing companies in a competitive environment.
There can be no assurance that our efforts will be successful or that we will ultimately be able to attain profitability.
We
have no GD-T cell-based therapeutic candidates approved for commercial sale and have not generated any revenue from sales of our GD-T
cell-based therapeutic candidates, and do not anticipate generating any revenue from sales of our GD-T cell-based therapeutic candidates
until sometime after we receive regulatory approval, if at all, for the commercial sale of a GD-T cell-based therapeutic candidate. We
intend to fund future operations through our existing and future collaboration and licensing agreements for other therapeutic targets
and through additional equity financings. Our ability to generate revenue and achieve profitability depends on our success in many factors,
including:
| |
● |
completing
research regarding, and preclinical and clinical development of, our GD-T cell-based therapeutic candidates; |
| |
|
|
| |
● |
obtaining
regulatory approvals and marketing authorizations for our GD-T cell-based therapeutic candidates for which we complete clinical trials; |
| |
|
|
| |
● |
developing
sustainable and scalable manufacturing and supply processes for our GD-T cell-based therapeutic candidates, including establishing
and maintaining commercially viable supply relationships with third parties and pursuing our own commercial manufacturing capabilities
and infrastructure; |
| |
|
|
| |
● |
launching
and commercializing GD-T cell-based therapeutic candidates for which we obtain regulatory approvals and marketing authorizations,
either directly or with a collaborator or distributor; |
| |
|
|
| |
● |
obtaining
market acceptance of our GD-T cell-based therapeutic candidates as viable treatment options; |
| |
|
|
| |
● |
addressing
any competing technological and market developments; |
| |
|
|
| |
● |
identifying,
assessing, acquiring and/or developing new GD-T cell-based therapeutic candidates; |
| |
|
|
| |
● |
maintaining,
protecting, and expanding our portfolio of intellectual property rights, including patents, trade secrets and know-how; and |
| |
|
|
| |
● |
attracting,
hiring and retaining qualified personnel. |
Even
if one or more of our GD-T cell-based therapeutic candidates is approved for commercial sale, we anticipate incurring significant costs
associated with commercializing any approved GD-T cell-based therapeutic candidate. Our expenses will increase beyond our current expectations
if the U.S. Food and Drug Administration, the FDA, or the United Kingdom Medicines and Healthcare products Regulatory Agency, the MHRA,
or any other regulatory agency require changes to our manufacturing processes or assays, or for us to perform preclinical programs and
clinical or other types of trials in addition to those that we currently anticipate. If we are successful in obtaining regulatory approvals
to market one or more of our GD-T cell-based therapeutic candidates, our revenue will be dependent, in part, upon the size of the markets
in the territories for which we gain regulatory approval, the accepted price for the GD-T cell-based therapeutic candidate, the ability
to get reimbursement at any price, and whether we own the commercial rights for that territory. If the number of our addressable disease
patients is not as significant as we estimate, the indication approved by regulatory authorities is narrower than we expect, or the reasonably
accepted population for treatment is narrowed by competition, physician choice or treatment guidelines, we may not generate significant
revenue from sales or supplies of such GD-T cell-based therapeutic candidates, even if approved. If we are not able to generate revenue
from the sale of any approved GD-T cell-based therapeutic candidates, we may never become profitable.
If
we fail to obtain additional financing as needed, we may be unable to complete the development and commercialization of our GD-T cell-based
product candidates.
Our
operations have required substantial amounts of cash since inception. We expect to continue to spend substantial amounts to continue
the development of our GD-T cell-based therapeutic candidates, including for clinical trials. We expect to use our operating capital
to advance and accelerate the clinical development of our therapeutic candidates, therefore, changing circumstances beyond our control
may cause us to increase our spending significantly faster than we currently anticipate, we believe we will require additional capital,
likely in significant amounts, for the further development and commercialization of our GD-T cell-based therapeutic candidates.
We
cannot be certain that additional funding will be available on acceptable terms, or at all. We have no committed source of additional
capital. If we are unable to raise additional capital in sufficient amounts or on terms acceptable to us, we may have to significantly
delay, scale back or discontinue the development or commercialization of our GD-T cell-based therapeutic candidates or other research
and development initiatives. Our license and supply agreements may also be terminated if we are unable to meet the milestone obligations
under these agreements. We could be required to seek collaborators for our GD-T cell-based therapeutic candidates at an earlier stage
than otherwise would be desirable or on terms that are less favorable than might otherwise be available or relinquish or license on unfavorable
terms our rights to our GD-T cell-based therapeutic candidates in markets where we otherwise would seek to pursue development or commercialization
ourselves. Any of the above events could significantly harm our business, prospects, financial condition and results of operations and
cause the price of either of our ADSs or Warrants, or both, to decline.
We,
as well as our independent registered public accounting firm, have expressed substantial doubt about our ability to continue as a going
concern.
Our
recurring losses from operations and negative cash flow raise substantial doubt about our ability to continue as a going concern. As
a result, our independent registered public accounting firm included an explanatory paragraph in its report on our financial statements
for the year ended December 31, 2023 with respect to this uncertainty.
Our
ability to continue as a going concern ultimately is dependent upon our generating cash flow from sales that are sufficient to fund
operations or finding adequate financing to support our operations. To date, we have had no product revenues and relied on
equity-based financing from the sale of securities subscribed by our founders and related parties and in various private placements,
and receipts from collaboration partners. Our research and development plans may not be successful in creating a marketable product,
and our business plan may not be successful in achieving a sustainable business and generating revenues. We completed our initial
public offering (“IPO”) in February 2022, which, together with additional funds that we have raised since then and funds
we plan to raise during 2024, we believe will provide funding to enable us to progress our planned clinical trial program in our
lead product, OmnImmune®, into 2024 and beyond. We have no firm arrangements in place for all the anticipated, required
financing to be able to fund our operations during and beyond 2024 and otherwise to implement fully our business plan. If we are
unable to continue our operations as planned, we may have to curtail some or all of our business plan and operations. In such case, investors
will lose all or a portion of their investment.
We
anticipate needing additional financing over the longer term to execute our business plan and fund operations, which additional financing
may not be available on reasonable terms or at all.
The
proceeds from our February 2022 IPO, together with additional capital raised since then and funds that we plan to continue to raise during
2024, are expected to provide capital to further develop our drug product candidates and fund our overall business plan until at least
the end of 2024. We will require additional capital in the future to fully develop our technologies and potential products to the stage
of a commercial launch. We cannot give now any indication of the amount of future funding that we will need or give any assurance that
we will be able to obtain all the necessary funding that we may need. We may pursue additional funding through various financing sources,
including the private and public sale of our equity and debt securities, licensing fees for our product candidates, joint ventures with
capital partners and project type financing. We also may seek government-based financing, such as development and research grants. There
can be no assurance that funds will be available on commercially reasonable terms, if at all. If financing is not available on satisfactory
terms, we may be unable to pursue further our business plan and we may be unable to continue operations, in which case investors may
lose their entire investment. Alternatively, we may consider changes in our business plan that might enable us to achieve aspects of
our business objectives and lead to some commercial success with a smaller amount of capital, but we cannot assure that changes in our
business plan will result in revenues or maintain value in an investment in the company.
On March 22, 2024, the Group had cash on hand of $1.7 million (£1.4 million), which will not be sufficient
to enable the Group to meet the cash requirements required to enable it to conduct its business plan through the going concern period
(being to April 1, 2025) (“Going Concern Period”). With existing resources, we expect to be able to fund current operations
to May 2024.
Risks
Related to Development, Clinical Testing and Commercialization of Our Investigational Therapies and Any Future Therapeutic Candidates
Our
GD-T cell therapies represent a novel approach to cancer treatment that could result in heightened regulatory scrutiny, delays in clinical
development, or delays in or our ability to achieve regulatory approval or commercialization of our therapeutic candidates.
Our
products are novel cancer treatment approaches that carry inherent development risks. We are therefore constantly evaluating and adapting
our therapeutic candidates following the results obtained during development work and the ongoing clinical trials. Further development,
characterization and evaluation may be required, depending on the results obtained, in particular where such results suggest any potential
safety risk for patients. The need to develop further assays, or to modify in any way the protocols related to our therapeutic candidates
to improve safety or effectiveness, may delay a clinical program, regulatory approval or commercialization, if approved at all, of any
therapeutic candidate. Consequently, this may have a material impact on our ability to receive milestone payments and/or generate revenues
from our therapeutic candidates and raise equity capital. In addition, given the novelty of our GD-T cell therapeutic candidates, the
end users and medical personnel require a substantial amount of education and training in their administration of our cell therapy. Regulatory
authorities have very limited experience with commercial cell therapies for disease treatment. As a result, regulators may be more risk
averse or require substantial dialogue and education as part of the normal regulatory approval process for each stage of development
of our therapeutic candidates.
GD-T
cell therapy creates significantly increased risk in terms of side-effect profile, ability to satisfy regulatory requirements associated
with clinical trials, and the long-term efficacy of administered cells.
Development
of a pharmaceutical or biologic therapy product has inherent risks based on differences in patient population and responses to therapy
and treatment. The mechanism of action and impact on other systems and tissues within the human body following administration of GD-T
cell therapy products is not completely understood, which means that we cannot predict the long-term effects of treatment with the GD-T
cell therapy product. We are aware that certain patients may not respond to GD-T cell therapy and other patients may relapse. The percentage
of the patient population in which these events may occur is unknown, but the inability of patients to respond and the possibility of
relapse may impact our ability to conduct clinical trials, to obtain regulatory approvals, if at all, and to successfully commercialize
our therapeutic products.
Our
GD-T cell therapeutic candidates and their application are not fully scientifically understood and are still undergoing validation and
investigation. The utility of our GD-T cell products may depend on persistence, potency, durability and infiltration capacity of the
GD-T cells within a patient’s body. The level of persistence and the factors affecting such persistence, potency and infiltration
capacity in patients are not completely understood, which presents an additional risk to the ongoing development and use of our therapeutic
candidates. Certain steps involved in validating and carrying out testing require access to samples (for example tissue samples or cell
samples) from third parties. Such samples may be obtained from universities or research institutions and will often be provided subject
to satisfaction of certain terms and conditions. There can be no guarantee that we will be able to obtain samples in sufficient quantities
to enable development of and use of the full preclinical safety testing program for CAR-T therapeutic candidates undergoing development.
In addition, the terms under which such samples are available may not be acceptable to us or may restrict our use of any generated results
or require us to make payments to third parties.
Our
products, before they can be commercialized, will require regulatory approval.
We
cannot commercialize a product candidate until the appropriate regulatory authorities have reviewed and approved the product candidate.
Approval by the FDA, the MHRA and comparable other regulatory authorities is lengthy and unpredictable, and depends upon numerous factors.
Approval policies, regulations, or the type and amount of clinical data necessary to gain approval may change during the course of a
product candidate’s clinical development and may vary among jurisdictions, which may cause delays in the approval or the decision
not to approve an application. We have not obtained commercialization regulatory approval for any product candidate, and it is possible
that any of our product candidates will never obtain regulatory approval.
Applications
for product candidates we may develop could fail to receive regulatory approval for many reasons, including but not limited to:
| |
● |
our
inability to demonstrate to the satisfaction of the regulatory authorities that a product candidate we develop is safe and effective; |
| |
|
|
| |
● |
the
regulatory authorities may disagree with the design or implementation of our clinical trials; |
| |
|
|
| |
● |
the
population studied in the clinical program may not be sufficiently broad or representative to assure safety in the full population
for which we seek approval; |
| |
|
|
| |
● |
the
regulatory authorities’ requirement for additional preclinical studies or clinical trials; |
| |
|
|
| |
● |
the
regulatory authorities may disagree with our interpretation of data from nonclinical studies or clinical trials; |
| |
|
|
| |
● |
the
data collected from clinical trials may not be sufficient to support the submission of a new drug application, or NDA, or other submission
for regulatory approval; |
| |
● |
we
may be unable to demonstrate to the regulatory authorities that a product candidate’s risk-benefit ratio for its proposed indication
is acceptable; |
| |
|
|
| |
● |
the
regulatory authorities may fail to approve the manufacturing processes, test procedures and specifications, or facilities of third-party
manufacturers with which we contract for clinical and commercial supplies; and |
| |
|
|
| |
● |
the
approval policies or regulations of the regulatory authorities may change in a manner that renders our clinical trial design or data
insufficient for approval. |
The
lengthy approval process, as well as the unpredictability of the results of clinical trials, may result in our failing to obtain regulatory
approval to market a product candidate in the United States, the UK, the EU or elsewhere, which would significantly harm our business,
prospects, financial condition and results of operations.
We
may encounter substantial delays in completing our clinical trials, which in turn will result in additional costs and may ultimately
prevent successful or timely completion of the clinical development and commercialization of our product candidates.
We
must conduct extensive clinical trials to demonstrate the safety and efficacy of the product candidates in humans before commercialization.
Clinical testing is expensive, time-consuming and uncertain as to outcome. We cannot guarantee that any clinical trials will be conducted
as planned or completed on schedule, if at all. A failure of one or more clinical trials can occur at any stage of testing. Events that
may prevent successful or timely completion of clinical development include:
| |
● |
delays
in reaching, or any failure to reach, a consensus with regulatory agencies on study design; |
| |
|
|
| |
● |
delays
in obtaining FDA required Institutional Review Board, or IRB, approval at each clinical trial site; |
| |
|
|
| |
● |
delays
in recruiting a sufficient number of suitable patients to participate in our clinical trials; |
| |
|
|
| |
● |
imposition
of a clinical hold by regulatory agencies, after an inspection of our clinical trial operations or study sites; |
| |
|
|
| |
● |
failure
by third parties or us to adhere to clinical trial, regulatory or legal requirements; |
| |
|
|
| |
● |
failure
to perform in accordance with good clinical practices (“GCP”), or applicable regulatory guidelines in other countries; |
| |
|
|
| |
● |
delays
in the testing, validation, manufacturing and delivery of sufficient quantities of our product candidates to the clinical sites; |
| |
|
|
| |
● |
delays
in having patients’ complete participation in a study or return for post-treatment follow-up; |
| |
|
|
| |
● |
clinical
trial sites or patients dropping out of a trial; |
| |
|
|
| |
● |
delay
or failure to address any patient safety concerns that arise during the course of a trial; |
| |
|
|
| |
● |
unanticipated
costs or increases in costs of clinical trials of our product candidates; |
| |
|
|
| |
● |
occurrence
of serious adverse events associated with the product candidate that are viewed to outweigh its potential benefits; or |
| |
|
|
| |
● |
changes
in regulatory requirements and guidance that require amending or submitting new clinical protocols. |
We
could also encounter delays if a clinical trial is suspended or terminated by us or by regulators and related reviewing authorities such
as IRBs of the institutions in which such trials are being conducted, by an independent Safety Review Board. Suspension or termination
of a clinical trial might be due to a number of factors, including failure to conduct the clinical trial in accordance with regulatory
requirements or our clinical protocols, inspection of the clinical trial operations or trial site resulting in the imposition of a clinical
hold, unforeseen safety issues or adverse side effects, or failure to demonstrate a benefit from using a therapy. In addition, if we
make manufacturing or formulation changes to our product candidates, we may need to conduct additional studies to bridge our modified
product candidates to earlier versions. In addition, any delays in completing our clinical trials will increase our costs, slow down
our product candidate development and approval process and jeopardize our ability to obtain regulatory approvals, commence product sales
and generate revenues. Any of these occurrences may significantly harm our business, prospects, financial condition and results of operations.
Manufacturing
and administering our GD-T cell-based therapeutic candidates is complex, and we may encounter difficulties in production, particularly
with respect to process development or scaling-up of our manufacturing capabilities. If we encounter such difficulties, our ability to
supply of our GD-T cell therapeutic candidates for clinical trials or for commercial purposes could be delayed or stopped.
Manufacturing
and administrating our GD-T cell-based therapeutics candidates is complex and highly regulated. The manufacture process of our GD-T cell-based
therapeutics involves complex processes, including peripheral blood mononuclear cell isolation from leukapheresis material, stimulation
of the GD-T cells, expansion of the cells to obtain a desired dose, and ultimately infusion of the cells to the patient’s body.
On occasions the GD-T cell therapeutic could be genetically modified, which could involve manufacturing of lentiviral vectors containing
the gene of our interest (for example Chimeric Antigen Receptor) and transducing the cells or a method such as electroporation or nucleofection
of a plasmid containing the gene of interest to the cells. As a result of the complexities, our manufacturing and supply costs are likely
to be higher than those in more traditional manufacturing processes and the manufacturing process is less reliable and more difficult
to reproduce. Our manufacturing process is, and will be, susceptible to product loss or failure due to logistical issues, including manufacturing
issues associated with the differences in patients’ white blood cells, interruptions in the manufacturing process, contamination,
equipment or reagent failure, supplier error and variability in GD-T cell-based therapeutic candidate and patient characteristics. Even
minor deviations from normal manufacturing processes could result in reduced production yields, product defects, and other supply disruptions.
If microbial, viral or other contaminations are discovered in our GD-T cell-based therapeutic candidates or in the manufacturing facilities
in which our GD-T cell based therapeutic candidates are made or administered, the manufacturing facilities may need to be closed for
an extended period of time to investigate and remedy the contamination. As our GD-T cell-based therapeutic candidates progress through
preclinical programs and clinical trials towards approval and commercialization, it is expected that various aspects of the manufacturing
and administration process will be altered in an effort to optimize processes and results.
We
have identified some improvements to our manufacturing and administration processes, but these changes may not achieve the intended objectives,
and could cause our GD-T cell-based therapeutic candidates to perform differently and affect the results of planned clinical trials or
other future clinical trials. The changes may require amendments to be made to regulatory applications which may further delay the timeframes
under which modified manufacturing processes can be used for any GD-T cell-based therapeutic candidate. For example, we are planning
to introduce automated enclosed systems to our production process. This will require development work to ensure that these modifications
do not alter the characteristics of the product. If the GD-T cell-based therapeutic candidate manufactured under the new process has
a worse safety or efficacy profile than the prior investigational product, we may need to re-evaluate the use of that manufacturing process,
which could significantly delay the progress of our clinical trials.
Developing
a commercially viable process is a difficult and uncertain task and there are risks associated with scaling to the level required for
advanced clinical trials or commercialization, including, among others, increased costs, potential problems with process scale-out, process
reproducibility, stability issues, lot consistency, and timely availability of reagents or raw materials. We may ultimately be unable
to reduce the expenses associated with our GD-T cell-based therapeutic candidates to levels that will allow us to achieve a profitable
return on investment. If we are unable to demonstrate that our commercial scale product is comparable to the product used in clinical
trials, we may not receive regulatory approval for that product without additional clinical trials. Even if we are successful, our manufacturing
capabilities could be affected by increased costs, unexpected delays, equipment failures, labor shortages, natural disasters, power failures
and numerous other factors that could prevent us from realizing the intended benefits of our manufacturing strategy, which in turn could
have a material adverse effect on our business.
We
may seek expedited approval in the European Union and United States for our therapeutic candidates, but we may not be able to obtain
or maintain such designation.
The
FDA and the European Medicines Agency, the EMA, have established programs to expedite drug development and regulatory review. The FDA
has four main expedited programs: fast track (introduced in 1987), accelerated approval (1992), priority review (1992), and breakthrough
therapy (2012). A priority review designation in North America will direct overall attention and resources to the evaluation of applications
for drugs that, if approved, would be significant improvements in the safety or effectiveness of the treatment, diagnosis, or prevention
of serious conditions when compared to standard applications. Significant improvement may be demonstrated by the following examples:
| |
● |
evidence
of increased effectiveness in treatment, prevention, or diagnosis of condition; |
| |
● |
elimination
or substantial reduction of a treatment-limiting drug reaction; |
| |
|
|
| |
● |
documented
enhancement of patient compliance that is expected to lead to an improvement in serious outcomes; or |
| |
|
|
| |
● |
evidence
of safety and effectiveness in a new subpopulation. |
We
intend to seek breakthrough therapy designation for some or all of our therapeutic candidates, but there can be no assurance that we
will receive breakthrough therapy designation. Additionally, other treatments from competing companies may obtain the designations and
impact our ability to develop and commercialize our therapeutic candidates, which may adversely impact our business, financial condition
or results of operation. We may also seek fast track designation. If a drug or biologic candidate is intended for the treatment of a
serious or life-threatening condition or disease and the drug demonstrates the potential to address unmet medical needs for the condition,
the sponsor may apply for fast track designation. Under the fast track program, the sponsor of a new drug or biologic candidate may request
that the FDA designate the candidate for a specific indication as a fast track drug or biologic concurrent with, or after, the submission
of the IND for the candidate. The FDA must determine if the drug or biologic candidate qualifies for fast track designation within 60
days of receipt of the sponsor’s request. Even if we do apply for and receive fast track designation, we may not experience a faster
development, review or approval process compared to conventional FDA procedures. The FDA may withdraw fast track designation if it believes
that the designation is no longer supported by data from our clinical development program. We may also seek accelerated approval for
products that have obtained fast track designation. Under the FDA’s fast track and accelerated approval programs, the FDA may approve
a drug or biologic for a serious or life-threatening illness that provides meaningful therapeutic benefit to patients over existing treatments
based upon a surrogate endpoint that is reasonably likely to predict clinical benefit, or on a clinical endpoint that can be measured
earlier than irreversible morbidity or mortality, that is reasonably likely to predict an effect on irreversible morbidity or mortality
or other clinical benefit, taking into account the severity, rarity, or prevalence of the condition and the availability or lack of alternative
treatments. For drugs granted accelerated approval, post-marketing confirmatory trials have been required to describe the anticipated
effect on irreversible morbidity or mortality or other clinical benefit. These confirmatory trials must be completed with due diligence.
The
EMA has three programs, the accelerated assessment (2005), conditional marketing authorization (2006), and the Priority Medicines (PRIME)
scheme (2016). These programs are intended to prioritize the most important medicines for faster access by patients. As part of its marketing
authorization process, the EMA may grant conditional marketing authorizations for certain categories of medicinal products on the basis
of less complete data than is normally required, when doing so may meet unmet medical needs of patients and may serve the interest of
public health. In these cases, it is possible for the Committee for Medicinal Products for Human Use, the CHMP, to recommend the granting
of a marketing authorization, subject to certain specific obligations to be reviewed annually, which is referred to as a conditional
marketing authorization. This may apply to medicinal products for human use that fall under the jurisdiction of the EMA, including those
that aim at the treatment, the prevention, or the medical diagnosis of seriously debilitating diseases or life-threatening diseases and
those designated as orphan medicinal products. A conditional marketing authorization may be granted when the CHMP finds that, although
comprehensive clinical data referring to the safety and efficacy of the medicinal product have not been supplied, the risk-benefit balance
of the medicinal product is positive. The granting of a conditional marketing authorization is restricted to situations in which only
the clinical part of the application is not yet fully complete. Incomplete preclinical or quality data may only be accepted if duly justified
and only in the case of a product intended to be used in emergency situations in response to public-health threats. Conditional marketing
authorizations are valid for one year, on a renewable basis. The holder will be required to complete ongoing trials or to conduct new
trials with a view to confirming that the benefit-risk balance is positive. In addition, specific obligations may be imposed in relation
to the collection of pharmacovigilance data. Granting a conditional marketing authorization allows medicines to reach patients with unmet
medical needs earlier than might otherwise be the case and will ensure that additional data on a product are generated, submitted, assessed
and acted upon. Although we may seek a conditional marketing authorization for our therapeutic candidates, the EMA or CHMP may ultimately
not agree that the requirements for conditional marketing authorization have been satisfied and hence delay the commercialization of
our therapeutic candidates.
In
the European Union, accelerated assessment can reduce the timeframe for EMA’s CHMP to review a marketing-authorization application.
Applications may be eligible for accelerated assessment if the CHMP decides the product is of major interest for public health and therapeutic
innovation. The evaluation of a marketing-authorization application can take up to 210 days. However, the CHMP can reduce the timeframe
to 150 days if the applicant can provide sufficient justification for an accelerated assessment. The Priority Medicines (PRIME) scheme
was introduced by the EMA in 2016 to support the development of medicines addressing unmet medical needs which offer a therapeutic advantage
over existing treatments. To be accepted in the PRIME scheme, the treatments must meet the eligibility criteria for accelerated assessment
including a strongly substantiated mechanism of action, supportive preclinical data, and first-in-human tolerance data. PRIME has been
compared to the U.S. Breakthrough Therapy Designation.
Withdrawal
of expedited approval will delay trials and likely increase cost.
The
FDA or EMA may withdraw expedited approval of our therapeutic candidate or indication approved under the accelerated approval pathway
if, for example:
| |
● |
the
trial or trials required to verify the predicted clinical benefit of our therapeutic candidate fail to verify such benefit or do
not demonstrate sufficient clinical benefit to justify the risks associated with the drug; |
| |
|
|
| |
● |
other
evidence demonstrates that our therapeutic candidate is not shown to be safe or effective under the conditions of use; |
| |
|
|
| |
● |
we
fail to conduct any required post-approval trial of our therapeutic candidate with due diligence; or |
| |
|
|
| |
● |
we
disseminate false or misleading promotional materials relating to the relevant therapeutic candidate. |
Obtaining
and maintaining regulatory approval of our therapeutic candidates in one jurisdiction does not mean that we will be successful in obtaining
regulatory approval of our therapeutic candidates in other jurisdictions.
We
plan on submit marketing applications in multiple jurisdictions and countries, including the UK, the EU and the United States. Regulatory
authorities in each jurisdiction have requirements for approval of therapeutic candidates with which we must comply prior to marketing
in those jurisdictions. Obtaining regulatory approvals and compliance with regulatory requirements of multiple jurisdictions and countries
could result in significant delays, difficulties and costs for us and could delay or prevent the introduction of our therapeutic candidates
in certain countries. If we fail to comply with the regulatory requirements in international markets and/or receive applicable marketing
approvals, our target market will be reduced and our ability to realize the full market potential of our therapeutic candidates will
be harmed.
Obtaining
and maintaining regulatory approval of our therapeutic candidates in one jurisdiction does not guarantee that we will be able to obtain
or maintain regulatory approval in any other jurisdiction, while a failure or delay in obtaining regulatory approval in one jurisdiction
may have a negative effect on the regulatory approval process in others. For example, even if the FDA grants marketing approval of our
key products in the United States, comparable regulatory authorities in other jurisdictions must also approve the manufacturing, marketing
and promotion of our products in those countries. Approval procedures vary among jurisdictions and may require additional preclinical
programs or clinical trials. In many jurisdictions a therapeutic candidate must be approved for reimbursement before it can be approved
for sale in that jurisdiction. In some cases, the price that we intend to charge for our therapeutic candidates is also subject to approval.
We
may face difficulty in enrolling patients in our clinical trials.
We
may find it difficult to enroll patients in our clinical trials. For example, in our TCB-001 clinical trial we experienced a high screen
failure rate. Identifying and qualifying patients, including testing of patients for their GD-T cells’ proliferation capacity,
to participate in clinical trials of our therapeutic candidates, are critical to our success. The timing of our current and future clinical
trials depends on the speed at which we can recruit patients to participate in testing our therapeutic candidates. If patients are unwilling
to participate in our trial(s) because of negative publicity from adverse reactions or for other reasons, including competitive clinical
trials for similar patient populations, the timeline for recruiting patients, conducting trials and obtaining regulatory approval of
potential products may be delayed or prevented. These delays could result in increased costs, delays in advancing our product development,
delays in testing the effectiveness of our technology or termination of the clinical trials altogether. We may not be able to identify,
recruit and enroll a sufficient number of patients, or those with required or desired characteristics to achieve sufficient diversity
in a given trial in order to complete our clinical trials in a timely manner. Patient enrolment is affected by factors including:
| |
● |
eligibility
criteria for the trial in question; |
| |
|
|
| |
● |
severity
of the disease under investigation; |
| |
|
|
| |
● |
design
of the trial protocol; |
| |
|
|
| |
● |
trial
duration and number and complexity of visits and procedures; |
| |
|
|
| |
● |
size
of the patient population; |
| |
|
|
| |
● |
perceived
risks and benefits of the therapeutic candidate under trial; |
| |
|
|
| |
● |
novelty
of the therapeutic candidate and acceptance by oncologists; |
| |
|
|
| |
● |
proximity
and availability of clinical trial sites for prospective patients; |
| |
|
|
| |
● |
availability
of competing therapies and clinical trials; |
| |
|
|
| |
● |
efforts
to facilitate timely enrolment in clinical trials; |
| |
|
|
| |
● |
patient
referral practices of physicians; |
| |
|
|
| |
● |
ability
to monitor patients adequately during and after treatment; and |
| |
|
|
| |
● |
macro
political or economic factors that impact the availability and efficiency of available trial centers. |
If
we have difficulty enrolling a sufficient number of patients to conduct our clinical trials as planned, we may need to delay, limit or
terminate ongoing or planned clinical trials, any of which would have an adverse effect on our business.
The
outcome of clinical trials is uncertain and our clinical trials may fail to demonstrate adequately the safety and efficacy of any of
our T cell therapeutic candidates, which would prevent or delay regulatory approval and commercialization.
There
is a risk in any clinical trial that side effects from our therapeutic candidates will require a hold on, or termination of, our clinical
program(s) or further adjustments to our clinical program(s) in order to progress our therapeutic candidates. Our T cell therapeutic
candidates will require evidence that they are safe before permitting clinical trials to commence and evidence that the therapeutic candidates
are safe and effective before granting any regulatory approval. In particular, because our therapeutic candidates are subject to regulation
as biological products, we will need to demonstrate that they are safe, pure and potent for use in each target indication. The therapeutic
candidate must demonstrate an acceptable risk versus benefit profile in its intended patient population and for its intended use. The
risk/benefit profile required for product licensure will vary depending on these factors and may include not only the ability to show
tumor shrinkage, but also adequate duration of response, a delay in the progression of the disease and/or an improvement in survival.
For example, response rates from the use of our therapeutic candidates will not be sufficient to obtain regulatory approval unless we
can also show an adequate duration of response.
We
may not be able to submit INDs, or the foreign equivalent outside of the United States, to continue our CAR-T clinical trials.
We
are currently conducting preclinical development of our CAR-T therapeutic candidates. Progression of our CAR-T therapeutic candidates
from pre-clinical to clinical development (first-in-human, phase 1) is inherently risky and dependent on the results obtained in preclinical
programs, the results of other clinical programs and results of third-party programs that utilize common components used for production
and administration of our therapeutic candidates. If results are not available when expected or problems are identified during therapy
development, we may experience significant delays in development of pipeline products and of existing clinical programs, which may impact
our ability to receive regulatory approval. This may also impact our ability to achieve certain financial milestones and the expected
timeframes to market any of our therapeutic candidates. Failure to submit further INDs or the foreign equivalent and commence additional
clinical programs will significantly limit our opportunity to generate revenue.
Our
research and development efforts may not result in the progression of our product candidates into clinical trials.
Our
research and development efforts and our selection of the product candidates to pursue remain subject to all of the risks associated
with the development of new treatment modalities. Development of the underlying technology and appropriate assay development may be affected
by unanticipated technical or other problems, among other development and research issues, and the possible insufficiency of funds needed
in order to complete development of these products. Safety, regulatory and efficacy issues, clinical hurdles or challenges also may result
in delays and cause us to incur additional expenses that will increase our need for capital and result in additional losses. If we cannot
complete, or if we experience significant delays in developing our medical products for use in potential commercial applications, particularly
after incurring significant expenditures, our business may fail and investors may lose the entirety of their investment.
We
will need to obtain regulatory approval for our product candidates, which is time consuming, costly and complicated. We may not obtain
regulatory approval.
We
cannot commercialize a product candidate until the appropriate regulatory authorities have reviewed and approved the product candidate.
Approval by the FDA and comparable foreign regulatory authorities is lengthy and unpredictable, and depends upon numerous factors. Approval
policies, regulations, or the type and amount of clinical data necessary to gain approval may change during the course of a product candidate’s
clinical development and may vary among jurisdictions, which may cause delays in the approval or the decision not to approve an application.
We have not obtained regulatory approval for any product candidate, and it is possible that any of our product candidates will never
obtain regulatory approval.
Applications
for product candidates we may develop could fail to receive regulatory approval for many reasons. For example, under FDA regulation,
approval may not be obtained for many reasons such as:
| |
● |
our
inability to demonstrate to the satisfaction of the FDA or comparable foreign regulatory authorities that a product candidate we
may develop is safe and effective; |
| |
|
|
| |
● |
the
FDA or comparable foreign regulatory authorities may disagree with the design or implementation of our clinical trials; |
| |
|
|
| |
● |
the
population studied in the clinical program may not be sufficiently broad or representative to assure safety in the full population
for which we seek approval; |
| |
|
|
| |
● |
the
FDA’s or comparable foreign regulatory authorities’ requirement for additional preclinical studies or clinical trials; |
| |
|
|
| |
● |
the
FDA or comparable foreign regulatory authorities may disagree with our interpretation of data from nonclinical studies or clinical
trials; |
| |
|
|
| |
● |
the
data collected from clinical trials of our product candidates may not be sufficient to support the submission of a new drug application,
or NDA, or other submission for regulatory approval in the United States or elsewhere; |
| |
|
|
| |
● |
we
may be unable to demonstrate to the FDA or comparable foreign regulatory authorities that a product candidate’s risk-benefit
ratio for its proposed indication is acceptable; |
| |
|
|
| |
● |
the
FDA or comparable foreign regulatory authorities may fail to approve the manufacturing processes, test procedures and specifications,
or facilities of third-party manufacturers with which we contract for clinical and commercial supplies; and |
| |
|
|
| |
● |
the
approval policies or regulations of the FDA or comparable foreign regulatory authorities may change in a manner that renders our
clinical trial design or data insufficient for approval. |
The
lengthy approval process, as well as the unpredictability of the results of clinical trials, may result in our failing to obtain regulatory
approval to market a product candidate in the United States or elsewhere, which would significantly harm our business, prospects, financial
condition and results of operations.
We
are heavily dependent on the success of our lead product candidate, OmnImmune®, and our subsequent product development program, If
we are unable to successfully complete clinical development, obtain regulatory approval for, or commercialize these products, or experience
delays in doing so, our business will be materially harmed.
Our
future success is dependent on our ability to successfully develop, obtain regulatory approval for, and commercialize our aforementioned
lead product candidate which is in its clinical development phase. Before we can generate any revenues from sales of products, we will
be required to conduct additional clinical development and conduct longer-term clinical trials and, if our clinical trial of these products
is successful, we will be required to seek and obtain regulatory approval, secure adequate manufacturing supply to support larger clinical
trials and commercial sales and build a commercial organization, or to work with commercialization partners. Further, the success of
these products will depend on patent and trade secret protection, acceptance of these products by patients, the medical community and
third-party payers, its ability to compete with other therapies, healthcare coverage and reimbursement, and maintenance of an acceptable
safety profile following approval, among other factors. If we do not achieve any of these factors in a timely manner or at all, we could
experience significant delays or an inability to successfully commercialize our products which would materially harm our business.
Laboratory
conditions differ from clinical conditions and commercial conditions, which could affect the effectiveness of our potential products.
Failures to effectively move from laboratory to the field would harm our business.
Observations
and developments that may be achievable under laboratory circumstances may not be replicated in commercial settings or in the use of
any of the proposed products in the field. The failure of our product candidates under development or other future product candidates
to be able to be tested, approved and manufactured in available manufacturing facilities or to be able to meet the demands of users in
the field would harm our business.
Results
of earlier studies may not be predictive of future clinical trial results, and initial studies may not establish an adequate safety or
efficacy profile for our drugs and other product candidates that we may pursue to justify proceeding to advanced clinical trials or an
application for regulatory approval.
The
results of preclinical studies and clinical trials of our product candidates may not be predictive of the results of later-stage clinical
trials, and interim results of a clinical trial do not necessarily predict final results. Additionally, any positive results generated
in our Phase 1b/2a clinical trials in adults would not ensure that we will achieve similar results in larger, pivotal clinical trials
or in clinical trials in general populations. In addition, preclinical and clinical data are often susceptible to various interpretations
and analyses, and many companies that have believed their product candidates performed satisfactorily in preclinical studies and clinical
trials have nonetheless failed to obtain marketing approval. Product candidates in later stages of clinical trials may fail to show the
desired safety and efficacy despite having progressed through nonclinical studies and initial clinical trials. A number of companies
in the biopharmaceutical industry have suffered significant setbacks in advanced clinical trials due to lack of efficacy or adverse safety
profiles, notwithstanding promising results in earlier studies, and we cannot be certain that we will not face similar setbacks. Even
if early-stage clinical trials are successful, we may need to conduct additional clinical trials for product candidates in additional
patient populations or under different treatment conditions before we are able to seek approvals from the FDA and regulatory authorities
outside the United States to market and sell these product candidates. Our failure to demonstrate the required characteristics to support
marketing approval for our product candidates in any ongoing or future clinical trials would substantially harm our business, prospects,
financial condition and results of operations.
We
manufacture and test all our therapeutic candidates in-house, and may experience logistic issues.
The
manufacture, testing and release of TCB’s cell therapies for clinical trials may not meet with the regulatory requirements and
result in the delay of clinical trials. Logistical issues which may prevent timely completion of manufacture and testing include:
| |
● |
failure
in integrity of facility infrastructure; |
| |
|
|
| |
● |
failure
of High Efficiency Particulate Absorbing (HEPA) filters to prevent airborne cross-contamination; |
| |
|
|
| |
● |
delays
in the procuring test materials/reagents due to supplier, shipping issues or discontinued supply; |
| |
|
|
| |
● |
failure
by third parties to notify a change in material product specifications that are not GMP (“Good Manufacturing Practice”)
compliant; |
| |
|
|
| |
● |
equipment
failure within production, quality control and stores; |
| |
|
|
| |
● |
failure
of quality control equipment; |
| |
|
|
| |
● |
delays
in cleanroom supplies from third parties such as PPE or cleaning reagents; |
| |
|
|
| |
● |
failure
in the cleanroom resulting in insufficient quantities of our product candidates being available to the clinical sites; |
| |
|
|
| |
● |
increase
in our costs of materials; |
| |
|
|
| |
● |
failure
of third party specialist couriers to deliver the product to clinical sites; |
| |
|
|
| |
● |
failure
due to resource issues associated with personnel illness; and |
| |
|
|
| |
● |
failure
in recruitment of cleanroom operators and quality staff as we progress through clinical trials. |
We
conduct and manage clinical studies using internal staff trained to perform such studies and loss of these staff may delay our clinical
program.
We
are highly dependent upon the principal members of our management team and the members of our scientific team. These people have significant
experience and knowledge within our operational sector, and the loss of any team member could impair our ability to design, identify,
and develop clinical trials, new intellectual property and new scientific or product ideas.
We
expect to operate in a highly competitive, ever-evolving, market.
The
broader market for our products is becoming more focused and potentially more competitive. Over time, we believe this field will become
subject to more rapid change and new drugs, therapies and other products will emerge. We may not be able to compete effectively against
these companies or their products. We may find ourselves in competition with companies that have competitive advantages over us, such
as:
| |
● |
significantly
greater name recognition; |
| |
|
|
| |
● |
established
relations with healthcare professionals, customers and third-party payors; |
| |
● |
established
distribution networks; |
| |
|
|
| |
● |
additional
lines of products, and the ability to offer rebates, higher discounts or incentives to gain a competitive advantage; |
| |
|
|
| |
● |
greater
experience in conducting research and development, manufacturing, clinical trials, obtaining regulatory approval for products, and
marketing approved products; and |
| |
|
|
| |
● |
greater
financial and human resources for product development, sales and marketing, and patent litigation. |
Many
of our competitors, either alone or with their strategic collaborators, have substantially greater financial, technical and human resources
than we do. Accordingly, our competitors may be more successful than we are in obtaining approval for treatments and achieving widespread
market acceptance and may render our treatments obsolete or non-competitive. Mergers and acquisitions in the biotechnology and pharmaceutical
industries may result in even more resources being concentrated among a smaller number of our competitors. These competitors also compete
with us in recruiting and retaining qualified scientific and management personnel and establishing clinical study sites and patient registration
for clinical studies, as well as in acquiring technologies complementary to, or necessary for, our programs. Smaller or early-stage companies
may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies.
Rapidly
changing medical technology within the life sciences could make the product candidates that we are developing obsolete.
The
medical industry is characterized by rapid and significant medical technological and therapy changes, frequent new product candidates
and product introductions and enhancements and evolving industry standards. Our future success will depend on our ability to continually
develop and then improve the product candidates that we design and to develop and introduce new product candidates that address the evolving
needs of the physicians and patients on a timely and cost-effective basis. Any new product candidates and products developed by us may
not be accepted in the intended markets. Our inability to gain market acceptance of new products could harm our future operating results.
The
market opportunities for certain of our product candidates may be small, due to the fact that the products may be limited to those patients
who are ineligible for or have failed prior treatments, and our projections regarding the size of the addressable market may be incorrect.
Cancer
therapies are sometimes characterized as first line, second line or third line, and the FDA often approves new therapies initially only
for third line use. When blood cancers are detected, they are treated with first line of therapy with the intention of curing the cancer.
This generally consists of chemotherapy, radiation, antibody drugs, tumor targeted small molecules, or a combination of these. In addition,
sometimes a bone marrow transplantation can be added to the first line therapy after the combination chemotherapy is given. If the patient’s
cancer relapses, then they are given a second line or third line therapy, which can consist of more chemotherapy, radiation, antibody
drugs, tumor targeted small molecules, or a combination of these, or bone marrow transplant. Generally, the higher the line of therapy,
the lower the chance of a cure. With third or higher line, the goal of the therapy in the treatment of lymphoma and myeloma is to control
the growth of the tumor and extend the life of the patient, as a cure is unlikely to happen. Patients are generally referred to clinical
trials in these situations.
Our
projections of both the number of people who have the cancers we are targeting, as well as the size of the patient population subset
of people with these cancers in a position to receive first, second, third and fourth line therapy and who have the potential to benefit
from treatment with our product candidates, are based on our beliefs and estimates. These estimates have been derived from a variety
of sources, including scientific literature, surveys of clinics, patient foundations, or market research and may prove to be incorrect.
Further, new studies may change the estimated incidence or prevalence of these cancers. The number of patients may turn out to be fewer
than expected. Additionally, the potentially addressable patient population for our product candidates may be limited or may not be amenable
to treatment with our product candidates. Even if we obtain significant market share for our product candidates, because the potential
target populations are small, we may never achieve significant revenues without obtaining regulatory approval for additional indications
or as part of earlier lines of therapy.
We
rely on third parties to support our clinical trials. If these third parties do not successfully carry out their contractual duties or
meet expected deadlines, we may not be able to obtain regulatory approval of or commercialize our product candidates.
We
depend and will continue to depend upon independent investigators and collaborators, such as universities, medical institutions, CROs
(“Contract Research Organizations”) and strategic partners to support our clinical trials under agreements with the Company.
We
negotiate budgets and contracts with CROs and study sites, which may result in delays to our development timelines and increased costs.
We will rely heavily on third parties over the course of our clinical trials, and we control only certain aspects of their activities.
Nevertheless, we are responsible for ensuring that each of our studies is conducted in accordance with applicable protocol, legal, regulatory
and scientific standards, and its reliance on third parties does not relieve us of our regulatory responsibilities. TCB and these third
parties are required to comply with GCPs, which are regulations and guidelines enforced by regulatory authorities for product candidates
in clinical development. Regulatory authorities enforce these GCPs through periodic inspections of trial sponsors, principal investigators
and trial sites. If we or any of these third parties fail to comply with applicable GCP regulations, the clinical data generated in our
clinical trials may be deemed unreliable and the FDA or comparable foreign regulatory authorities may require us to perform additional
clinical trials before approving our marketing applications. We cannot assure that, upon inspection, such regulatory authorities will
determine that any of our clinical trials comply with the GCP regulations. In addition, our clinical trials must be conducted with biologic
product produced under cGMPs and will require a large number of test patients. Our failure or any failure by these third parties to comply
with these regulations or to recruit a sufficient number of patients may require us to repeat clinical trials, which would delay the
regulatory approval process. Moreover, our business may be implicated if any of these third parties violates federal or state fraud and
abuse or false claims laws and regulations or healthcare privacy and security laws.
Any
third parties supporting our clinical trials are and will not be our employees and, except for remedies available to us under our agreements
with such third parties, we cannot control whether or not they devote sufficient time and resources to our ongoing preclinical, clinical
and nonclinical programs. These third parties may also have relationships with other commercial entities, including our competitors,
for whom they may also be conducting clinical studies or other drug development activities, which could affect their performance on our
behalf. If these third parties do not successfully carry out their contractual duties or obligations or meet expected deadlines, if they
need to be replaced or if the quality or accuracy of the clinical data they obtain is compromised due to the failure to adhere to our
clinical protocols or regulatory requirements or for other reasons, our clinical trials may be extended, delayed or terminated and we
may not be able to complete development of, obtain regulatory approval of or successfully commercialize our product candidates. As a
result, our financial results and the commercial prospects for our product candidates would be harmed, our costs could increase and our
ability to generate revenue could be delayed.
If
any of our relationships with trial sites, or any CRO that we may use in the future, terminates, we may not be able to enter into arrangements
with alternative trial sites or CROs or do so on commercially reasonable terms. Switching or adding third parties to conduct our clinical
trials will involve substantial cost and require extensive management time and focus. In addition, there is a natural transition period
when a new third party commences work. As a result, delays occur, which can materially impact our ability to meet its desired clinical
development timelines.
We
do not have any current sales, marketing, commercial manufacturing and distribution capabilities or arrangements, and will need to create
these as we move towards commercialization of our products.
We
do not yet have commercial sales, marketing, manufacturing and distribution capabilities or arrangements. We will need to develop all
of the foregoing or partner with organizations who have expertise in all the foregoing. We do not have any corporate experience in establishing
these commercial sized capabilities. We believe that setting up the commercialization aspects of a company such as ours, in our field,
will take a substantial amount of capital and time. Therefore, we may seek development and marketing partners and license our drug technologies
or product candidates to others in order to avoid our having to provide the marketing, manufacturing and distribution capabilities within
our organization. There can be no assurance that we will find any development and marketing partners or companies that are interested
in licensing our drug technology or any of our product candidates or products. If we are unable to establish and maintain adequate sales,
marketing, manufacturing and distribution capabilities, independently or with others, we will not be able to generate product revenue,
and may not become profitable.
We
may rely on third parties to manufacture our clinical product supplies, and we may have to rely on third parties to produce and process
our product candidates, if approved.
Although
to date, we have used our internal capabilities to manufacture clinical trial supplies, we do not yet have sufficient information to
reliably estimate the cost of commercially manufacturing and processing of our product candidates. The actual cost to manufacture and
process our product candidates could materially and adversely affect the commercial viability of our product candidates. As a result,
we may never be able to develop a commercially viable product.
Our
manufacturing plant is located in Scotland and as we develop clinical trials outside the UK and in particular into the USA we anticipate
that we will rely on a limited number of third-party manufacturers for commercial production, but this will expose us to the following
risks.
| |
● |
We
may be unable to identify manufacturers on acceptable terms or at all because the number of potential manufacturers is limited and
the regulatory authorities may have questions regarding any replacement contractor. This may require new testing and regulatory interactions.
In addition, a new manufacturer would have to be educated in, or develop substantially equivalent processes for, production of our
products. |
| |
|
|
| |
● |
Third-party
manufacturers might be unable to timely formulate and manufacture our product or produce the quantity and quality required to meet
our clinical and commercial needs, if any. |
| |
|
|
| |
● |
Future
contract manufacturers may not perform as agreed or may not remain in the contract manufacturing business for the time required to
supply our clinical trials or to successfully produce, store and distribute our products. |
| |
|
|
| |
● |
Manufacturers
are subject to strict compliance with cGMP and other government regulations and corresponding foreign standards. We do not have control
over third-party manufacturers’ compliance with these regulations and standards. |
| |
|
|
| |
● |
We
may not own, or may have to share, the intellectual property rights to any improvements made by our third-party manufacturers in
the manufacturing process for our products. |
| |
|
|
| |
● |
Third-party
manufacturers could breach or terminate their agreement(s) with us. |
Contract
manufacturers would also be subject to the same risks we face in developing our own manufacturing capabilities, as described above. Each
of these risks could delay our clinical trials, the regulatory approval, if any, of our product candidates or the commercialization of
our product candidates or result in higher costs or deprive us of potential product revenue. In addition, we will rely on third parties
to perform release tests on our product candidates prior to delivery to patients. If these tests are not appropriately done and test
data are not reliable, patients could be put at risk of serious harm.
Cell-based
therapies rely on the availability of specialty materials, which may not be available to us on acceptable terms or at all.
Our
product candidates require specialized materials, including viral vectors that deliver the targeting moiety (CAR) and other genes to
the product candidate. We currently manufacture some of our requirements through contract manufacturers, some of which are manufactured
by companies with limited resources and experience to support a commercial product, and the suppliers may not be able to deliver materials
to our specifications. In addition, those suppliers normally support blood-based hospital businesses and generally do not have the capacity
to support commercial products manufactured under cGMP by biopharmaceutical firms. The suppliers may be ill-equipped to support our needs,
especially in non-routine circumstances like an FDA inspection or medical crisis, such as widespread contamination. We also do not have
contracts with many of these suppliers, and we may not be able to contract with them on acceptable terms or at all. Accordingly, we may
experience delays in receiving key materials to support clinical or commercial manufacturing.
In
addition, some materials utilized in the manufacture of our candidates are currently available from a single supplier, or a small number
of suppliers. For example, principal suppliers for the purchase of equipment and reagents critical for the manufacture of our product
candidates include Cytiva (Global Life Sciences Solutions Operations UK Ltd), Wilson Wolf Manufacturing Corporation, Octapharma AG, Sexton
Biotechnologies, Miltenyi Biotech, Thermo Fisher Scientific and other suppliers. We cannot be sure that these suppliers will remain in
business or that they will not be purchased by one of our competitors or another company that is not interested in continuing to produce
these materials for our intended purpose. In addition, the lead time needed to establish a relationship with a new supplier can be lengthy,
and we may experience delays in meeting demand in the event that we must switch to a new supplier. The time and effort to qualify a new
supplier could result in additional costs, diversion of resources or reduced manufacturing yields, any of which would negatively impact
our operating results. Further, we may be unable to enter into agreements with a new supplier on commercially reasonable terms, which
could have a material adverse impact on our business.
Product
liability lawsuits against us could cause us to incur substantial liabilities and to limit commercialization of any products that we
may develop.
We
face an inherent risk of product liability exposure related to the testing of our product candidates in human clinical trials and will
face an even greater risk if we commercially sell any products that we may develop. If we cannot successfully defend ourselves against
claims that our product candidates or products caused injuries, we will incur substantial liabilities. Regardless of merit or eventual
outcome, liability claims may result in, for example:
| |
● |
reduced
resources of our management to pursue our business strategy; |
| |
|
|
| |
● |
decreased
demand for any product candidates or products that we may develop; |
| |
|
|
| |
● |
injury
to our reputation and significant negative media attention; |
| |
|
|
| |
● |
withdrawal
of clinical trial participants; |
| |
|
|
| |
● |
initiation
of investigations by regulators; |
| |
|
|
| |
● |
product
recalls, withdrawals or labelling, marketing or promotional restrictions; |
| |
|
|
| |
● |
significant
costs to defend the resulting litigation; |
| |
|
|
| |
● |
substantial
monetary awards paid to clinical trial participants or patients; |
| |
|
|
| |
● |
loss
of revenue; and |
| |
|
|
| |
● |
the
inability to commercialize any products that we may develop. |
We
currently hold £5.0 million in clinical study liability annual insurance cover for each clinical study, with a per patient limit
of £5.0 million, which may not be adequate to cover all liabilities that we may incur. We may need to increase our insurance coverage
as we expand our clinical trials or if we commence commercialization of our product candidates. Insurance coverage is increasingly expensive.
We may not be able to maintain insurance coverage at a reasonable cost or in an amount adequate to satisfy any liability that may arise.
Risks
Related to Governmental Regulations
Changes
and uncertainties in the tax system in the countries in which we have operations could materially adversely affect our financial condition
and results of operations, and reduce net returns to our shareholders.
We
conduct business and file income tax returns in multiple jurisdictions. Our consolidated effective income tax rate could be materially
adversely affected by several factors, including: changing tax laws, regulations and treaties, or the interpretation thereof; tax policy
initiatives and reforms under consideration (such as those related to the Organization for Economic Co-Operation and Development’s,
or OECD, Base Erosion and Profit Shifting, or BEPS, Project, the European Commission’s state aid investigations and other initiatives);
the practices of tax authorities in jurisdictions in which we operate; the resolution of issues arising from tax audits or examinations
and any related interest or penalties. Such changes may include (but are not limited to) the taxation of operating income, investment
income, dividends received or (in the specific context of withholding tax) dividends paid.
We
are unable to predict what tax reform may be proposed or enacted in the future or what effect such changes would have on our business,
but such changes, to the extent they are brought into tax legislation, regulations, policies or practices in jurisdictions in which we
operate, could increase the estimated tax liability that we have expensed to date and paid or accrued on our balance sheets, and otherwise
affect our financial position, future results of operations, cash flows in a particular period and overall or effective tax rates in
the future in countries where we have operations, reduce post-tax returns to our shareholders and increase the complexity, burden and
cost of tax compliance.
Changes
in our business strategy or operations may result in grant income being repaid to government grant awarding bodies.
We
have been awarded and received grant income from government agencies with respect to a number of research and development programs totaling
£5.6 million since incorporation through December 31, 2023. In some cases, the grant award contains commitments for the business
that extend beyond the specific program period. If the Company changes strategy or the nature of its operations, some grant awarding
bodies may view this as a breach of the original terms of the grant and all or part of the original grant award may become subject to
repayment. In the event of our having to return funds under prior grant awards, the Company may be required to repay up to an aggregate
of £5.6 million.
Tax
authorities may disagree with our positions and conclusions regarding certain tax positions, or may apply existing rules in an unforeseen
manner, resulting in unanticipated costs, taxes or non-realization of expected benefits.
A
tax authority may disagree with tax positions that we have taken, which could result in increased tax liabilities. For example, Her Majesty’s
Revenue & Customs, or HMRC, the United States Internal Revenue Service, the IRS, or another tax authority could challenge our allocation
of income by tax jurisdiction and the amounts paid between our affiliated companies pursuant to our intercompany arrangements and transfer
pricing policies, including amounts paid with respect to our intellectual property development. Similarly, a tax authority could assert
that we are subject to tax in a jurisdiction where we believe we have not established a taxable connection, often referred to as a “permanent
establishment” under international tax treaties, and such an assertion, if successful, could increase our expected tax liability
in one or more jurisdictions.
A
tax authority may take the position that material income tax liabilities, interest and penalties are payable by us, for example where
there has been a technical violation of contradictory laws and regulations that are relatively new and have not been subject to extensive
review or interpretation, in which case we expect that we might contest such assessment. High-profile companies can be particularly vulnerable
to aggressive application of unclear requirements. Many companies must negotiate their tax bills with tax inspectors who may demand higher
taxes than applicable law appears to provide. Contesting such an assessment may be lengthy and costly and if we were unsuccessful in
disputing the assessment, the implications could increase our anticipated effective tax rate, where applicable.
We
may be unable to use net operating loss and tax credit carryforwards and certain built-in losses to reduce future tax payments or benefit
from favorable UK tax legislation.
As
a UK incorporated and tax resident entity, we are subject to UK corporate taxation. Due to the nature of our business, we have generated
losses since inception and therefore have not paid any UK corporation tax. As of December 31, 2023, we had cumulative carryforward tax
trading losses of £11.1 million. Subject to any relevant utilization criteria and restrictions (including those that limit the
percentage of profits that can be reduced by carried forward losses and those that can restrict the use of carried forward losses where
there is a change of ownership of more than half of our ordinary shares (including those represented by the ADSs) and a major change
in the nature, conduct or scale of the trade), we expect these to be eligible for carry forward and utilization against future operating
profits arising in the United Kingdom. The use of loss carryforwards in relation to UK profits incurred on or after April 1, 2017 are
limited each year to £5.0 million per group plus, broadly, an incremental 50% of UK taxable profits.
As
a company that carries out extensive research and development activities, we seek to benefit from the UK research and development tax
relief programs, being the Small and Medium-sized Enterprises R&D tax relief program, or SME Program, and, to the extent that our
projects are grant funded or relate to work subcontracted to us by third parties, the Research and Development Expenditure Credit program,
or RDEC Program. Under the SME Program, we may be able to surrender the trading losses that arise from our qualifying research and development
activities for a cash rebate of up to 14.5% of such qualifying research and development expenditures. The majority of our research, clinical
trials management and manufacturing development activities are eligible for inclusion within these tax credit cash rebate claims. We
may not be able to continue to claim payable research and development tax credits in the future if we cease to qualify as a SME, based
on size criteria concerning employee headcount, turnover and gross assets.
We
may benefit in the future from the UK’s “patent box” regime, which allows certain profits attributable to revenue from
patented products (and other qualifying income) to be taxed at an effective rate of 10% by giving an additional tax deduction. We own
several patents which cover our investigational therapies, and accordingly, future upfront fees, milestone fees, product revenue and
royalties could be eligible for this deduction. When taken in combination with the enhanced relief available on our research and development
expenditures, we expect a long-term rate of corporation tax lower than the statutory to apply to us. If, however, there are unexpected
adverse changes to the UK research and development tax credit regime or the “patent box” regime, or for any reason we are
unable to qualify for such advantageous tax legislation, or we are unable to use net operating loss and tax credit carryforwards and
certain built-in losses to reduce future tax payments then our business, results of operations and financial condition may be adversely
affected. This may impact our ongoing requirement for investment and the timeframes within which additional investment is required.
Failure
to comply with United States health and data protection laws and regulations could lead to enforcement actions, including civil or criminal
penalties, private litigation, and adverse publicity and could negatively affect our operating results and business.
We
and any potential collaborators are subject to data protection laws and regulations, such as laws and regulations that address privacy
and data security. In the United States, numerous federal and state laws and regulations, including state data breach notification laws,
state health information privacy laws, and federal and state consumer protection laws, govern the collection, use, disclosure, and protection
of health-related and other personal information. In addition, we may obtain health information from third parties, including research
institutions from which we obtain clinical trial data, which are subject to privacy and security requirements under HIPAA, as amended
by HITECH. To the extent that we act as a business associate to a healthcare provider engaging in electronic transactions, we may also
be subject to the privacy and security provisions of HIPAA, as amended by HITECH, which restricts the use and disclosure of patient-identifiable
health information, mandates the adoption of standards relating to the privacy and security of patient-identifiable health information,
and requires the reporting of certain security breaches to healthcare provider customers with respect to such information. Additionally,
many states have enacted similar laws that may impose more stringent requirements on entities like ours. Depending on the facts and circumstances,
we could be subject to significant civil, criminal, and administrative penalties if we obtain, use, or disclose individually identifiable
health information maintained by a HIPAA-covered entity in a manner that is not authorized or permitted by HIPAA.
Additionally,
in June 2018, the State of California enacted the California Consumer Privacy Act of 2018, or CCPA, which came into effect on January
1, 2020 and provides new data privacy rights for consumers (as that term is broadly defined) and new operational requirements for companies,
which may increase our compliance costs and potential liability. The CCPA gives California residents expanded rights to access and delete
their personal information, opt out of certain personal information sharing, and receive detailed information about how their personal
information is used. The CCPA provides for civil penalties for violations, as well as a private right of action for data breaches that
is expected to increase data breach litigation. While there is currently an exception for protected health information that is subject
to HIPAA and clinical trial regulations, as currently written, the CCPA may impact certain of our business activities. The CCPA could
mark the beginning of a trend toward more stringent state privacy legislation in the United States, which could increase our potential
liability and adversely affect our business.
Compliance
with U.S. and foreign privacy and data protection laws and regulations could require us to take on more onerous obligations in our contracts,
restrict our ability to collect, use and disclose data, or in some cases, impact our ability to operate in certain jurisdictions. Failure
to comply with these laws and regulations could result in government enforcement actions (which could include civil, criminal and administrative
penalties), private litigation, and/or adverse publicity and could negatively affect our operating results and business. Moreover, clinical
trial subjects, employees and other individuals about whom we or our potential collaborators obtain personal information, as well as
the providers who share this information with us, may limit our ability to collect, use and disclose the information. Claims that we
have violated individuals’ privacy rights, failed to comply with data protection laws, or breached our contractual obligations,
even if we are not found liable, could be expensive and time-consuming to defend and could result in adverse publicity that could harm
our business.
European
data collection is governed by restrictive privacy and security regulations governing the use, processing and cross-border transfer of
personal information.
The
collection, use, storage, disclosure, transfer, or other processing of personal data (including health data processed in the context
of clinical trials) (i) regarding individuals in the EU, and/or (ii) carried out in the context of the activities of our establishment
in any EU member state, is subject to the EU General Data Protection Regulation, or GDPR, which became effective on May 25, 2018, as
well as other national data protection legislation in force in relevant member states (including the Data Protection Act 2018 in the
UK).
The
GDPR is wide-ranging in scope and imposes numerous additional requirements on companies that process personal data, including imposing
special requirements in respect of the processing of health and other sensitive data, requiring that consent of individuals to whom the
personal data relates is obtained in certain circumstances, requiring additional disclosures to individuals regarding data processing
activities, requiring that safeguards are implemented to protect the security and confidentiality of personal data, creating mandatory
data breach notification requirements in certain circumstances, and requiring that certain measures (including contractual requirements)
are put in place when engaging third-party processors. The GDPR also imposes strict rules on the transfer of personal data to countries
outside the EEA, including the United States, and permits data protection authorities to impose large penalties for violations of the
GDPR, including potential fines of up to €20 million or 4% of annual global revenue, whichever is greater. The GDPR provides individuals
with various rights in respect of their personal data, including rights of access, erasure, portability, rectification, restriction and
objection. The GDPR also confers a private right of action on data subjects and consumer associations to lodge complaints with supervisory
authorities, seek judicial remedies, and obtain compensation for damages resulting from violations of the GDPR.
The
GDPR may increase our responsibility and liability in relation to personal data that we process where such processing is subject to the
GDPR. While we have taken steps to comply with the GDPR, and implementing legislation in applicable EU member states, including by seeking
to establish appropriate lawful bases for the various processing activities we carry out as a controller or joint controller, reviewing
our security procedures and those of our vendors and collaborators, and entering into data processing agreements with relevant vendors
and collaborators, we cannot be certain that our efforts to achieve and remain in compliance have been, and/or will continue to be, fully
successful.
Following
the UK’s withdrawal from the EU on January 31, 2020 and following the end the of the transitional arrangements on December 31,
2020, it is likely that the data protection obligations of the GDPR will continue to apply to UK-based organizations’ processing
of personal data in substantially unvaried form, for at least the short term thereafter.
Risks
Related to Our Business Operations, Managing Growth and Employee Matters
We
may have difficulty managing growth in our business.
Because
of our small size, growth in accordance with our business plan, if achieved, will place a significant strain on our financial, technical,
operational and management resources. As we expand our activities, there will be additional demands on these resources. The failure to
continue to upgrade our technical, administrative, operating and financial control systems or the occurrence of unexpected expansion
difficulties, including issues relating to our research and development activities and retention of experienced scientists, managers
and engineers, could have a material adverse effect on our business, financial condition and results of operations and our ability to
timely execute our business plan. If we are unable to implement these actions in a timely manner, our results may be adversely affected.
We
depend upon our key personnel and our ability to attract and retain employees
We
are heavily dependent on the ongoing employment and involvement of certain key employees in particular our principal executive officers
(i) Bryan Kobel, our Chief Executive Officer and Martin Thorp, our Chief Financial Officer.
We
anticipate, overtime, a requirement to expand our current personnel, who will be based in the UK, the EU and the USA, potentially very
rapidly in order to achieve our planned business activities and aims to further engage in clinical trials. Such expansion is dependent
on our ability to recruit experienced and suitably trained employees or consultants, and to retain such employees on a long-term basis.
Our ability to take our existing pipeline of GD-T cell therapeutics and to meet the demands of our clinical programs may be compromised
or delayed if we are unable to recruit sufficient personnel on a timely basis.
The
loss of key managers and senior scientists could delay our research and development activities. In addition, our ability to compete in
the highly competitive pharmaceutical industry depends upon our ability to attract and retain highly qualified management, scientific
and medical personnel. Many other companies and academic institutions that we compete against for qualified personnel have greater financial
and other resources, different risk profiles and a longer history in the industry than we do. Therefore, we might not be able to attract
or retain these key persons on conditions that are economically acceptable. Moreover, some qualified prospective employees may choose
not to work for us due to negative perceptions regarding the therapeutic use of psilocybin or other objections to the therapeutic use
of a controlled substance. Furthermore, we will need to recruit new managers and qualified scientific personnel to develop our business
if we expand into fields that will require additional skills. Our inability to attract and retain these key people could prevent us from
achieving our objectives and implementing our business strategy, which could have a material adverse effect on our business and prospects.
In
addition, certain key academic and scientific personnel play a pivotal role in our collaborative partners’ research and development
activities. If any of those key academic and scientific personnel who work on development of our research programs, our investigational
GD-T cell therapy and any future therapeutic candidates leave our collaborative partners, the development of our research programs, our
investigational GD-T cell therapy and any future therapeutic candidates may be delayed or otherwise adversely affected.
We
will need to grow the size and capabilities of our organization, and we may experience difficulties in managing this growth.
As
of March 7, 2024, we have 41 full-time equivalent employees. As our development and commercialization plans and strategies develop, and
as we transition into operating as a public company, we will have to add a significant number of additional managerial, operational,
financial, and other personnel. Future growth will impose significant added responsibilities on members of management, including:
| |
● |
identifying,
recruiting, integrating, maintaining, and motivating additional employees; |
| |
|
|
| |
● |
managing
our internal development efforts effectively, including the clinical and regulatory review process for our GD-T therapeutic candidates,
while complying with our contractual obligations to contractors and other third parties; and |
| |
|
|
| |
● |
improving
our operational, financial and management controls, reporting systems, and procedures. |
Our
future financial performance and our ability to commercialize our GD-T therapeutic candidates will depend, in part, on our ability to
effectively manage any future growth, and our management may also have to divert a disproportionate amount of its attention away from
day-to-day activities in order to devote a substantial amount of time to managing these growth activities.
If
we fail to maintain proper and effective internal controls, our ability to produce accurate financial statements on a timely basis could
be impaired, which would adversely affect our business and our stock price.
Ensuring
that we have adequate internal financial and accounting controls and procedures in place to produce accurate financial statements on
a timely basis is a costly and time-consuming effort that needs to be re-evaluated frequently. We may discover material weaknesses in
our internal financial and accounting controls and procedures that need improvement from time to time.
Management
is responsible for establishing and maintaining adequate internal control over financial reporting to provide reasonable assurance regarding
the reliability of our financial reporting and the preparation of financial statements for external purposes. Management does not expect
that our internal control over financial reporting will prevent or detect all errors and all fraud. A control system, no matter how well
designed and operated, can provide only reasonable, not absolute, assurance that the control system’s objectives will be met. Because
of the inherent limitations in all control systems, no evaluation of controls can provide absolute assurance that misstatements due to
error or fraud will not occur or that all control issues and instances of fraud, if any, within our company will have been detected.
Pursuant
to Section 404(a) of the Sarbanes-Oxley Act of 2002, or Section 404, we are required to furnish a report by our senior management on
our internal control over financial reporting. However, while we remain an EGC we are not required
to include an attestation report on internal control over financial reporting issued by our independent registered public accounting
firm (Section 404(b)). To prepare for eventual compliance with Section 404(b), once we no longer qualify as an EGC, we are engaged in
a process to document and evaluate our internal control over financial reporting, which is both costly and challenging. In this regard,
we will need to continue to dedicate internal resources, potentially engage outside consultants, adopt a detailed work plan to assess
and document the adequacy of internal control over financial reporting, continue steps to improve control processes as appropriate, validate
through testing that controls are functioning as documented, and implement a continuous reporting and improvement process for internal
control over financial reporting. Despite our efforts, there is a risk that we will not be able to conclude, within the prescribed timeframe
or at all, that our internal control over financial reporting is effective as required by Section 404. If we identify one or more material
weaknesses, it could result in an adverse reaction in the financial markets due to a loss of confidence in the reliability of our financial
statements. In addition, if we are unable to produce accurate financial statements on a timely basis, investors could lose confidence
in the reliability of our financial statements, which could cause the market price of either of our ADSs or Warrants, or both, to decline
and make it more difficult for us to finance our operations and growth.
The
Company notes that the auditors identified that the Company experienced difficulty in the accounting for complex financial
instruments and leases, and the Company lacked adequate internal control over the account and assessment of complex financial
instruments following control deficiencies which they believed constituted a material weakness in the Company’s internal
control over financial reporting as of December 31, 2023. The Company recognizes this error as a material weakness and has
established controls to support assessment and review of accounting for complex financial instruments and leases.
A
pandemic, epidemic, or outbreak of an infectious disease, such as the COVID-19 pandemic, may materially and adversely affect our business,
including our preclinical studies, clinical trials, third parties on whom we rely, our supply chain, our ability to raise capital, our
ability to conduct regular business and our financial results.
We
are subject to risks related to public health crises such as the COVID-19 pandemic. The COVID-19 pandemic continues throughout the world.
The pandemic and policies and regulations implemented by governments in response to the pandemic, often directing businesses and governmental
agencies to cease non-essential operations at physical locations, prohibiting certain nonessential gatherings and ceasing non-essential
travel have also had a significant impact, both direct and indirect, on businesses and commerce, as worker shortages have occurred, supply
chains have been disrupted, facilities and production have been suspended, and demand for certain goods and services, such as medical
service and supplies, has spiked, while demand for other goods and services, such as travel, has fallen. The full extent to which COVID-19
will ultimately impact our business, preclinical trials and financial results will depend on future developments, which are highly uncertain
and cannot be accurately predicted, including new information which may emerge concerning the severity of COVID-19 and the actions to
contain COVID-19 or treat its impact, among others. Global health concerns, such as the COVID-19 pandemic, could also result in social,
economic, and labor instability in the countries in which we or the third parties with whom we engage operate.
In
response to the COVID-19 pandemic, we have taken temporary precautionary measures intended to help minimize the risk of the virus to
our employees, including flexibility for employees to work remotely where appropriate, suspending all non-essential travel worldwide
for our employees and discouraging employee attendance at industry events and in-person work-related meetings, all of which could negatively
affect our business. The extent of the impact of the COVID-19 pandemic on our preclinical studies or clinical trial operations, our supply
chain and manufacturing and our office-based business operations, will depend on future developments, which are highly uncertain and
cannot be predicted with confidence, such as the duration of the pandemic, the severity of the COVID-19 pandemic, or the effectiveness
of actions to contain and treat coronavirus.
While
we are working closely with our third-party manufacturers, distributors and other partners to manage our supply chain activities and
mitigate potential disruptions to current and any future therapeutic candidates as a result of the COVID-19 pandemic, if the COVID-19
pandemic continues and persists for an extended period of time, we expect there will be significant and material disruptions to our supply
chain and operations, and associated delays in the manufacturing and supply of current and any future therapeutic candidates. Any such
supply disruptions would adversely impact our ability to generate sales of and revenue from our approved products and our business, financial
condition, results of operations and growth prospects could be materially adversely affected.
The
COVID-19 pandemic may also affect employees of third-party CROs located in affected geographies that we rely upon to carry out our clinical
trials. As COVID-19 continues to be present and spread around the globe, we may experience additional disruptions that could severely
impact our business and clinical trials, including:
| |
● |
delays
or difficulties in enrolling patients in our clinical trials; |
| |
|
|
| |
● |
delays
or difficulties in clinical site initiation, including difficulties in recruiting clinical site investigators and clinical site staff; |
| |
|
|
| |
● |
diversion
of healthcare resources away from the conduct of clinical trials, including the diversion of sites or facilities serving as our clinical
trial sites and staff supporting the conduct of our clinical trials, including our trained therapists, or absenteeism due to the
COVID-19 pandemic that reduces site resources; |
| |
|
|
| |
● |
interruption
of key clinical trial activities, such as clinical trial site monitoring, due to limitations on travel imposed or recommended by
federal, state or national governments, employers and others or interruption of clinical trial subject visits and study procedures,
the occurrence of which could affect the integrity of clinical trial data; |
| |
|
|
| |
● |
risk
that participants enrolled in our clinical trials will acquire COVID-19 while the clinical trial is ongoing, which could impact the
results of the clinical trial, including by increasing the number of observed adverse events or patient withdrawals from our trials; |
| |
|
|
| |
● |
limitations
in employee resources that would otherwise be focused on conducting our clinical trials, including because of sickness of employees
or their families or the desire of employees to avoid contact with large groups of people; |
| |
|
|
| |
● |
delays
in receiving authorizations from regulatory authorities to initiate our planned clinical trials; |
| |
|
|
| |
● |
delays
in clinical sites receiving the supplies and materials needed to conduct our clinical trials; |
| |
|
|
| |
● |
interruption
in global shipping that may affect the transport of clinical trial materials, such as the cell therapy used in our clinical trials; |
| |
|
|
| |
● |
changes
in local regulations as part of a response to the COVID-19 pandemic which may require us to change the ways in which our clinical
trials are conducted, which may result in unexpected costs, or the discontinuation of the clinical trials altogether; |
| |
|
|
| |
● |
interruptions
or delays in preclinical studies due to restricted or limited operations at research and development laboratory facilities; |
| |
|
|
| |
● |
delays
in necessary interactions with local regulators, ethics committees and other important agencies and contractors due to limitations
in employee resources or forced furlough of government employees; and |
| |
|
|
| |
● |
refusal
of the FDA, the EMA, the MHRA or the other regulatory bodies to accept data from clinical trials in affected geographies outside
the United States or the EU or other relevant local geography. |
Any
negative impact the COVID-19 pandemic has on patient enrolment or treatment or the development of our investigational cell therapies
and any future therapeutic candidates could cause costly delays to clinical trial activities, which could adversely affect our ability
to obtain regulatory approval for and to commercialize our investigational cell therapies and any future therapeutic candidates, if approved,
increase our operating expenses, and have a material adverse effect on our financial results.
The
COVID-19 pandemic has also caused significant volatility in public equity markets and disruptions to the United States and global economies.
This increased volatility and economic dislocation may make it more difficult for us to raise capital on favorable terms, or at all.
We cannot currently predict the scope and severity of any potential business shutdowns or disruptions. If we or any of the third parties
with whom we engage, however, were to experience shutdowns or other business disruptions, our ability to conduct our business in the
manner and on the timelines presently planned could be materially and negatively affected, which could have a material adverse impact
on our business and our results of operations and financial conditions. To the extent the COVID-19 pandemic adversely affects our business
and financial results, it may also heighten many of the other risks described in this “Risk Factors” section, such as those
relating to the timing and completion of our clinical trials and our ability to obtain future financing.
Our
current operations are headquartered in one location, and we or the third parties upon whom we depend may be adversely affected by unplanned
natural disasters, as well as occurrences of civil unrest, and our business continuity and disaster recovery plans may not adequately
protect us from a serious disaster, including earthquakes, outbreak of disease or other natural disasters.
Our
current business operations are headquartered and principally located in our offices and laboratories in Glasgow, UK. Any unplanned event,
such as flood, fire, explosion, earthquake, extreme weather condition, medical epidemics, power shortage, telecommunication failure or
other natural or man-made accidents or incidents, including events of civil unrest that result in us being unable to fully utilize our
facilities, or the manufacturing facilities of our third-party contract manufacturers, may have a material and adverse effect on our
ability to operate our business, particularly on a daily basis, and have significant negative consequences on our financial and operating
conditions. Loss of access to these facilities may result in increased costs, delays in the development of our investigational GD-T cell
therapy or any future therapeutic candidates or interruption of our business operations.
The
disaster recovery and business continuity plans we have in place may prove inadequate in the event of a serious disaster or similar event.
We may incur substantial expenses as a result of the limited nature of our disaster recovery and business continuity plans, which, could
have a material adverse effect on our business. As part of our risk management policy, we maintain insurance coverage at levels that
we believe are appropriate for our business, but in the final result may not be sufficient to satisfy any damages and losses.
The
increasing use of social media platforms presents new risks and challenges.
Social
media is increasingly being used to communicate about our clinical development programs and the diseases our investigational GD-T cell
therapy or any future therapeutic candidates are being developed to treat, and we may use appropriate social media in connection with
our commercialization efforts of our investigational GD-T cell therapy following approval of our GD-T cell therapy or any future therapeutic
candidates, if any. Social media practices in the biopharmaceutical industry continue to evolve, and regulations and regulatory guidance
relating to such use are evolving and not always clear. This evolution creates uncertainty and risk of noncompliance with regulations
applicable to our business, resulting in potential regulatory actions against us, along with the potential for litigation related to
certain prohibited activities. For example, patients may use social media channels to comment on their experience in an ongoing clinical
trial or to report an alleged adverse event. When such disclosures occur, there is a risk that trial enrolment may be adversely impacted,
we fail to monitor and comply with applicable adverse event reporting obligations, or that we may not be able to defend our business
or the public’s legitimate interests in the face of the political and market pressures generated by social media due to restrictions
on what we may say about our investigational GD-T cell therapy or any future therapeutic candidates. There is also a risk of inappropriate
disclosure of sensitive information or negative or inaccurate posts or comments about us on any social networking website. If any of
these events were to occur or we otherwise fail to comply with applicable regulations, we could incur liability, face regulatory actions
or incur other harm to our business.
Risks
Related to Intellectual Property
If
we or our licensors are unable to protect our/their intellectual property, then our financial condition, results of operations and the
value of our drug technology and product candidates could be adversely affected.
Patents
and other proprietary rights are essential to our business, and our ability to compete effectively with other companies is dependent
upon the proprietary nature of our drug technologies and product candidates. We also rely upon trade secrets, know-how, continuing innovations
and licensing opportunities to develop, maintain and strengthen our competitive position. We seek to protect these, in part, through
confidentiality agreements with employees, consultants and other parties. Our success will depend in part on the ability of TCB and our
licensors to obtain, to maintain (including making periodic filings and payments) and to enforce patent protection for the licensed intellectual
property, in particular, those patents to which we have secured rights. We, and our licensors, may not successfully prosecute or continue
to prosecute the patent applications which we have licensed. Even if patents are issued in respect of these patent applications, TCB
or our licensors may fail to maintain these patents, may determine not to pursue litigation against entities that are infringing upon
these patents, or may pursue such enforcement less aggressively than we ordinarily would for our own patents. Without adequate protection
for the intellectual property that we own or license, other companies might be able to offer substantially identical products for sale,
which could unfavorably affect our competitive business position and harm our business prospects. Even if issued, patents may be challenged,
invalidated, or circumvented, which could limit our ability to stop competitors from marketing similar products or limit the length of
term of patent protection that we may have for our products.
Litigation
or third-party claims of intellectual property infringement or challenges to the validity of our patents would require us to use resources
to protect our rights and may prevent or delay our development, regulatory approval or commercialization of our product candidates.
If
we are the target of claims by third parties asserting that our product candidates and products or intellectual property infringe upon
the rights of others we may be forced to incur substantial expenses or divert substantial employee resources from our current business
endeavors. If successful, those claims could result in our having to pay substantial damages or could prevent us from developing one
or more product candidates or commercializing a product. Further, if a patent infringement suit were brought against us or our collaborators,
we or they could be forced to stop or delay research, development, manufacturing or sales of the product candidate or product that is
the subject of the suit.
If
we or our collaborators experience patent infringement claims, or if we elect to avoid potential claims others may be able to assert,
we or our collaborators may choose to seek, or be required to seek, a license from the third-party and would most likely be required
to pay license fees or royalties or both. These licenses may not be available on acceptable terms, or at all. Even if we or our collaborators
were able to obtain a license, the rights may be nonexclusive, which would give our competitors access to the same intellectual property.
Ultimately, we could be prevented from commercializing a product, or be forced to cease some aspect of our business operations if, as
a result of actual or threatened patent infringement claims, we or our collaborators are unable to enter into licenses on acceptable
terms. This could harm our business significantly. The cost to us of any litigation or other proceeding, regardless of its merit, even
if resolved in our favor, could be substantial. Some of our competitors may be able to bear the costs of such litigation or proceedings
more effectively than we can because of their having greater financial resources. Uncertainties resulting from the initiation and continuation
of patent litigation or other proceedings could have a material adverse effect on our ability to compete in the marketplace. Intellectual
property litigation and other proceedings may, regardless of their merit, also absorb significant management time and employee resources.
If
we are unable to obtain and maintain patent protection for our GD-T cell technologies and product candidates, or if the scope of the
patent protection obtained is not sufficiently broad, our competitors could develop and commercialize technology and biologics similar
or identical to ours, and our ability to successfully commercialize our technology and product candidates may be impaired.
Our
success depends on our ability to obtain and maintain patent protection in the United States, the European Union, Japan and other countries
with respect to our product candidates. We seek to protect our proprietary position by filing patent applications related to our technology
and product candidates in the major pharmaceutical markets, including the United States, major countries in Europe and Japan. If we do
not adequately protect our intellectual property, competitors may be able to use our technologies and erode or negate any competitive
advantage that we may have, which could harm our business and ability to achieve profitability.
To
protect our proprietary positions, we file patent applications related to our novel technologies and product candidates that are important
to our business. The patent application and prosecution process is expensive and time-consuming. We may not be able to file and prosecute
all necessary or desirable patent applications at a reasonable cost or in a timely manner. We may also fail to identify patentable aspects
of our research and development before it is too late to obtain patent protection. It is possible that defects of form in the preparation
or filing of our patents or patent applications may exist, or may arise in the future, such as with respect to proper priority claims,
inventorship, claim scope or patent term adjustments. If any current or future licensors or licensees are not fully cooperative or disagree
with us as to the prosecution, maintenance or enforcement of any patent rights, such patent rights could be compromised and we might
not be able to prevent third parties from making, using and selling competing products. If there are material defects in the form or
preparation of our patents or patent applications, such patents or applications may be invalid and unenforceable. Moreover, our competitors
may independently develop equivalent knowledge, methods and know-how. Any of these outcomes could impair our ability to prevent competition
from third parties.
Patent
applications are generally in the form of composition of matter or method patents. A composition of matter (COM) patent protects an actual
drug molecule or engineered cell or other therapeutic agent and will be infringed by a third party making any use of the protected composition.
COM patents provide de-facto protection for any and all uses of the protected composition and are generally held to be the strongest
and most valuable form of patent protection. Method patents protect, for example, a method of manufacturing a product or a method of
using it. They can be valuable but typically are more limited in scope than COM patents, particularly method of use patents which only
protect a particular application of a product. Where our patent applications are limited in their scope, such as a patent protecting
the method of use, such patent rights could be compromised and we might not be able to prevent third parties from making, using and selling
competing products.
Prosecution
of our owned and in-licensed patent portfolio is at a relatively early stage in some instances. This status of these patent rights is
discussed above.. Neither priority applications nor PCT applications can themselves give rise to issued patents. Rather, protection for
the inventions disclosed in these applications must be further pursued by applicable deadlines via applications that are subject to examination.
As applicable deadlines for the priority and PCT applications become due, we will need to decide whether to and in which countries or
jurisdictions to pursue patent protection for the various inventions claimed in these applications, and we will only have the opportunity
to pursue and obtain patents in those jurisdictions where we pursue protection.
It
is also possible that we will fail to identify patentable aspects of our research and development output before it is too late to obtain
patent protection. The patent applications that we own or in-license may fail to result in issued patents with claims that cover our
current and future product candidates in the countries in which we pursue patent protection. Our patent applications cannot be enforced
against third parties practicing the technology claimed in such applications unless, and until, a patent issues from such applications,
and then only to the extent the issued claims cover the technology.
If
the patent applications we hold or have in-licensed with respect to our development programs and product candidates fail to issue, if
their breadth or strength of protection is threatened, or if they fail to provide meaningful exclusivity for our current and future product
candidates, it could threaten our ability to commercialize our product candidates. Any such outcome could have a negative effect on our
business.
Patent
and other intellectual property rights may not be upheld, in which case we will suffer a loss of our intellectual property position and
the value of our assets.
The
patent position of biotechnology and pharmaceutical companies generally is highly uncertain. Changes in either the patent laws or interpretation
of the patent laws of the various jurisdictions in which we pursue patents may diminish the value of our patents or narrow the scope
of our patent protection. In addition, the protections offered by laws of different countries vary. No consistent policy regarding the
breadth of claims allowed in biotechnology and pharmaceutical patents has emerged to date in many jurisdictions. In addition, the determination
of patent rights with respect to pharmaceutical technologies, such as our cell technologies, commonly involves complex legal and factual
questions, which has in recent years been the subject of much litigation. As a result, the issuance, scope, validity, enforceability
and commercial value of our patent rights, whether owned or in-licensed, are highly uncertain. Furthermore, the scope, strength and enforceability
of our patent rights or the nature of proceedings that may be brought by or against us related to our patent rights may change as the
related patent and intellectual property laws change over time. Additionally, in the United States, one of the jurisdictions in which
we purse patent protection, the U.S. Supreme Court has ruled on several patent cases in recent years either narrowing the scope of patent
protection available in certain circumstances or weakening the rights of patent owners in certain situations. In addition to increasing
uncertainty with regard to our ability to obtain patents in the future, this combination of events has created uncertainty with respect
to the value of patents, once obtained. Depending on decisions by the U.S. Congress, the U.S. federal courts, and the USPTO, the laws
and regulations governing patents could change in unpredictable ways that could weaken our ability to obtain patents or to enforce any
patents that we might obtain in the future.
We
may be unaware of the rights of others which may ultimately be used to limit our intellectual property rights.
We
may not be aware of all third-party intellectual property rights potentially relating to our current and future product candidates. Publications
of discoveries in the scientific literature often lag behind the actual discoveries, and patent applications in many jurisdictions typically
are not published until 18 months or more after filing, or in some cases not at all. Therefore, we cannot be certain that we or our licensors
were the first to make the inventions claimed in our patents or pending patent applications, or that we or our licensors were the first
to file for patent protection of such inventions. Similarly, should we own or in-license any patents or patent applications in the future,
we may not be certain that we or the applicable licensor were the first to file for patent protection for the inventions claimed in such
patents or patent applications. As a result, the issuance, scope, validity and commercial value of our patent rights cannot be predicted
with any certainty. Moreover, in the United States, we may be subject to a third-party pre-issuance submission of prior art to the U.S.
Patent and Trademark Office, or USPTO, or become involved in opposition, derivation, re-examination, inter partes review
or interference proceedings, in the United States or elsewhere, challenging our patent rights or the patent rights of others. An adverse
determination in any such submission, proceeding or litigation could reduce the scope of, or invalidate, our patent rights, allow third
parties to commercialize our technology or product candidates and compete directly with us, without payment to us, or result in our inability
to manufacture or commercialize products without infringing third-party patent rights, which could significantly harm our business and
results of operations.
We
may be subject to claims by third parties asserting that we or our employees have misappropriated their intellectual property, or claiming
ownership of what we regard as our own intellectual property.
Many
of our employees were previously employed at universities or other biotechnology or pharmaceutical companies. Although we try to ensure
that our employees do not use the proprietary information or know-how of third parties in their work for us, we may be subject to claims
that these employees or we have inadvertently or otherwise used intellectual property, including trade secrets or other proprietary information,
of any such employee’s former employer. We may also in the future be subject to claims that we have caused an employee to breach
the terms of his or her non-competition or non-solicitation agreement. Litigation may be necessary to defend against these potential
claims.
In
addition, while it is our policy to require our employees and contractors who may be involved in the development of intellectual property
to execute agreements assigning such intellectual property to us, such employees and contractors may breach the agreement and claim the
developed intellectual property as their own.
If
we fail in defending any the claims we have made, in addition to paying monetary damages, we may lose valuable intellectual property
rights or personnel. A court could prohibit us from using technologies or features that are essential to our products if such technologies
or features are found to incorporate or be derived from the trade secrets or other proprietary information of the former employers. Even
if we are successful in prosecuting or defending against such claims, litigation could result in substantial costs and could be a distraction
to management. In addition, any litigation or threat thereof may adversely affect our ability to hire employees or contract with independent
service providers. Moreover, a loss of key personnel or their work product could hamper or prevent our ability to commercialize our products.
Technologies
and other proprietary rights for which we seek patent protection may not be obtained, which would potentially limit the value of our
intellectual property.
Our
pending and future patent applications, whether owned or in-licensed, may not result in patents being issued that protect our technology
or product candidates, in whole or in part, or which effectively prevent others from commercializing competitive technologies and products.
Even if our patent applications issue as patents, they may not issue in a form that will provide us with any meaningful protection against
competing products or processes sufficient to achieve our business objectives, prevent competitors from competing with us or otherwise
provide us with any competitive advantage. Our competitors may be able to circumvent our owned or licensed patents, should they issue,
by developing similar or alternative technologies or products in a non-infringing manner. Our competitors may seek approval to market
their own products similar to or otherwise competitive with our products. In these circumstances, we may need to defend and/or assert
our patents, including by filing lawsuits alleging patent infringement. In any of these types of proceedings, a court or other agency
with jurisdiction may find our patents invalid and/or unenforceable.
The
issuance of a patent is not conclusive as to its inventorship, scope, validity or enforceability, and our owned and licensed patents
may be challenged in the courts or patent offices in the jurisdictions in which we have filed for patent protection. Such challenges
may result in loss of exclusivity or freedom to operate or in patent claims being narrowed, invalidated or held unenforceable, in whole
or in part, which could limit our ability to stop others from using or commercializing similar or identical technology and products,
or limit the duration of the patent protection of our technology and products. In addition, given the amount of time required for the
development, testing and regulatory review of new product candidates, patents protecting such candidates might expire before or shortly
after such candidates are commercialized.
We
may be subject to claims challenging the inventorship or ownership of our owned or in-licensed patent rights and other intellectual property.
We
generally enter into confidentiality and intellectual property assignment agreements with our employees, consultants, outside scientific
collaborators, sponsored researchers and other advisors. However, these agreements may not be honored and may not effectively assign
intellectual property rights to us. For example, disputes may arise from conflicting obligations of consultants or others who are involved
in developing our technology and product candidates. Litigation may be necessary to defend against these and other claims challenging
inventorship or ownership. The owners of intellectual property in-licensed to us could also face such claims. If we or our licensors
fail in defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights, such as
exclusive ownership of, or right to use, valuable intellectual property. Such an outcome could have a material adverse effect on our
business. Even if we or our licensors are successful in defending against such claims, litigation could result in substantial costs and
be a distraction to management and other employees.
Third
parties may initiate legal proceedings alleging that we are infringing their intellectual property rights, the outcome of which would
be uncertain and could significantly harm our business.
We
believe that we have proprietary and modular T cell programming technology that does not infringe the intellectual property and other
proprietary rights of third parties. Numerous third-party U.S. and non-U.S. issued patents exist in the area of biotechnology, including
in the area of programmed T cell therapies. Some are patents held by our competitors. If any third-party patents cover our product candidates
or technologies, we may not be free to manufacture or commercialize our product candidates as planned.
There
is a substantial amount of intellectual property litigation in the biotechnology and pharmaceutical industries, and we may become party
to, or threatened with, litigation or other adversarial proceedings regarding intellectual property rights with respect to our technology
or product candidates, including interference proceedings before the relevant patent office. Intellectual property disputes arise in
a number of areas including with respect to patents, use of other proprietary rights and the contractual terms of license arrangements.
Third parties may assert claims against us based on existing or future intellectual property rights and claims may also come from competitors
against whom our own patent portfolio may have no deterrent effect. The outcome of intellectual property litigation is subject to uncertainties
that cannot be adequately quantified in advance. Other parties may allege that our product candidates or the use of our technologies
infringes patent claims or other intellectual property rights held by them or that we are employing their proprietary technology without
authorization. As we continue to develop and, if approved, commercialize our current and future product candidates, competitors may claim
that our technology infringes their intellectual property rights as part of business strategies designed to impede our successful commercialization.
There are and may in the future be additional third-party patents or patent applications with claims to, for example, materials, compositions,
formulations, methods of manufacture or methods for treatment related to the use or manufacture of any one or more of our product candidates.
If
we are found to infringe a third party’s intellectual property rights, we could be forced, including by court order, to cease developing,
manufacturing or commercializing the infringing product candidate or product. Alternatively, we may be required or may choose to obtain
a license from such third party in order to use the infringing technology and continue developing, manufacturing or marketing the infringing
product candidate. However, we may not be able to obtain any required license on commercially reasonable terms or at all. Even if we
were able to obtain a license, it could be non-exclusive, thereby giving our competitors access to the same technologies licensed to
us. In addition, we could be found liable for monetary damages, including treble damages and attorneys’ fees if we are found to
have willfully infringed a patent. A finding of infringement could prevent us from commercializing our product candidates or force us
to cease some of our business operations. Claims that we have misappropriated the confidential information or trade secrets of third
parties could have a similar negative effect on our business. Even if successful, the defense of any claim of infringement or misappropriation
is time-consuming, expensive and diverts the attention of our management from our ongoing business operations.
We
may need to license intellectual property from third parties, and such licenses may not be available or may not be available on commercially
reasonable terms.
A
third party may hold intellectual property rights, including patent rights, which are important or necessary to the development or manufacture
of our product candidates. It may be necessary for us to use the patented or proprietary technology of third parties to commercialize
our product candidates, in which case we would be required to obtain a license from these third parties. Such a license may not be available
on commercially reasonable terms, or at all, and we could be forced to accept unfavorable contractual terms. If we are unable to obtain
such licenses on commercially reasonable terms, our business could be harmed.
We
may become involved in lawsuits to protect or enforce our intellectual property, which could be expensive, time-consuming and unsuccessful.
Competitors
may infringe our patents, if issued, trademarks, copyrights or other intellectual property. To counter infringement or unauthorized use,
we may be required to file infringement claims, which can be expensive and time-consuming and divert the time and attention of our management
and scientific personnel. Any claims we assert against perceived infringers could provoke these parties to assert counterclaims against
us alleging that we infringe their patents, trademarks, copyrights or other intellectual property. In addition, in a patent infringement
proceeding, there is a risk that a court will decide that a patent of ours is invalid or unenforceable, in whole or in part, and that
we do not have the right to stop the other party from using the invention at issue. There is also a risk that, even if the validity of
such patents is upheld, the court will construe the patent’s claims narrowly or decide that we do not have the right to stop the
other party from using the invention at issue on the grounds that our patents do not cover the invention. An adverse outcome in a litigation
or proceeding involving our patents could limit our ability to assert our patents against those parties or other competitors, and may
curtail or preclude our ability to exclude third parties from making and selling similar or competitive products. Similarly, if we assert
trademark infringement claims, a court may determine that the marks we have asserted are invalid or unenforceable, or that the party
against whom we have asserted trademark infringement has superior rights to the marks in question. In this case, we could ultimately
be forced to cease use of such trademarks.
In
any infringement litigation, any award of monetary damages we receive may not be commercially valuable. Furthermore, because of the substantial
amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information
could be compromised by disclosure during litigation. Moreover, there can be no assurance that we will have sufficient financial or other
resources to file and pursue such infringement claims, which typically last for years before they are concluded. Some of our competitors
may be able to sustain the costs of such litigation or proceedings more effectively than we can because of their greater financial resources
and more mature and developed intellectual property portfolios. Even if we ultimately prevail in such claims, the monetary cost of such
litigation and the diversion of the attention of our management and scientific personnel could outweigh any benefit we receive as a result
of the proceedings. Accordingly, despite our efforts, we may not be able to prevent third parties from infringing, misappropriating or
successfully challenging our intellectual property rights. Uncertainties resulting from the initiation and continuation of patent litigation
or other proceedings could have a negative impact on our ability to compete in the marketplace.
Any
trademarks we may obtain may be infringed or successfully challenged, resulting in harm to our business.
We
expect to rely on trademarks as one means to distinguish any of our product candidates that are approved for marketing from the products
of our competitors. While we have a corporate trademark, we have not yet selected trademarks for our product candidates and have not
yet begun the process of applying to register trademarks for our product candidates. Once we select trademarks and apply to register
them, our trademark applications may not be approved. Third parties may oppose our trademark applications, or otherwise challenge our
use of the trademarks. In the event that our trademarks are successfully challenged, we could be forced to rebrand our products, which
could result in loss of brand recognition and could require us to devote resources to advertising and marketing new brands. Our competitors
may infringe our trademarks and we may not have adequate resources to enforce our trademarks.
In
addition, any proprietary name we propose to use with our clinical-stage product candidates or any other product candidate in the United
States must be approved by the FDA, regardless of whether we have registered it, or applied to register it, as a trademark. The FDA typically
conducts a review of proposed product names, including an evaluation of the potential for confusion with other product names. If the
FDA objects to any of our proposed proprietary product names, we may be required to expend significant additional resources in an effort
to identify a suitable proprietary product name that would qualify under applicable trademark laws, not infringe the existing rights
of third parties and be acceptable to the FDA.
If
we are unable to protect the confidentiality of our trade secrets, our business and competitive position would be harmed.
In
addition to seeking patent and trademark protection for our product candidates, we also rely on trade secrets, including unpatented know-how,
technology and other proprietary information, to maintain our competitive position. We seek to protect our trade secrets, in part, by
entering into non-disclosure and confidentiality agreements with parties who have access to them, such as our employees, consultants,
advisors and other third parties. We also enter into confidentiality and invention or patent assignment agreements with our employees
and consultants. Despite these efforts, any of these parties may breach the agreements and disclose our proprietary information, including
our trade secrets. Monitoring unauthorized uses and disclosures of our intellectual property is difficult, and we do not know whether
the steps we have taken to protect our intellectual property will be effective. In addition, we may not be able to obtain adequate remedies
for any such breaches. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and
time-consuming, and the outcome is unpredictable. In addition, some courts in the jurisdiction in which we operate or intend to operate
are less willing or unwilling to protect trade secrets.
Moreover,
our competitors may independently develop knowledge, methods and know-how equivalent to our trade secrets. Competitors could purchase
our products and replicate some or all of the competitive advantages we derive from our development efforts for technologies on which
we do not have patent protection. If any of our trade secrets were to be lawfully obtained or independently developed by a competitor,
we would have no right to prevent them, or those to whom they communicate it, from using that technology or information to compete with
us. If any of our trade secrets were to be disclosed to or independently developed by a competitor, our competitive position would be
harmed.
Obtaining
and maintaining our patent protection depends on compliance with various procedural, document submission, fee payment and other requirements
imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for non-compliance with these requirements.
Periodic
maintenance and annuity fees on any issued patent are due to be paid to the patent offices and patent agencies over the lifetime of the
patent to maintain the patents that have been issued. Additionally, these offices and agencies require compliance with a number of procedural,
documentary, fee payment and other similar provisions during the patent application process. While an inadvertent lapse can in many cases
be cured by payment of a late fee or by other means in accordance with the applicable rules, there are situations in which noncompliance
can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the
relevant jurisdiction. Non-compliance events that could result in abandonment or lapse of a patent or patent application include failure
to respond to official actions within prescribed time limits, non-payment of fees and failure to properly legalize and submit formal
documents. If we or our licensors fail to maintain the patents and patent applications covering our products or product candidates, our
competitors might be able to enter the market, which would harm our business. In addition, to the extent that we have responsibility
for taking any action related to the prosecution or maintenance of patents or patent application in-licensed from a third party, any
failure on our part to maintain the in-licensed rights could jeopardize our rights under the relevant license and may expose us to liability.
If
we fail to comply with our obligations in the agreements under which we license our development or commercialization rights to products
or drug technologies from third parties, we could lose license rights that are important to our business.
We
hold a license from UCL Business plc (“UCLB”) for its technology related to co-stimulatory CAR-T in GD-T cells. This is in
addition to the intellectual property that we own. Our license with UCLB is for a single CAR-T binder, where we pay an annual license
fee, certain performance-based milestone payments and a single-digit royalty on sales arising from use of that binder together with certain
cumulative sales-based milestone payments. Through December 31, 2023 we have paid UCLB approximately $0.65 million (taking into consideration
fluctuation in exchange rates) in license fee payments. Furthermore, the Company has a duty not to breach terms of the license agreement.
If we fail to meet specific obligations, the licensor will have the right to terminate the applicable license or modify certain terms
of the license agreement. Royalty provisions cease upon termination or upon expiry of the license which occurs, on a country-by-country
basis, upon the later of the tenth (10th) anniversary of the first commercial sale of a licensed product or the lapse, expiry,
or revocation of all patents.
Risks
Related Ownership of Our ADSs and Warrants
Control
by a limited number of shareholders may limit the ability of investors to influence the outcome of director elections and other transactions
requiring shareholder approval.
As
of March 22, 2024, we had outstanding warrants to acquire 1,956,918 ADSs, and share options to purchase 920,230 ADSs. A significant number of such warrants have exercise prices above our ADSs’ recent trading prices, but the holders have
the right, in certain circumstances, to effect a cashless exercise of such warrants. If a significant number of such warrants and share
options are exercised by the holders, such persons together, along with several other long term significant shareholders, may have influence
over corporate actions requiring shareholder approval, including the following actions:
| |
● |
to
elect our directors; |
| |
|
|
| |
● |
to
amend or prevent amendment of our articles of association; |
| |
|
|
| |
● |
to
effect or prevent a merger, sale of assets or other corporate transaction; and |
| |
|
|
| |
● |
to
influence the outcome of any other matter submitted to our shareholders for vote. |
These
persons’ share ownership or warrant ownership may discourage a potential acquirer from making a tender offer or otherwise attempting
to obtain control of our company, which in turn could reduce the market price of our ADSs and Warrants or prevent our shareholders from
realizing a premium over the market price of our ADSs and Warrants.
If
we fail to meet the requirements for continued listing on the Nasdaq Capital Market or Nasdaq, our ADSs could be delisted from trading,
which would decrease the liquidity of our ADSs and our ability to raise additional capital.
Our
ADSs are currently listed for quotation on The Nasdaq Capital Market. We are required to meet specified financial requirements in order
to maintain our listing on the Nasdaq Capital Market. These requirements include maintaining a minimum bid price of at least $1.00 per
share for our ADSs, which is referred to as the Bid Price Rule, and maintaining a minimum market value of listed securities, or the MVLS,
of $35,000,000. On July 12 and 15, 2022, we received deficiency letters from the Listings Qualifications Department of the Nasdaq Stock
Market notifying that we were not in compliance with the Bid Price Rule and the MVLS, respectively. On December 6, 2022, we received
written notification from the listing qualifications staff of the Nasdaq Stock Market, LLC (“Nasdaq”) indicating that the
Company regained compliance with the Bid Price Rule. On January 12, 2023, we received written notification from the listing qualifications
staff of the Nasdaq indicating that we have not regained compliance with the MVLS and that our
securities would be subject to delisting unless we timely request a hearing before a Nasdaq Hearings Panel (the “Panel”).
On March 9, 2023 the Company presented a formal plan to regain compliance to the Panel. On March 17, 2023, the Company announced that
the TC BioPharm (Holdings) plc has been granted a formal extension until June 30, 2023, to regain compliance under Nasdaq Listing Rule
5550(b)(2) or its alternative criteria. On July 27, 2023, the Company received a letter, dated July 26, 2023 (the “Letter”)
from Nasdaq notifying the Company that the Panel has concluded that the Company has regained compliance with Nasdaq’s continued
listing requirements. The Letter stated that, pursuant to Listing Rule 5815(d)(4)(A), the Company will be subject to a Panel Monitor
for a period of one year from the date of the Letter. If, within that one-year monitoring period, the Listing Qualifications staff (the
“Staff”) finds the Company again out of compliance with any continued listing requirement, notwithstanding Rule 5810(c)(2),
the Company will not be permitted to provide the Staff with a plan of compliance with respect to any deficiency and the Staff will not
be permitted to grant additional time for the Company to regain compliance with respect to any deficiency, nor will the Company be afforded
an applicable cure or compliance period. Instead, the Staff will issue a Delist Determination Letter and the Company will have an opportunity
to request a new hearing with the initial Panel or a newly convened Hearings Panel if the initial Panel is unavailable.
On
June 22, 2023, we received a written notification from Nasdaq indicating that the minimum closing bid price per share for our ADSs was
below $1.00 for a period of 30 consecutive business days and that we did not meet the minimum bid price requirement set forth in Nasdaq
Listing Rule 5550(a)(2). Pursuant to Nasdaq Listing Rule 5810(c)(3)(A), we had a compliance period of 180 calendar days, or until December
19, 2023 (the “Compliance Period”), to regain compliance with Nasdaq’s minimum bid price requirement. If at any time
during the Compliance Period, the closing bid price per share of our ADSs is at least $1.00 for a minimum of ten consecutive business
days, Nasdaq will provide us with a written confirmation of compliance and the matter will be closed. On December 28, 2023, we received
a letter from Nasdaq indicating that we had not regained compliance with the rule and are not eligible for a second 180 day period. On
January 2, 2024, we received written confirmation from Nasdaq that it has determined that for the last 10 consecutive business days,
from December 15, 2023 to December 29, 2023, the closing bid price of our securities was at $1.00 per share or greater. Accordingly,
we regained compliance with Listing Rule 5550(a)(2) and the matter was closed. If we are deficient in maintaining the necessary listing
requirements in the future, our common stock may be delisted. If our common stock is delisted, an active trading market for our common
stock may not be sustained and the market price of our common stock could decline. Delisting of our common stock could adversely affect
our ability to raise additional capital through the public or private sale of equity securities, would significantly affect the ability
of investors to trade our securities and would negatively affect the value and liquidity of our common stock. Delisting could also have
other negative results, including the potential loss of confidence by employees, the loss of institutional investor interest and fewer
business development opportunities. For these reasons and others, delisting would adversely affect the liquidity, trading volume and
price of our common stock, causing the value of an investment in us to decrease and having an adverse effect on our business, financial
condition and results of operations.
We
do not intend to list any of our securities on any public securities exchange in the United Kingdom. This may limit the information available
to our security holders.
Our
ordinary shares and public warrants are not listed in the United Kingdom. As a result, we are not, and will not be, subject to the reporting
and other requirements of companies listed on a securities exchange in the United Kingdom. Accordingly, there may be less publicly available
information concerning our company than there would be if we were a public company listed in the United Kingdom, notwithstanding our
reporting under the SEC rules.
An
active and liquid market for the ADSs and/or public warrants may fail to continue, which could harm the market price of the ADSs and/or
Warrants, and an investor may not be able to resell their ADSs and/ or warrants at or above the acquisition price.
An
active public trading market for the ADSs and public warrants on the United States securities markets may not continue or be sustained.
In the absence of an active trading market for the ADSs and/or warrants, investors may not be able to sell their ADSs and warrants at
or above the price they paid for their securities or at the time when they would like to sell.
The
market price of the ADSs and public warrants is volatile and investors could lose all or part of their investment.
The
price of the securities of publicly traded emerging pharmaceutical and drug discovery and development companies has been highly volatile
and is likely to remain highly volatile in the future. As a result of this volatility, investors may not be able to sell their ADSs and
warrants at or above the purchase price or when they want to sell their securities. The market price of the ADSs and Warrants may fluctuate
significantly due to a variety of factors, including the following:
| |
● |
positive
or negative results of testing and clinical trials by us, strategic partners or competitors; |
| |
|
|
| |
● |
delays
in entering into strategic relationships with respect to development or commercialization of our investigational GD-T cell therapy
or any future therapeutic candidates; |
| |
|
|
| |
● |
entry
into strategic relationships on terms that are not deemed to be favorable to us; |
| |
|
|
| |
● |
technological
innovations or commercial therapeutic introductions by competitors; |
| |
|
|
| |
● |
changes
in government regulations and healthcare payment systems; |
| |
|
|
| |
● |
developments
concerning proprietary rights, including patent and litigation matters; |
| |
|
|
| |
● |
public
concern relating to the commercial value or safety of any of our investigational GD-T cell therapy or any future therapeutic candidates; |
| |
|
|
| |
● |
negative
publicity or public perception of the use of GD-T cells as a treatment therapy; |
| |
|
|
| |
● |
financing
or other corporate transactions; |
| |
|
|
| |
● |
publication
of research reports or comments by securities or industry analysts; |
| |
|
|
| |
● |
the
trading volume of the ADSs and warrants on Nasdaq; |
| |
|
|
| |
● |
sales
of our ordinary shares, including through deposit of additional ordinary shares with the depositary for the ADSs, by us, members
of our senior management and directors or our shareholders or the anticipation that such sales may occur in the future; |
| |
|
|
| |
● |
general
market conditions in the pharmaceutical industry or in the economy as a whole; |
| |
|
|
| |
● |
general
economic, political, and market conditions and overall market volatility in the United States or the UK as a result of the COVID-19
pandemic or other pandemics or similar events; and |
| |
|
|
| |
● |
other
events and factors, many of which are beyond our control. |
These
and other market and industry factors may cause the market price and demand for our securities to fluctuate substantially, regardless
of our actual operating performance, which may limit or prevent investors from readily selling their ADSs and warrants and may otherwise
negatively affect the liquidity of the ADSs and warrants. In addition, the stock market in general, and pharmaceutical companies in particular,
have experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance
of these companies.
The
public warrants are speculative in nature.
The
public warrants merely represent the right to acquire our ordinary shares at a fixed cash price, for a limited period of time. If the
warrants are not exercised before they expire, in six years from date of issue, the warrants will never provide any value to the holder
thereof. It is usual that the price of a warrant in the public market is more volatile than that of the corresponding shares for which
it is exercisable. Therefore, investors should expect the price of a warrant to be fluctuate to a greater degree than our ADSs, and correspondingly
be more speculative.
Holders
of our public and private warrants will not have any rights of the holders of ordinary shares until such warrants are exercised.
The
public and private warrants do not confer any of the rights afforded to the holders of our ordinary shares, even those ordinary shares
held through ADSs, such as voting rights or the right to receive dividends, but rather represent the right to acquire ordinary shares
at a fixed price.
We
have a significant number of public warrants outstanding, with fifty warrants to purchase one ADS, which may be exercised at a current
cash exercise price of $500.00 per ADS. There is no assurance that they will be exercised and that they will provide funding for the
Company.
As
at March 8, 2024, there were approximately 16,166,260 public warrants outstanding where fifty of each warrant are exercisable for one
ordinary share. Following the ratio change twenty ordinary shares can be dematerialized into one ADS. For the public Warrants to be exercised
on a cash basis, we must maintain an effective registration statement with the SEC at the time of their exercise. It is not expected
that any of the derivative securities will be exercised for cash unless the price of an ADS in the market is substantially above the
then exercise price. There can be no assurance that our ADS price will be sufficiently high on a sustained basis to encourage warrant
holders to exercise their derivative securities.
The
public warrants have a number of restrictions and reset provisions which may limit aspects of our operations and capital raising.
The
public warrant terms have restrictions on our ability to issue ordinary shares in a number of situations. For example, for the period
during which the lock up agreements are in place, we have limits on our ability to issue ordinary shares under our incentive plans. Another
restriction, one on our capital raising, is an exercise price reset provision; if we issue any ordinary shares, including instruments
convertible into ordinary shares, at a per share price or conversion price less than the exercise price, the then the exercise price
of the warrants will be reduced to the lower issue price permanently. The warrants have anti-dilution provisions including those for
recapitalization transactions such as a reverse stock split, stock dividend and forward stock split, and protective provisions in the
event of a rights offering, cash or asset dividend, and fundamental transactions consummated by the company where it is not the survivor.
The warrant has buy-in protection and cash penalties if we do not issue the securities underlying them on a timely basis. The six-year
term and number of warrants in combination with the registration obligation will be an overhang on the market while the warrants are
outstanding. This overhang may limit our ability to raise capital when needed at a price that represents the value of the company. The
warrants do not have a redemption provision by which we can either encourage their exercise or terminate the warrants.
We
have a significant number of private, Series E Warrants outstanding each warrant to purchase one ADS, which may be exercised at
a current cash exercise price of £1.5814 per ADS. There is no assurance that they will be exercised and that they will provide
funding for the Company.
As
at March 22, 2024, there were approximately 1,750,000 private Series E warrants with an exercise price of £1.5814, each of which
currently is exercisable for 20 ordinary shares. For these private Warrants to be exercised on a cash basis, we must maintain an effective
registration statement with the SEC at the time of their exercise. It is not expected that any of the derivative securities will be exercised
for cash unless the price of an ADS in the market is substantially above the then exercise price. There can be no assurance that our
ADS price will be sufficiently high on a sustained basis to encourage warrant holders to exercise their derivative securities, such as
the private warrants.
The
private warrants have a number of restrictions and reset provisions which may limit aspects of our operations and capital raising.
The
private warrant terms have restrictions on our ability to issue ordinary shares in a number of situations. For example, for the period
during which the lock up agreements are in place, we have limits on our ability to issue ordinary shares. The private warrants have anti-dilution
provisions including those for recapitalization transactions such as a reverse stock split, stock dividend and forward stock split, and
protective provisions in the event of a rights offering, cash or asset dividend, and fundamental transactions consummated by the company
where it is not the survivor. The private warrants have buy-in protection and cash penalties if we do not issue the securities underlying
them on a timely basis. The term and number of warrants in combination with the registration obligation will be an overhang on the market
while the private warrants are outstanding. This overhang may limit our ability to raise capital when needed at a price that represents
the value of the company. The private warrants do not have a redemption provision by which we can either encourage their exercise or
terminate the private Warrants.
We
incur increased costs as a result of operating as a Scottish public company listed in the U.S., and our board of directors is required
to devote substantial time to compliance requirements and corporate governance practices.
As
a Scottish public company listed in the U.S., we incur significant legal, accounting and other expenses that we did not incur as a private
company. In addition, the Sarbanes-Oxley Act, the Dodd-Frank Wall Street Reform and Consumer Protection Act, the listing requirements
of Nasdaq, and other applicable securities rules and regulations impose various requirements on foreign reporting public companies, including
the establishment and maintenance of effective disclosure and financial controls and corporate governance practices. Our board of directors,
management and other personnel devote a substantial amount of time to these compliance initiatives. Moreover, these rules and regulations
increase our annual legal and financial compliance costs and make some activities more time-consuming and costly. For example, we expect
that these rules and regulations may make it more difficult and more expensive for us to obtain director and officer liability insurance,
which in turn could make it more difficult for us to attract and retain qualified members of our board of directors.
However,
these rules and regulations are often subject to varying interpretations, in many cases due to their lack of specificity, and, as a result,
their application in practice may evolve over time as new guidance is provided by regulatory and governing bodies. This could result
in continuing uncertainty regarding compliance matters and higher costs necessitated by ongoing revisions to disclosure and governance
practices.
Pursuant
to Section 404(a) of the Sarbanes-Oxley Act, or Section 404, we are required to furnish a report by our board of directors on our internal
control over financial reporting. However, while we remain an emerging growth company, we are
not required to include an attestation report on internal control over financial reporting issued by our independent registered public
accounting firm. To achieve compliance with Section 404(b) within the prescribed period, we will be engaged in a process to document
and evaluate our internal controls over financial reporting, which is both costly and challenging. In this regard, we will need to continue
to dedicate internal resources, potentially engage outside consultants and adopt a detailed work plan to assess and document the adequacy
of internal control over financial reporting, continue steps to improve control processes as appropriate, validate through testing that
controls are functioning as documented and implement a continuous reporting and improvement process for internal control over financial
reporting. Despite our efforts, there is a risk that we will not be able to conclude that our internal
controls over financial reporting are effective as required by Section 404. If we identify one or more material weaknesses, it could
result in an adverse reaction in the financial markets due to a loss of confidence in the reliability of our financial statements.
We
are implementing appropriate accounting policies, processes and controls to comply with our expected expansion in scale of operations
and with Section 404. These activities include identifying and recruiting additional individuals with requisite expertise to assist in
implementation activities designed to strengthen our internal control over financial reporting to avoid future control deficiencies and
initiating the design and implementation of improvements to our financial control environment to address our future needs. However, we
cannot give assurance that the measures we have taken to date, and actions we plan to take in the future, will be sufficient to prevent
or avoid potential future additional material weaknesses in our controls.
If
we are unsuccessful in building an appropriate accounting infrastructure, we may not be able to prepare and disclose, in a timely manner,
our financial statements and other required disclosures, or comply with existing or new reporting requirements. Any failure to report
our financial results on an accurate and timely basis could result in sanctions, lawsuits, delisting of our shares from Nasdaq or other
adverse consequences that would materially harm our business. If we cannot provide reliable financial reports or prevent fraud, our business
and results of operations could be harmed and investors could lose confidence in our reported financial information. Any of the foregoing
occurrences, should they come to pass, could negatively impact the public perception of our company, which could have a negative impact
on the price of our publicly traded securities or our ability to sell other securities.
We
are an “emerging growth company” under the federal securities laws and we cannot be certain if the reduced disclosure requirements
applicable to emerging growth companies will make our securities less attractive to investors.
We
are an “emerging growth company,” as defined in Section 2(a) of the Securities Act, and we may take advantage of certain
exemptions from various reporting requirements that are applicable to public companies that are not “emerging growth companies”
including, but not limited to, not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley
Act, reduced disclosure obligations regarding executive compensation, and exemptions from the requirements of holding a nonbinding advisory
vote on executive compensation and shareholder approval of any golden parachute payments not previously approved. We cannot predict if
investors will find our securities less attractive because we may rely on these exemptions. Investors may be unable to compare our business
with other companies in our industry if they believe that our reporting is not as transparent as other companies in our industry. If
some investors, including persons considering an investment in the company, find our securities less attractive as a result, there may
be a less active trading market for our public securities, and the prices of these securities may be more volatile.
We
will remain an “emerging growth company” for up to five years, although we will lose that status sooner if our revenues exceed
$1.07 billion (or equivalent), if we issue more than $1 billion (or equivalent) in non-convertible debt in a three-year period, or if
the market value of our ordinary shares that is held by non-affiliates exceeds $700 million (or equivalent) as of any June 30.
Holders
of our ordinary shares will have their rights as a shareholder governed by Scottish law, and those rights differ from the rights of shareholders
under U.S. law.
We
are a public limited company under the laws of Scotland and United Kingdom. Therefore, the rights of holders of our ordinary shares,
including those represented by ADSs, are governed by the corporate law of Scotland and the United Kingdom and by our memorandum of association
and articles. The statutory framework that governs the Company is the Companies Act 2006 which is a UK-wide act and references to the
“UK Law” are to UK-wide legislation. These rights differ from the typical rights of shareholders in U.S. corporations. In
certain cases, facts that, under U.S. law, would entitle a shareholder in a U.S. corporation to claim damages may not give rise to a
cause of action or claim for damages under Scottish law. For example, the rights of shareholders to bring proceedings against the Company
or against our directors or officers in relation to public statements are more limited under Scottish law and UK Law than under the civil
liability provisions of the U.S. securities laws.
Investors
may face difficulties in protecting their interests, and their ability to protect their rights through the U.S. federal courts may be
limited, because we are incorporated outside the United States, conduct most of our operations outside the United States, and most of
our directors and senior management reside outside the United States.
We
are incorporated and have our registered office in, and are currently existing under the laws of, Scotland. In addition, most of our
tangible assets are located, and most of our senior management and certain of our directors reside, outside of the United States. As
a result, it may not be possible to serve process within the United States on certain directors or us or to enforce judgments obtained
in U.S. courts against such directors or us based on civil liability provisions of the securities laws of the United States.
The
United States and the UK do not currently have a treaty providing for recognition and enforcement of judgments (other than arbitration
awards) in civil and commercial matters. Consequently, a final judgment for payment of money given by a court in the United States, whether
or not predicated solely upon U.S. securities laws, would not automatically be recognized or enforceable in the UK. In addition, uncertainty
exists as to whether courts of Scotland would entertain original actions brought in Scotland against us or our directors or senior management
predicated upon the securities laws of the U.S. or any state in the U.S. Any final and conclusive monetary judgment for a definite sum
obtained against us in U.S. courts would be treated by the courts of Scotland as a cause of action in itself and sued upon as a debt
at common law so that no retrial of the issues would be necessary, provided that certain requirements are met.
Whether
these requirements are met in respect of a judgment based upon the civil liability provisions of the U.S. securities laws, including
whether the award of monetary damages under such laws would constitute a penalty, is subject to determination by the court making such
decision. If the courts of Scotland give a judgment for the sum payable under a U.S. judgment, the Scottish judgment will be enforceable
by methods generally available for this purpose. These methods generally permit the courts of Scotland discretion to prescribe the manner
of enforcement.
As
a result, U.S. investors may not be able to enforce against us or certain of our directors any judgments obtained in U.S. courts in civil
and commercial matters, including judgments under the U.S. federal securities laws.
As
a Scottish public limited company, certain capital structure decisions will require shareholder approval, which may limit our flexibility
to manage our capital structure.
Scottish
law provides that a board of directors may only allot shares (or grant rights to subscribe for or to convert any security into shares)
with the prior authorization of shareholders, such authorization stating the aggregate nominal amount of shares that it covers and being
valid for a maximum period of five years, each as specified in the articles of association or relevant ordinary resolution passed by
shareholders at a general meeting. Once allotted, the board of directors are free to issue the shares without further shareholder approval.
The authority from our shareholders to allot additional shares for a period of five years from January 14, 2022 was included in the ordinary
resolution passed by our shareholders on January 14, 2022, which authorization will need to be renewed upon expiration (i.e., at least
every five years) but may be sought more frequently for additional five-year terms (or any shorter period).
Scottish
law also generally provides shareholders with pre-emptive rights when new shares are issued for cash. However, it is possible for the
articles of association, or for shareholders to pass a special resolution at a general meeting, being a resolution passed by the members
(or of a class of members) of a company by a majority of not less than 75%, to disapply pre-emptive rights. Such a disapplication of
pre-emptive rights may be for a maximum period of up to five years from the date of adoption of the articles of association, if the disapplication
is contained in the articles of association, but not longer than the duration of the authority to allot shares to which this disapplication
relates or from the date of the shareholder special resolution, if the disapplication is by shareholder special resolution. In either
case, this disapplication would need to be renewed by our shareholders upon its expiration (i.e., at least every five years). Such authority
from our shareholders to disapply pre-emptive rights for a period of five years was included in the special resolution passed by our
shareholders on, January 14, 2022, which disapplication will need to be renewed upon expiration (i.e., at least every five years) to
remain effective, but may be sought more frequently for additional five-year terms (or any shorter period).
Scottish
law also generally prohibits a public company from repurchasing its own shares without the prior approval of shareholders by ordinary
resolution, being a resolution passed by a simple majority of votes cast, and other formalities. Such approval may be for a maximum period
of up to five years.
Our
business and results of operations may be negatively impacted by the UK’s withdrawal from the EU.
The
UK withdrew from the EU effective on January 31, 2020, and the transition period ended on December 31, 2020, which we refer to as Brexit.
The future regulations that will apply in the UK following the transition period (including financial laws and regulations, tax and free
trade agreements, intellectual property rights, data protection laws, supply chain logistics, environmental, health and safety laws and
regulations medicine licensing and regulations, immigration laws and employment laws), have yet to be fully addressed and continue to
be in transition, subject to change. The overall lack of clarity on future UK laws and regulations and their interaction with the EU
laws and regulations may negatively impact foreign direct investment in the UK, increase costs, depress economic activity and restrict
access to capital. As we are headquartered in the UK and have operations and clinical trials in the United Kingdom and EU, it is possible
that Brexit may impact some or all of our current operations and otherwise how we conduct business. For example, Brexit may impact our
ability to freely move employees from our headquarters in the UK to other locations in Europe, and it may impact the ability of European
therapists to move freely to the UK in order to complete part of their training or work on our clinical trials there.
The
long-term effects of Brexit will depend in part on the agreements the UK made during the Brexit transition period and thereafter to retain
access to markets in the EU. The Brexit withdrawal from the EU is unprecedented, and it is unclear how the UK’s access to the European
single market for goods, capital, services and labor within the EU, or single market, and the wider commercial, legal and regulatory
environment, will impact our current and future operations (including business activities conducted by third parties and contract manufacturers
on our behalf) and clinical activities in the UK In addition to the foregoing, our UK operations support our current and future operations
and clinical activities in the EU and European Economic Area, or EEA, and these operations and clinical activities could be disrupted
by Brexit.
We
may also face new regulatory costs and challenges that could have an adverse effect on our operations as a result of Brexit. The UK could
lose the benefits of global trade agreements negotiated by the EU on behalf of its member states, which may result in increased trade
barriers that could make our doing business in the EU and the EEA more difficult. Since the regulatory framework in the UK covering quality,
safety and efficacy of therapeutic substances, clinical trials, marketing authorization, commercial sales and distribution of therapeutic
substances is derived from EU directives and regulations, Brexit could materially impact the future regulatory regime with respect to
the approval of our GD-T cell therapy or any future therapeutic candidates in the UK. For instance, in November 2017, EU member states
voted to move the EMA, the EU’s regulatory body, from London to Amsterdam. Operations in Amsterdam commenced in March 2019, and
the move itself may cause significant disruption to the regulatory approval process in Europe. It remains to be seen how, if at all,
Brexit will impact regulatory requirements for therapeutic candidates and therapies in the UK. Any delay in obtaining, or an inability
to obtain, any regulatory approvals, as a result of Brexit or otherwise, would delay or prevent us from commercializing our investigational
GD-T cell therapy or future therapeutic candidates in the UK and/or the EU and restrict our ability to generate revenue and achieve and
sustain profitability. We may be forced to restrict or delay efforts to seek regulatory approval in the UK and/or EU for GD-T cell therapy
or any future therapeutic candidates, which could significantly and materially harm our business.
We
expect that Brexit, in the near and middle term will lead to certain legal uncertainties and potentially divergent national laws and
regulations as the UK determines which EU laws to replicate or replace, including those related to data privacy and the regulation of
medicinal products, as described above. Any of these effects of Brexit, and others we cannot anticipate, could negatively impact our
business and results of operations.
Our
business may be subject to risks related to possible Scottish independence from the UK
The
possibility of Scottish independence from the UK creates a range of uncertainties for Scotland based business in general, which would
require careful assessment by the board of directors and management as political events develop. There could be changes in currency,
taxation, general legislation, regulations and trading arrangements and agreements, together with economic prospects more generally.
It is not possible to predict the effect of Scottish independence if it were to occur and the changes introduced could have only limited
effect on the business, be beneficial to the business or could have a material adverse effect on the business’ revenue, financial
condition, profitability, prospects and results of operations.
A
transfer of ordinary shares, other than one effected by means of the transfer of book-entry interests, such as through our ADS program,
may be subject to United Kingdom stamp duty.
The
transfer of our ordinary shares effected by means of the transfer of book entry interests through our ADS program will generally not
be subject to United Kingdom stamp duty. However, if an investor holds its ordinary shares directly rather than beneficially through
the ADS program, any transfer of ordinary shares (including into the ADS program with a view to trading) would be likely to be subject
to United Kingdom stamp duty currently at the rate of 1.5% of the higher of the price paid or the market value of the shares acquired.
General
Risk Factors
Exchange
rate fluctuations may materially affect our results of operations and financial condition.
Due
to the international scope of our operations, the financial reporting of our assets, earnings and cash flows are influenced by movements
in exchange rates of several currencies, when comparing our results among different currencies such as the U.S. dollar, the pound sterling
and the euro. Our reporting currency and our functional currency is the pound sterling and the majority of our operating expenses are
paid in pound sterling. We regularly acquire services, consumables and materials in U.S. dollars, pound sterling and the euro. Further
potential future revenue may be derived from non-United Kingdom jurisdictions, particularly from the United States. As a result, our
business and the value of our ordinary shares, including those represented by the ADSs and underlying the Warrants, may be affected by
fluctuations in foreign exchange rates between the pound sterling and these other currencies, which may have a significant impact on
our results of operations and cash flows from period to period. Currently, we do not have any exchange rate hedging arrangements in place.
Collaborations,
whether through joint ventures, licensing, development arrangements, and other forms of agreements, will be important to our overall
business development.
In
common with many development stage biotechnology companies an element of our business plan is consider entering into collaborative arrangements
with larger pharmaceutical and biotechnology companies. We expect that future collaborations will provide us with important expertise,
aid in product development, conducting drug trials, facilitate market entry and may provide some level of funding or future revenue.
Notwithstanding our belief that collaborations will be beneficial to us, any collaboration arrangement may by their nature pose a number
of risks, including the following:
| |
● |
collaborators
have significant discretion in determining the efforts and resources that they will apply to a project; |
| |
|
|
| |
● |
collaborators
may not perform their obligations as expected; |
| |
|
|
| |
● |
collaborators
may dispute the amounts of payments owed; |
| |
|
|
| |
● |
collaborators
may not pursue development and commercialization of any product candidates that achieve regulatory approval or may elect not to continue
or renew development or commercialization programs or license arrangements |
In
the past, we have entered into collaborative arrangements with two partners, bluebird bio, Inc. (USA) and Nipro Corporation (Japan),
which involved funded or partly funded preclinical collaboration. Neither collaboration involve us in any current clinical or development
activity or are generating any current cash receipts for us. It is uncertain if these collaborations will generate any future cash receipts
or obligations for TCB.
As
a company based outside of the United States, our business is subject to economic, political, regulatory and other risks associated with
international operations.
Our
business is based in the United Kingdom and is subject to risks associated with conducting business outside of the United States. Many
of our suppliers and clinical trial relationships are located outside the United States, primarily in the United Kingdom and in the EU.
Accordingly, our future results could be harmed by a variety of factors, including:
| |
● |
economic
weakness, including inflation, or political change; |
| |
|
|
| |
● |
differing
and changing regulatory requirements for product approvals; |
| |
|
|
| |
● |
differing
jurisdictions could present different issues for securing, maintaining or obtaining freedom to operate in such jurisdictions; |
| |
|
|
| |
● |
potentially
reduced protection for intellectual property rights; |
| |
|
|
| |
● |
difficulties
in compliance with different, complex and changing laws, regulations and court systems of multiple jurisdictions and compliance with
a wide variety of foreign laws, treaties and regulations; |
| |
● |
changes
in non-U.S. currency exchange rates of the pound sterling, U.S. dollar, euro and currency controls; |
| |
|
|
| |
● |
changes
in a specific country’s or region’s political or economic environment, including the implications of the recent action
of the United Kingdom withdrawing from the European Union and efforts related to Scottish independence; |
| |
|
|
| |
● |
customs,
tariffs and trade barriers, trade protection measures, import or export licensing requirements or other restrictive actions by governments; |
| |
|
|
| |
● |
differing
medical product reimbursement regimes and price controls; |
| |
|
|
| |
● |
compliance
with tax, employment, immigration and labor laws for employees living or traveling abroad, including, for example, the variable tax
treatment in different jurisdictions of options granted under our share option schemes or equity incentive plans; |
| |
|
|
| |
● |
workforce
uncertainty in countries where labor unrest is more common than in the United Kingdom and the United States; |
| |
|
|
| |
● |
difficulties
associated with staffing and managing international operations, including differing labor relations; and |
| |
|
|
| |
● |
business
interruptions resulting from geo-political actions, including war and terrorism, or natural disasters including earthquakes, typhoons,
floods and fires. |
Unstable
market and economic factors could adversely affect our business, financial condition or results of operations.
Uncertain
or unfavorable global economic or market conditions, such as a recession, an economic slowdown, inflation or reduced growth rates, could
significantly impact our operating results or lead to significant reductions in funding sources available to the Company, which could
adversely affect our business, results of operations or financial condition. Our operations have required substantial amounts of cash
since inception. We expect to continue to spend substantial amounts to continue the development of our GD-T cell-based therapeutic candidates,
including for future clinical trials. In the event of unstable markets and unfavorable market conditions, we cannot be certain that additional
funding will be available on acceptable terms, or at all. We have no committed source of additional capital. If we are unable to raise
additional capital in sufficient amounts or on terms acceptable to us, we may have to significantly delay, scale back or discontinue
the development or commercialization of our GD-T cell-based therapeutic candidates or other research and development initiatives. Furthermore,
uncertain or unfavorable global economic or market conditions may cause our manufacturers, suppliers, distributors, contractors, logistics
providers and other external business partners to suffer financial or operational difficulties, which could impact their ability to provide
us with or distribute finished product, raw and packaging materials or services in a timely manner or at all. We could also face difficulty
collecting or recovering accounts receivables from third parties facing financial or operational difficulties.
The
conflict between Russia and Ukraine currently does not have any material impact on the company.
Our
operations primarily are undertaken in the United Kingdom and the resources we use primarily are domestically available. We have not
specifically sourced any resources for our operations from the Ukraine. We may be generally impacted by the macro-economic effects of
international sanctions and the effects of inflation, as it would affect all businesses.
Computer
system failures, cyber-attacks or deficiencies in our or related parties’ cyber security could result in a material disruption
of our product development programs, compromise sensitive information related to our business or trigger contractual and legal obligations,
any of which could potentially expose us to liability or reputational harm or otherwise adversely affect our business and financial results.
We
have implemented our security measures designed to protect the information (including but not limited to intellectual property, proprietary
business information and personal information) in our possession, custody or control. Our internal computer systems and those of current
and future third parties (such as vendors, CROs, collaborators or others) on which we rely may fail and are vulnerable to breakdown,
breach, interruption or damage from computer viruses, computer hackers, malicious code, employee error or malfeasance, theft or misuse,
denial-of-service attacks, sophisticated nation-state and nation-state-supported actors, unauthorized access, natural disasters, terrorism,
war, telecommunication and electrical failures or other compromise. Despite our security practices, there is a risk that we may be subject
to phishing and other cyberattacks in the future. The risk of a security breach or disruption, particularly through cyber-attacks or
cyber intrusion, including by computer hackers, foreign governments and cyber terrorists, has generally increased as the number, intensity
and sophistication of attempted attacks and intrusions from around the world have increased. We may not be able to anticipate all types
of security threats, and we may not be able to implement preventive measures effective against all such security threats. The techniques
used by cyber criminals change frequently, may not be recognized until launched, and can originate from a wide variety of sources, including
outside groups such as external service providers, organized crime affiliates, terrorist organizations or hostile foreign governments
or agencies. Our information technology and other internal infrastructure systems, including corporate firewalls, servers, leased lines
and connection to the Internet, face the risk of systemic failure that could disrupt our operations. If such an event were to occur and
cause interruptions in our operations, it could result in a material disruption of our development programs and our business operations.
For example, the loss of clinical trial data from completed or future clinical trials could result in delays in our regulatory approval
efforts and significantly increase our costs to recover or reproduce the data. Likewise, we rely on third parties for the manufacture
of our product candidates or any future product candidates and to conduct clinical trials, and similar events relating to their computer
systems could also have a material adverse effect on our business. To the extent that any disruption or security breach were to result
in a loss of, or damage to, our data or applications, or inappropriate use, disclosure of or access to confidential or proprietary information,
we could incur liability, our competitive position could be harmed and the further development and commercialization of our product candidates
or any future product candidates could be hindered or delayed. If we were to experience a significant cybersecurity breach of our information
systems or data, the costs associated with the investigation, remediation and potential notification of the breach to counterparties,
data subjects, regulators or others could be material. In addition, our remediation efforts may not be successful. Moreover, if the information
technology systems of our vendors, CROs, collaborators or other contractors or consultants become subject to disruptions or security
breaches, we may have insufficient recourse against such third parties and we may have to expend significant resources to mitigate the
impact of such an event, and to develop and implement protections to prevent future events of this nature from occurring. If we do not
allocate and effectively manage the resources necessary to build and sustain the proper technology and cybersecurity infrastructure,
we could suffer significant business disruption, including transaction errors, supply chain or manufacturing interruptions, processing
inefficiencies, data loss or the loss of or damage to intellectual property or other proprietary information. Furthermore, any such event
that leads to unauthorized access, use, or disclosure of personal information, including personal information regarding clinical trial
participants or employees, could harm our reputation, compel us to comply with federal and/or state breach notification laws and foreign
law equivalents, cause us to breach our contractual obligations, subject us to mandatory corrective action, and otherwise subject us
to liability under laws, regulations and contracts that protect the privacy and security of personal information, which could result
in significant legal and financial exposure and reputational damages. As cyber threats continue to evolve, we may be required to incur
significant additional expenses in order to enhance our protective measures or to remediate any information security vulnerability.
In
addition, in response to the ongoing COVID-19 pandemic, varying parts of our workforce are currently working remotely on a part- or full-time
basis. This could increase our cyber security risk, create data accessibility concerns, and make us more susceptible to communication
disruptions. Any of the foregoing could have a material adverse effect on our business, financial condition, results of operations or
prospects.
If
we are not able to maintain and enhance our reputation and brand recognition, our business, financial condition and results of operations
will be harmed.
We
believe that maintaining and enhancing our reputation and brand recognition is critical to our relationships with existing and future
third-party therapy locations, therapists, patients and collaborators, and to our ability to attract clinics to become our third-party
therapy locations offering our therapies. The promotion of our brand may require us to make substantial investments, and we anticipate
that, as our market becomes increasingly competitive, these marketing initiatives may become increasingly difficult and expensive. Brand
promotion and marketing activities may not be successful or yield increased revenue, and to the extent that these activities yield increased
revenue, the increased revenue may not offset the expenses we incur and our business, financial condition and results of operations could
be harmed. In addition, any factor that diminishes our reputation or that of our management, including failing to meet the expectations
of our network of third-party therapy locations, therapists and patients, could harm our reputation and brand and make it substantially
more difficult for us to attract new third-party therapy locations, therapists and patients. If we do not successfully maintain and enhance
our reputation and brand recognition, our business may not grow and we could lose our relationships with third-party therapy sites, therapists
and patients, which would harm our business, financial condition and results of operations.
We
are subject to anti-corruption laws, export control laws, customs laws, sanctions laws and other laws governing our operations. If we
fail to comply with these laws, we could be subject to civil or criminal penalties, other remedial measures and legal expenses, be precluded
from manufacturing our products and developing and selling our investigational therapies or any future therapeutic candidates or be required
to develop and implement costly compliance programs, which could adversely affect our business, results of operations and financial condition.
Our
operations are subject to anti-corruption laws, including the UK Bribery Act 2010, or Bribery Act, the U.S. Foreign Corrupt Practices
Act of 1977, as amended, or FCPA, and other anti-corruption laws that apply in countries where we do business and may do business in
the future. The Bribery Act, FCPA and these other laws generally prohibit us, our officers, and our employees and intermediaries from
bribing, being bribed or making other prohibited payments to government officials or other persons to obtain or retain business or gain
some other business advantage.
The
Bribery Act, the FCPA and these other laws generally prohibit us and our employees and intermediaries from authorizing, promising, offering,
or providing, directly or indirectly, a financial or other advantage to government officials or other persons to induce them to improperly
perform a relevant function or activity (or reward them for such behavior).
Under
the Bribery Act, we may also be liable for failing to prevent a person associated with us from committing a bribery offense. We, along
with those acting on our behalf and our commercial partners, operate in a number of jurisdictions that pose a high risk of potential
Bribery Act or FCPA violations, and we participate in collaborations and relationships with third parties whose corrupt or illegal activities
could potentially subject us to liability under the Bribery Act, FCPA or local anti-corruption laws, even if we do not explicitly authorize
or have actual knowledge of such activities. In addition, we cannot predict the nature, scope or effect of future regulatory requirements
to which our international operations might be subject or the manner in which existing laws might be administered or interpreted.
Compliance
with the FCPA, in particular, is expensive and difficult, particularly in countries in which corruption is a recognized problem. In addition,
the FCPA presents particular challenges in the pharmaceutical industry, because, in many countries, hospitals are operated by the government,
and doctors and other hospital employees are considered foreign officials. Certain payments to hospitals in connection with clinical
trials and other work have been deemed to be improper payments to government officials and have led to FCPA enforcement actions. Need
to dedicate additional resources to comply with numerous laws and regulations in each jurisdiction in which we plan to operate.
We
may in the future operate in jurisdictions that pose a high risk of potential Bribery Act or FCPA violations, and we may participate
in collaborations and relationships with third parties whose actions could potentially subject us to liability under the Bribery Act,
FCPA or local anti-corruption laws. In addition, we cannot predict the nature, scope or effect of future regulatory requirements to which
our international operations might be subject or the manner in which existing laws might be administered or interpreted. If we expand
our operations, we will need to dedicate additional resources to comply with numerous laws and regulations in each jurisdiction in which
we plan to operate.
We
are also subject to other laws and regulations governing our international operations, including regulations administered by the governments
of the UK and the U.S., and authorities in the EU, including applicable export control regulations, economic sanctions on countries and
persons, customs requirements and currency exchange regulations, collectively referred to as the Trade Control laws. In addition, various
laws, regulations and executive orders also restrict the use and dissemination outside of the United States, or the sharing with certain
non-U.S. nationals, of information classified for national security purposes, as well as certain products and technical data relating
to those products. If we expand our presence outside of the U.S., it will require us to dedicate additional resources to comply with
these laws, and these laws may preclude us from manufacturing our products and developing and selling our investigational therapies or
any future therapeutic candidates outside of the United States, which could limit our growth potential and increase our development costs.
There
is no assurance that we will be completely effective in ensuring our compliance with all applicable anti-corruption laws, including the
Bribery Act, the FCPA or other legal requirements, including Trade Control laws. If we are not in compliance with the Bribery Act, the
FCPA and other anti-corruption laws or Trade Control laws, we may be subject to criminal and civil penalties, disgorgement and other
sanctions and remedial measures, and legal expenses, which could have an adverse impact on our business, financial condition, results
of operations and liquidity. Any investigation of any potential violations of the Bribery Act, the FCPA, other anti-corruption laws or
Trade Control laws by UK, U.S. or other authorities could also have an adverse impact on our reputation, our business, results of operations
and financial condition.
Because
we are subject to environmental, health and safety laws and regulations, we may become exposed to liability and substantial expenses
in connection with environmental compliance or remediation activities which may adversely affect our business and financial condition.
Our
operations, including our research, development, testing and manufacturing activities, are subject to numerous environmental, health
and safety laws and regulations. These laws and regulations govern, among other things, the controlled use, manufacture, handling, release
and disposal of and the maintenance of a registry for, hazardous materials, such as chemical solvents, human cells, carcinogenic compounds,
mutagenic compounds and compounds that have a toxic effect on reproduction, laboratory procedures and exposure to blood-borne pathogens.
We
may incur significant costs to comply with these current or future environmental and health and safety laws and regulations. Furthermore,
if we fail to comply with such laws and regulations, we could be subject to fines or other sanctions.
As
with other companies engaged in activities similar to ours, we face a risk of environmental liability inherent in our current and historical
activities, including liability relating to releases of or exposure to hazardous materials and, as a result, may incur material liability
as a result of such release or exposure. Environmental, health and safety laws and regulations are becoming more stringent. We may incur
substantial expenses in connection with any current or future environmental compliance or remediation activities, in which case, our
production and development efforts may be interrupted or delayed and our financial condition and results of operations may be materially
adversely affected. In the event of an accident involving such hazardous materials, an injured party may seek to hold us liable for damages
that result.
Our
internal computer systems, or those of our future collaborators or other contractors or consultants, may fail or suffer security breaches,
which could result in a significant disruption of our product development programs and our ability to operate our business effectively.
Our
internal computer systems and those of our current and any future collaborators and other contractors or consultants are vulnerable to
damage from computer viruses, unauthorized access, natural disasters, terrorism, war and telecommunication and electrical failures. While
we have not experienced any significant system failure, accident or security breach to date, if such an event were to occur and cause
interruptions in our operations, it could result in a disruption of our development programs and our business operations, whether due
to a loss of our trade secrets or other proprietary information or other similar disruptions. For example, the loss of clinical trial
data from completed or future clinical trials could result in delays in our regulatory approval efforts and significantly increase our
costs to recover or reproduce the data. To the extent that any disruption or security breach were to result in a loss of, or damage to,
our data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur liability, our competitive
position could be harmed and the further development and commercialization of our product candidates could be delayed.
We
do not intend to pay dividends on our ordinary shares, including those represented by ADSs, so any returns will be limited to the market
value of the ADSs.
We
currently anticipate that we will retain future earnings for the development, operation, and expansion of our business and do not anticipate
declaring or paying any cash dividends for the foreseeable future. In addition, we may enter into agreements that prohibit us from paying
cash dividends without prior written consent from our contracting parties, or which include other terms prohibiting or limiting the amount
of dividends that may be declared or paid on our ordinary shares, including those represented by the ADSs. Furthermore, under UK corporate
law, a company’s accumulated realized profits, so far as not previously utilized by distribution or capitalization, must exceed
its accumulated realized losses so far as not previously written off in a reduction or reorganization of capital duly made (on a non-consolidated
basis), before dividends can be paid. In the future, were our dividend policy to change, a dividend or distribution may still be restricted
from being declared and paid. For these reasons, any return to shareholders may therefore be limited to the appreciation of their shares,
which may never occur.
Investors
in our ADSs may not receive distributions on our ordinary shares or any other value applicable to them if it is illegal or impractical
to make them available to holders of ADSs.
The
depositary for the ADSs has agreed to pay to the ADS holders the cash dividends or other distributions it or the custodian receives on
our ordinary shares or other deposited securities after deducting its fees and expenses. ADS holders will receive these distributions
in proportion to the number of our ordinary shares that the ADSs represent. In accordance with the limitations set forth in the deposit
agreement, however, it may be unlawful or impractical to make a distribution available to holders of ADSs. We have no obligation to take
any other action to permit distribution on the ADSs, ordinary shares, rights or anything else to holders of the ADSs. This means that
ADS holders may not receive the distributions we make on our ordinary shares or any value from them if it is unlawful or impractical
to make them available to the ADS holder. These restrictions may have an adverse effect on the value of the ADSs.
Holders
of the ADSs will not have the same voting rights as the holders of our ordinary shares, and may not receive voting materials or any other
documents that would need to be provided to our shareholders pursuant in time to be able to exercise their right to vote.
Holders
of the ADSs will not be able to exercise voting rights attaching to the ordinary shares represented by the ADSs. The deposit agreement
provides that, upon receipt of notice of any meeting of holders of our ordinary shares, the depositary will fix a record date for the
determination of ADS holders who shall be entitled to give instructions for the exercise of voting rights. Upon our request, the depositary
shall distribute to the holders as of the record date: (i) the notice of the meeting or solicitation of consent or proxy sent by us;
and (ii) a statement as to the manner in which instructions may be given by the holders. We cannot guarantee that ADS holders will receive
the voting materials in time to ensure that they can instruct the depositary to vote the ordinary shares underlying their ADSs.
ADS
holders will not be able to exercise their right to vote directly as a holder of ordinary shares, unless they surrender the ADSs they
hold to the depositary and withdraw the ordinary shares underlying such ADSs. Holders of ADSs may not know about the meeting far enough
in advance to cancel the ADSs and withdraw those ordinary shares. In addition, the depositary and its agents are not responsible for
failing to carry out voting instructions or for the manner of carrying out voting instructions. As a result, holders of ADSs may not
be able to exercise their right to vote, and there may be nothing they can do if the ordinary shares underlying their ADSs are not voted
as they requested or if their shares cannot be voted.
Holders
of Ordinary Shares and ADSs may not be able to participate in equity offerings we may conduct from time to time.
All shareholders
and holders of ADSs, including those in the United States have had pre-emption rights waived and therefore the presumption is that shareholders
and holders of ADSs do not have any right of future participation. Even in the case where preferential subscription rights have not been
cancelled or limited, shareholders and ADS holders may not be entitled to exercise such rights, unless the offering is registered or
the ordinary shares are qualified for sale under the relevant regulatory framework. As a result, there is the risk that investors in
ADSs may suffer dilution of their holdings should they not be permitted to participate in preference right equity or other offerings
that we may conduct in the future.
Holders
of ADSs may be subject to limitations on the transfer of their ADSs and the withdrawal of the underlying ordinary shares.
ADSs
are transferable on the books of the depositary. However, the depositary may close its books at any time or from time to time when it
deems expedient in connection with the performance of its duties. The depositary may refuse to deliver, transfer or register transfers
of ADSs generally when our books or the books of the depositary are closed, or at any time if we or the depositary think it is advisable
to do so because of any requirement of law, government or governmental body, or under any provision of the deposit agreement, or for
any other reason, subject to the right of ADS holders to cancel their ADSs and withdraw the underlying ordinary shares. Temporary delays
in the cancellation of the ADSs and withdrawal of the underlying ordinary shares may arise because the depositary has closed its transfer
books or we have closed our transfer books, the transfer of ordinary shares is blocked to permit voting at a shareholders meeting or
we are paying a dividend on our ordinary shares. In addition, ADS holders may not be able to cancel their ADSs and withdraw the underlying
ordinary shares when they owe money for fees, taxes and similar charges and when it is necessary to prohibit withdrawals in order to
comply with any laws or governmental regulations that apply to ADSs or to the withdrawal of ordinary shares or other deposited securities.
Holders
of ADSs may not be entitled to a jury trial with respect to claims arising under the deposit agreement, which could result in less favorable
outcomes to the plaintiff(s) in any such action.
The
deposit agreement governing the ADSs representing our ordinary shares provides that, to the fullest extent permitted by law, owners and
holders of ADSs irrevocably waive the right to a jury trial of any claim they may have against us or the depositary arising out of or
relating to the ADSs or the deposit agreement.
If
this jury trial waiver provision is not permitted by applicable law, an action could proceed under the terms of the deposit agreement
with a jury trial. If we or the depositary oppose a jury trial demand based on the waiver, the court would determine whether the waiver
was enforceable based on the facts and circumstances of that case in accordance with the applicable state and federal law. To our knowledge,
the enforceability of a contractual pre-dispute jury trial waiver in connection with claims arising under the federal securities laws
has not been finally adjudicated by the United States Supreme Court. However, we believe that a contractual pre-dispute jury trial waiver
provision is generally enforceable, including under the law of the State of New York, which governs the deposit agreement, by a federal
or state court in the City and County of New York, which has non-exclusive jurisdiction over matters arising under the deposit agreement.
In determining whether to enforce a contractual pre-dispute jury trial waiver provision, courts will generally consider whether a party
knowingly, intelligently and voluntarily waived the right to a jury trial. We believe that this is the case with respect to the deposit
agreement and the ADSs. It is advisable that investors consult legal counsel regarding the jury waiver provision before entering into
the deposit agreement.
If
holders or beneficial owners of ADSs bring a claim against us or the depositary in connection with matters arising under the deposit
agreement or the ADSs, including claims under federal securities laws, such holder or beneficial owner may not be entitled to a jury
trial with respect to such claims, which may have the effect of limiting and discouraging lawsuits against us and/or the depositary.
If a lawsuit is brought against us and/or the depositary under the deposit agreement, it may be heard only by a judge or justice of the
applicable trial court, which would be conducted according to different civil procedures and may result in different outcomes than a
trial by jury would have had, including results that could be less favorable to the plaintiff(s) in any such action, depending on, among
other things, the nature of the claims, the judge or justice hearing such claims, and the venue of the hearing.
No
condition, stipulation or provision of the deposit agreement or ADSs serves as a waiver by any holder or beneficial owner of ADSs or
by us or the depositary of compliance with U.S. federal securities laws and the rules and regulations promulgated thereunder.
Holders
of ADSs have limited choice of forum, which could limit their ability to obtain a favorable judicial forum for complaints against us,
the depositary or our respective directors, officers or employees.
The
deposit agreement governing the ADSs provides that: (i) the deposit agreement and the ADSs will be interpreted in accordance with the
law of the State of New York; and (ii) as an owner of ADSs, the investor irrevocably agrees that any legal action arising out of the
deposit agreement and the ADSs involving us or the depositary may only be instituted in a state or federal court sitting in the City
and County of New York. Any person or entity purchasing or otherwise acquiring any the ADSs, whether by transfer, sale, operation of
law or otherwise, shall be deemed to have notice of and have irrevocably agreed and consented to these provisions. This choice of forum
provision may increase costs and limit the ability to bring a claim in a judicial forum that the ADS holder finds favorable for disputes
with us, the depositary or our and the depositary’s respective directors, officers or employees, which may discourage such lawsuits
against us, the depositary and our and the depositary’s respective directors, officers or employees. However, it is possible that
a court could find such choice of forum provisions to be inapplicable or unenforceable. The enforceability of similar choice of forum
provisions has been challenged in legal proceedings. It is possible that a court could find this type of provisions to be inapplicable
or unenforceable.
To
the extent that any such claims may be based upon federal law claims, Section 27 of the Exchange Act creates exclusive federal jurisdiction
over all suits brought to enforce any duty or liability created by the Exchange Act or the rules and regulations thereunder. Furthermore,
Section 22 of the Securities Act creates concurrent jurisdiction for federal and state courts over all suits brought to enforce any duty
or liability created by the Securities Act or the rules and regulations thereunder. Accordingly, actions by our ADS holders to enforce
any duty or liability created by the Exchange Act, the Securities Act or the respective rules and regulations thereunder must be brought
in a federal court. Our ADS holders will not be deemed to have waived our compliance with the federal securities laws and the regulations
promulgated thereunder.
If
we are a “passive foreign investment company,” or a PFIC, in any particular year, a U.S. shareholder may be subject to adverse
U.S. federal income tax consequences.
Under
the Internal Revenue Code of 1986, as amended, or the Code, we will be a PFIC for any taxable year in which, after the application of
certain look-through rules with respect to our subsidiaries, either (i) 75% or more of our gross income consists of passive income or
(ii) 50% or more of the average quarterly value of our assets consists of assets that produce, or are held for the production of, passive
income (including cash). Passive income includes, among other things, dividends, interest, certain non-active rents and royalties, and
capital gains. Based on our operations, income, assets and certain estimates and projections, including as to the relative values of
our assets and the treatment of amounts in respect of refundable tax credits from governmental entities we received, or are or may become
entitled to receive, as gross income that is not passive income, we do not believe that we were a PFIC in 2020 and do not expect to be
a PFIC for our 2021 taxable year. However, the determination whether we are a PFIC is a fact-intensive determination that must be made
on an annual basis applying principles and methodologies that are in some circumstances unclear, and whether we will be a PFIC in 2022
or any future taxable year is uncertain because, among other things, (i) we currently own a substantial amount of passive assets, including
cash, (ii) the valuation of our assets that generate non-passive income for PFIC purposes, including our intangible assets, is uncertain
and may depend in part of the market price of the ADSs or, if applicable, our ordinary shares from time to time, which may fluctuate
substantially, (iii) the treatment of amounts in respect of refundable tax credits from governmental entities we received, or are or
may become entitled to receive, as gross income that is not passive income is uncertain, and (iv) the composition of our income may vary
substantially over time. Accordingly, there can be no assurance that we will not be a PFIC for any taxable year, and our U.S. counsel
expresses no opinion with respect to our PFIC status, or with respect to our expectations regarding our PFIC status in 2023 or any future
taxable year.
If
we are a PFIC for any taxable year during which a U.S. investor holds ADSs or ordinary shares, we would continue to be treated as a PFIC
with respect to that U.S. investor for all succeeding years during which the U.S. investor holds the ADSs or ordinary shares, even if
we ceased to meet the threshold requirements for PFIC status, unless certain exceptions apply. Such a U.S. investor may be subject to
adverse U.S. federal income tax consequences, including (i) the treatment of all or a portion of any gain on the disposition of the ADSs
or ordinary shares as ordinary income (and therefore ineligible for the preferential rates that apply to capital gains with respect to
some U.S. investors), (ii) the application of a deferred interest charge on such gain and the receipt of certain dividends on the ADSs
or the ordinary shares and (iii) compliance with certain reporting requirements. We do not intend to provide the information that would
enable investors to make a qualified electing fund election, or a QEF Election, with respect to their holding of ADSs or ordinary shares
that could mitigate the adverse U.S. federal income tax consequences to a U.S. investor should we be classified as a PFIC.
If
we are a controlled foreign corporation for U.S. federal income tax purposes, there could be adverse U.S. federal income tax consequences
to certain U.S. holders who own, directly, indirectly or by attribution, ten percent or more of our ordinary shares.
Each
“Ten Percent Shareholder” (as defined below) in a non-U.S. corporation that is classified as a “controlled foreign
corporation,” or a CFC, for U.S. federal income tax purposes generally is required to include in income for U.S. federal tax purposes
such Ten Percent Shareholder’s pro rata share of the CFC’s “Subpart F income”, investment of earnings in U.S.
property, and “global intangible low-taxed income”, even if the CFC has made no distributions to its shareholders. In addition,
a Ten Percent Shareholder that realizes gain from the sale or exchange of shares in a CFC may be required to classify a portion of such
gain as dividend income rather than capital gain. A non-U.S. corporation generally will be classified as a CFC for U.S. federal income
tax purposes if Ten Percent Shareholders own, directly, indirectly or constructively (through attribution), more than 50% of either the
total combined voting power of all classes of stock of such corporation entitled to vote or of the total value of the stock of such corporation.
A
“Ten Percent Shareholder” is a United States person (as defined by the Code) who owns or is considered to own 10% or more
of the total combined voting power of all classes of stock entitled to vote of such corporation or 10% or more of the total value of
the stock of such corporation. The determination of CFC status is complex and includes attribution rules, the application of which is
not entirely certain. A failure by a United States shareholder of a CFC to comply with its reporting obligations may subject the United
States shareholder to significant monetary penalties and other adverse tax consequences, and may extend the statute of limitations. We
cannot provide any assurances that we will assist U.S. holders in determining whether we or any of our non- U.S. subsidiaries are CFCs
or whether any holder is a Ten Percent Shareholder. We also cannot guarantee that we will furnish information that may be necessary to
comply with the aforementioned obligations. U.S. holders should consult their own advisors regarding the potential application of these
rules.
| Item
1B. |
Unresolved
Staff Comments |
None.
Risk
Management and Strategy
TC
BioPharm recognizes the critical importance of developing, implementing, and maintaining robust cybersecurity measures to safeguard our
information systems and protect the confidentiality, integrity, and availability of our data.
Managing
Material Risks & Integrated Overall Risk Management
Currently,
TC BioPharm does not have a formalized cybersecurity risk management process. However, the organization is working toward implementing
a framework for assessing, identifying, and managing material risks from cybersecurity threats. The IT department will work to continuously
evaluate and address cybersecurity risks in alignment with our business objectives and operational needs. This includes assessing cybersecurity
risk as part of an overall risk assessment and considering the likelihood and potential consequences of each risk. Plans also include
the identification of critical cybersecurity risks (e.g., malware, phishing, ransomware, and unauthorized access) and the implementation
of formalized mitigants to address those risks such as cybersecurity policies and incident response strategy.
Third-Party
Risk
TC
BioPharm does not currently engage with third parties in connection with cybersecurity risk management. Third-party consultants including
cybersecurity auditors are being considered for future engagement, at which point any risks stemming from the use of third parties will
be incorporated in the cybersecurity risk assessment.
Risks
from Cybersecurity Threats
TC
BioPharm has not encountered any cybersecurity risks or incidents that have materially impacted our business strategy, operational results,
or financial condition. We remain dedicated to maintaining a strong cybersecurity posture by continually analyzing and improving our
security procedures to reduce potential risks. This approach to cybersecurity is critical for protecting sensitive information and guaranteeing
the reliability of our business operations.
Governance
Board
of Directors Oversight
The
Board of Directors is acutely aware of the critical nature of managing risks associated with cybersecurity threats. The Board is committed
to effective governance in managing risks associated with cybersecurity threats because we recognize the significance of these threats
to our operational integrity and stakeholder confidence.
The
Board of Directors is informed of relevant cybersecurity risks and related updates during quarterly Board meetings by the CFO who is
also a Board member. There have been no critical, time sensitive cybersecurity updates thus far, but if this did occur it would be escalated
to the Board immediately.
Management’s
Role Managing Risk
Within
the management team, the responsibility for assessing and managing cybersecurity risk falls under the purview of the Senior IT Manager,
whose background includes a Qualification in Network System Engineering coupled with over two decades of experience in fortifying cybersecurity
for various companies. Moreover, TC BioPharm partners with a Managed IT service provider. This enriches our collective capabilities by
leveraging their extensive experience and specialized skills. This collaboration significantly bolsters our overall cybersecurity stance,
creating a well-rounded and robust defense against emerging threats.
Monitoring
Cybersecurity Incidents
TC
BioPharm’s Senior IT Manager monitors cybersecurity events and logs for unusual activity or potential security breaches within
the network. E-mail alerts are sent in real time as notification for any suspicious activity including phishing attempts, suspicious
attachments, and other e-mail-related security concerns. The Senior IT Manager also actively utilizes the WatchGuard & Webroot Advance
Security tools to conduct regular and thorough scans of the network infrastructure. The tools help identify vulnerabilities, malware,
and other potential threats, enabling a proactive measure to prevent and mitigate cybersecurity incidents.
Reporting
to Board of Directors
The
CFO holds biweekly meetings with the Senior IT Manager to discuss any pertinent information regarding cybersecurity risks. This includes
briefings on existing threat scenarios, updates on incident response efforts, and recommendations for enhancing our cybersecurity stance.
As a member of the Board of Directors, the CFO communicates key information to the remaining Board members.
Our
corporate headquarters and most of our operations, including our research and manufacturing facilities, are located at Maxim 1, 2 Parklands
Way, Holytown, Motherwell, ML1 4WR, United Kingdom. The lease for this space expires February 28, 2029 and covers a total leasable area
of approximately 26,300 square feet. We believe that our office facilities and the production and research facilities in the United Kingdom
are sufficient to meet our current needs.
| Item
3. |
Legal
Proceedings |
From
time to time, we are involved in various disputes, claims, suits, investigations, and legal proceedings arising in the ordinary course
of business. We believe that the resolution of current pending legal matters will not have a material adverse effect on our business,
financial condition, results of operations or cash flows. Nonetheless, we cannot predict the outcome of these proceedings, as legal matters
are subject to inherent uncertainties, and there exists the possibility that the ultimate resolution of these matters could have a material
adverse effect on our business, financial condition, results of operations or cash flows.
In
accordance with the terms of a Convertible Loan Note (‘Note’) on August 9, 2022 (the Conversion Date) the Company issued
183,820 Ordinary Shares and 367,640 listed warrants to the Note holder in full satisfaction of the Note in the aggregate amount of $781,233.
The holder filed a claim in the English courts on June 19, 2023 asserting that notice was provided such that the Company should have
paid it the value of the Note in cash, rather than by settling it through the issuance of Ordinary Shares and listed warrants. The holder
is demanding payment of the face value of the Note, together with interest, (approximately $860,000). The litigation process is in its
early stages and is not expected to conclude until late 2024 or later. The Company has retained English solicitors and is contesting the claim
in its entirety. The Company believes that it acted correctly under the terms of the Note and has accounted for the transaction on that
basis, and that no further amounts are payable to the holder.
| Item
4. |
Mine
Safety Disclosures |
None.
PART
II
| Item
5. |
Market
for Registrants Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities |
Our
share capital currently consists of ordinary shares. We may issue new classes of shares with such rights or restrictions as may be determined
by special (75%) resolution of the shareholders, including shares which are to be redeemed, or are liable to be redeemed at our option or
the option of the holder of such shares. The ADSs and public Warrants have been listed on the Nasdaq Capital Market under the symbols
“TCBP” and “TCBPW,” respectively, since February 11, 2022.
Dividends
We
have never declared or paid a dividend, and we do not anticipate declaring or paying dividends in the foreseeable future. We intend to
retain all available funds and any future earnings to fund the development and expansion of our business. Under Scottish law, among other
things, we may only pay dividends if we have sufficient distributable reserves (on a non-consolidated basis), which are our accumulated
realized profits that have not been previously distributed or capitalized less our accumulated realized losses, so far as such losses
have not been previously written off in a reduction or reorganization of capital.
Recent
Sales of Unregistered Securities
During
the year ended December 31, 2023, all sales of unregistered securities by the Company have been previously reported on a Form 6-K as
the Company qualified as a foreign private issuer during this period.
Issuer
Purchases of Equity Securities
We
did not repurchase any of our equity securities during the period covered by this Annual Report.
| Item
7. |
Management’s
Discussion and Analysis of Financial Condition and Results of Operations |
You
should read the following discussion and analysis of our financial condition and results of operations in conjunction with the financial
statements (prepared in accordance with accounting principles generally accepted in the United States (“U.S. GAAP”) and related
notes included elsewhere in this Annual Report on Form 10-K (this “Form 10-K”). The following discussion contains forward-looking
statements that are subject to risks and uncertainties. See “Special Note Regarding Forward-Looking Statements” for a discussion
of the uncertainties, risks, and assumptions associated with those statements. Actual results could differ materially from those discussed
in or implied by forward-looking statements as a result of various factors, including those discussed below and elsewhere in this Form
10-K, particularly in the section entitled “Risk Factors.” Unless we state otherwise or the context otherwise requires, the
terms “we,” “us,” “our” and the “Company” refer to TC BioPharm (Holdings) Limited and
its subsidiaries.
Overview
We
are a clinical-stage biopharmaceutical company with a cell-based product pipeline capable of treating a variety of disorders including
cancer and infectious disease. We are currently developing a pipeline of unmodified allogeneic GD-T therapies and next generation GD
CAR-T treatments with a number of advantages over conventional approaches. We own our main patent families in the GD CAR-T space, providing
robust IP protection and manufactures all products in-house, leading to a much lower cost of goods than competitor products.
Components
of Our Results of Operations
Revenues
We
do not have any approved products. Accordingly, we have not generated any revenue from the sale of products, and we do not expect to
generate any such revenue unless and until we obtain regulatory approvals for, and commercialize any of, our product candidates. In the
future, we will seek to generate revenue primarily from product sales and, potentially, regional or global collaborations with strategic
partners, which may produce license fee income.
During
the year ended December 31, 2022, we had two collaboration agreements. Revenue arose under these contracts as a result of (i) our recharging
development costs incurred by us under those agreements to our partners and (ii) on upfront payments received under those collaboration
agreements, which were taken to revenue on a straight-line basis over the estimated term over which the services promised will be provided.
This term was estimated by management at the inception of each contract and re-evaluated at each reporting date. The Company ceased to have an effective obligation to
continue to provide unpaid services from December 7, 2022. Thus, noting that a) the Company did not have any additional obligations to the collaboration partner and 2) the upfront payment (that
was recognized as deferred revenue) was non-refundable we recognized any remaining deferred revenue upon termination of the contract in
December of 2022.
Since
inception through December 31, 2023, the Company has received £14.5 million in pre-clinical payments connected with CAR-T development
partnerships. These partnerships are no longer actively being progressed and it is unlikely that we will receive any future milestone
revenues.
Operating
Expenses
We
classify our operating expenses into two categories: research and development expenses and administrative expenses. Personnel costs,
including salaries, benefits, bonuses and share-based payment expense, comprise a significant component of each of these expense categories.
We allocate expenses associated with personnel costs based on the function performed by the respective employees.
Research
and Development Expenses
The
largest component of our total operating expenses since inception has been costs related to our research and development activities,
including the preclinical and clinical development of our product candidates.
Research
and development costs are expensed as incurred, with our development activities not yet at the point at which capitalization can occur
under GAAP. Our research and development expense primarily consist of:
| |
● |
consumable
costs related to research and development of pharmaceutical or biologic therapy products for preclinical studies and clinical trials; |
| |
|
|
| |
● |
costs
related to manufacturing active pharmaceutical or biologic therapy products for preclinical studies and clinical trials; |
| |
|
|
| |
● |
salaries
and personnel-related costs, including bonuses, benefits and any share-based payment expense, for our personnel performing research
and development activities or managing those activities that have been out-sourced; |
| |
|
|
| |
● |
fees
paid to consultants and other third parties who support our product candidate development; |
| |
● |
third
party costs incurred in connection with preclinical studies and clinical trials from investigative sites and contract research organizations,
or CROs; |
| |
|
|
| |
● |
other
costs incurred in seeking regulatory approval of our product candidates; |
| |
|
|
| |
● |
costs
of related office space allocated to our research and development function, materials and equipment; and |
| |
|
|
| |
● |
payments
under our license agreements. |
The
successful development of our product candidates is highly uncertain. Product candidates in later stages of clinical development generally
have higher development costs than those in earlier stages of clinical development, primarily due to the increased size and duration
of later-stage clinical trials. In addition, the cost of development of our CAR-T range of products is likely to be substantially higher
than costs incurred historically in the development of our unmodified products. Accordingly, we expect research and development costs
to increase significantly for the foreseeable future as programs progress. However, we do not believe that it is possible at this time
to accurately project total program-specific expenses through commercialization. We are also unable to predict when, if ever, material
net cash inflows will commence from our product candidates to offset these expenses. Our expenditures on current and future preclinical
and clinical development programs are subject to numerous uncertainties in timing and cost to completion.
The
duration, costs and timing of clinical trials and development of our product candidates will depend on a variety of factors including:
| |
● |
the
scope, rate of progress, results and expenses of our ongoing and future clinical trials, preclinical studies and research and development
activities; |
| |
|
|
| |
● |
the
potential need for additional clinical trials or preclinical studies requested by regulatory agencies; |
| |
|
|
| |
● |
potential
uncertainties in clinical trial enrolment rates or drop-out or discontinuation rates of patients; |
| |
|
|
| |
● |
competition
with other drug development companies in, and the related expense of, identifying and enrolling patients in our clinical trials and
contracting with third-party manufacturers for the production of the drug product needed for our clinical trials; |
| |
|
|
| |
● |
the
achievement of milestones requiring payments under in-licensing agreements; |
| |
|
|
| |
● |
any
significant changes in government regulation; |
| |
|
|
| |
● |
the
terms and timing of any regulatory approvals; |
| |
|
|
| |
● |
the
expense of filing, prosecuting, defending and enforcing patent claims and other intellectual property rights; and |
| |
|
|
| |
● |
the
ease, cost and ability to market, commercialize and achieve market acceptance for any of our product candidates, if approved. |
We
track research and development expenses on a program-by-program basis for both clinical-stage and preclinical product candidates. Manufacturing,
clinical trial and preclinical research and development expenses are assigned or allocated to individual product candidates. We do not
allocate employee costs or facility expenses, including depreciation or other indirect costs, to specific programs because these costs
are deployed across multiple programs and, as such, are not separately classified. We use internal resources primarily to oversee research
and development as well as for managing our preclinical development, process development, manufacturing and clinical development activities.
Administrative
Expenses
Administrative
expenses consist of personnel costs, other administrative expenses and other expenses for outside professional services, including legal,
audit and accounting services. Personnel costs consist of salaries, bonuses, benefits and share-based compensation expense. Other administrative
expenses include office space-related costs not otherwise allocated to research and development expense, professional fees and costs
of our information systems. We anticipate that our administrative expenses will continue to increase in the future as we increase our
headcount to support our continued research and development and potential commercialization of our product candidates. We expect to continue
to incur additional expenses as a public company, including expenses related to compliance with the rules and regulations of the SEC
and Nasdaq, additional insurance expenses, and expenses related to investor relations activities and other administrative and professional
services.
Administrative
Expenses – Costs to prepare for listing
Administrative
costs to prepare for our IPO consist of professional services such as legal, investor relations, accounting, audit, and other administrative
services.
Change
in fair value of derivative liability
The gain/loss relates to the movement in the estimated fair value of the embedded derivative related to the issue of Convertible
Loan Notes, calculated by using a Black Scholes option pricing model at the end of each reporting period. The gain/loss relates to
the movement in the estimated fair value of our warrants, calculated by using a Black Scholes option pricing model at the end of
each reporting period. As it pertains to the issued warrants, it is important to note that as of December 31, 2023 they were
primarily equity classified and therefore are no longer required to be re-measured to fair value at the end of each reporting
period.
Interest
Expense
Interest
expense includes the effective interest charge accrued in relation to the Convertible Loan Notes. Interest expense is offset by interest income related to interest earned on our cash and cash equivalents and short-term deposits.
Income
Tax Credit
We
are subject to corporate taxation in the United Kingdom. Due to the nature of our business, we have generated losses since inception.
Our income tax credit recognized represents the sum of the research and development tax credits recoverable in the United Kingdom.
As
a company that carries out extensive research and development activities, we benefit from the United Kingdom research and development
tax credit regime and are able to surrender some of our losses for a cash rebate of up to 33.35% of expenditures related to eligible
research and development projects. Qualifying expenditures largely comprise clinical trial and manufacturing costs, employment costs
for relevant staff and consumables incurred as part of research and development projects. Certain subcontracted qualifying research and
development expenditures are eligible for a cash rebate of up to 21.68%. A large portion of costs relating to our research and development,
clinical trials and manufacturing activities are eligible for inclusion within these tax credit cash rebate claims.
There
can be no certainty that we will be able to continue to claim research and development tax credits in the future. Tax losses that have
not been utilized to offset taxable income or surrendered in connection with the aforementioned research and development tax credits
are carried forward to be offset against future taxable profits. In the event we generate revenues in the future, we may benefit from
the United Kingdom’s government’s “patent box” initiative that allows profits attributable to revenues from patents
and/or patented products registered in the United Kingdom or European Union to be taxed at a lower rate than other streams of revenue.
The current rate of tax for relevant streams of revenue for companies receiving this relief is 10%.
Results
of Operations
Comparison
of the Years Ended December 31, 2023, and 2022
The
following table summarizes our results of operations:
| | |
For the Year
Ended | | |
For the Year
Ended | | |
| | |
| |
| | |
December
31, 2023 | | |
December
31, 2022 | | |
£
Change | | |
%
Change | |
| | |
| | |
| | |
| | |
| |
| Revenue | |
£ | - | | |
£ | 3,844,532 | | |
£ | (3,844,532 | ) | |
| (100 | )% |
| | |
| | | |
| | | |
| | | |
| | |
| Operating expenses: | |
| | | |
| | | |
| | | |
| | |
| Research and development
expenses | |
| 7,771,391 | | |
| 7,592,470 | | |
| 178,921 | | |
| 2 | % |
| Administrative expenses | |
| 6,467,932 | | |
| 7,030,972 | | |
| (563,040 | ) | |
| (8 | )% |
| Administrative expenses
- costs to prepare for listing | |
| - | | |
| 1,305,087 | | |
| (1,305,087 | ) | |
| (100 | )% |
| Total
operating expenses | |
| 14,239,323 | | |
| 15,928,529 | | |
| (1,689,206 | ) | |
| (11 | )% |
| Loss
from operations | |
| (14,239,323 | ) | |
| (12,083,997 | ) | |
| (2,155,326 | ) | |
| 18 | % |
| | |
| | | |
| | | |
| | | |
| | |
| Other income (expense): | |
| | | |
| | | |
| | | |
| | |
| Loss on modification of
convertible loan | |
| (645,845 | ) | |
| (140,344 | ) | |
| (505,501 | ) | |
| 360 | % |
| Change in fair value of derivative liability | |
| 8,052,581 | | |
| 16,064,945 | | |
| (8,012,364 | ) | |
| (50 | )% |
| Foreign currency losses | |
| (80,070 | ) | |
| (120,974 | ) | |
| 40,904 | | |
| (34 | )% |
| Interest expense | |
| (83,025 | ) | |
| (6,753,231 | ) | |
| 6,670,206 | | |
| (99 | )% |
| Total
other income (expense), net | |
| 7,243,641 | | |
| 9,050,396 | | |
| (1,806,755 | ) | |
| (20 | )% |
| Net loss before income taxes | |
| (6,995,682 | ) | |
| (3,033,601 | ) | |
| (3,962,081 | ) | |
| 131 | % |
| Income tax credit | |
| 1,088,729 | | |
| 1,720,000 | | |
| (631,271 | ) | |
| (37 | )% |
| | |
| | | |
| | | |
| | | |
| | |
| Net
loss | |
£ | (5,906,953 | ) | |
£ | (1,313,601 | ) | |
£ | (4,593,352 | ) | |
| 350 | % |
Revenue
Revenue
decreased by £3.8 million for the year ended December 31, 2023 compared to for the year ended December 31, 2022. Revenue from
upfront payments in connection with collaboration agreements is recognized over the estimated term over which the services promised
will be provided. This term was estimated by management at the inception of each contract and evaluated at each reporting date.
Management reviewed the status of the contract and specific contractual terms and concluded that at December 31, 2022, no further
services were required to be provided under the contract. As such, the remaining deferred revenue, which reflected a non-refundable upfront payment received from our collaboration
partner was fully recognized during December 2022. Thus, no revenue stemming from the upfront payment was recognized during the year ended December 31, 2023.
Research
and Development Expenses
| | |
Year
Ended December 31, | | |
| | |
| |
| | |
2023 | | |
2022
| | |
| | |
| |
| | |
£’000’s | | |
£’000’s
| | |
£
Change | | |
%
Change | |
| Direct research and development expenses by
program: | |
| | | |
| | | |
| | | |
| | |
| Unmodified
cell therapy programs(1) | |
£ | 3,212 | | |
£ | 1,257 | | |
£ | 1,955 | | |
| 155.5 | % |
| Other research and development
programs(2) | |
| 64 | | |
| 136 | | |
| (72 | ) | |
| (52.7 | )% |
| | |
| | | |
| | | |
| | | |
| | |
| Total direct research and development expense | |
| 3,276 | | |
| 1,393 | | |
| 1,883 | | |
| 135.2 | % |
| | |
| | | |
| | | |
| | | |
| | |
| Research and development and unallocated costs: | |
| | | |
| | | |
| | | |
| | |
| Personnel related (including
share-based compensation) | |
| 3,365 | | |
| 4,504 | | |
| (1,139 | ) | |
| (25.3 | )% |
| Indirect research and development
expense(3) | |
| 1,130 | | |
| 1,695 | | |
| (565 | ) | |
| (33.4 | )% |
| | |
| | | |
| | | |
| | | |
| | |
| Total research and development
expenses | |
£ | 7,771 | | |
£ | 7,592 | | |
£ | 179 | | |
| 2.4 | % |
| |
(1) |
Unmodified
cell therapy programs include OmnImmune® and ImmuniStim® |
| |
(2) |
Other
research and development programs include expenditure on areas such as our CAR-T program and induced pluripotent stem cells (iPSCs). |
| |
(3) |
Indirect
research and development expense includes property related costs and depreciation and amortization. |
Research
and development expenses increased by 2.4% to £7.8 million for the year ended December 31, 2023 from £7.6 million for the year
ended December 31, 2022 reflecting the increased work on unmodified cell therapy programs, which increased by 156%. Personnel costs partially
offset the increase, which decreased by 25% for the year ended December 31, 2023 compared to December 31, 2022.
General
and administrative
| | |
Year
Ended December 31, | | |
| | |
| |
| | |
2023 | | |
2022 | | |
£
Change | | |
%
Change | |
| | |
£’000’s | | |
£’000’s | | |
| | |
| |
| Share-based compensation expense | |
| 295 | | |
| 719 | | |
£ | (424 | ) | |
| (59.0 | )% |
| Employee-related costs | |
| 2,443 | | |
| 2,396 | | |
| 47 | | |
| 2.0 | % |
| Legal & professional fees | |
| 3,383 | | |
| 3,569 | | |
| (186 | ) | |
| (5.2 | )% |
| Other expenses | |
| 347 | | |
| 347 | | |
| - | | |
| 0.0 | % |
| Total
administrative expenses | |
| 6,468 | | |
| 7,031 | | |
| (563 | ) | |
| (8.0 | )% |
Administrative
expenses decreased by 8% to £6.5 million for the year ended December 31, 2023 from £7.0 million for the year ended December
31, 2022. The decrease was primarily driven by a decrease in share-based compensation expense and a decrease in legal and professional
fees.
Loss
on modification of convertible loan
The
change in the loss on modification of convertible loan primarily relates to modifications made to the loan in 2023 as compared to
2022. The convertible loan was fully settled (via either cash settlement or conversion) during the year ended December 31, 2023.
Change
in fair value of derivative liability
The change in fair value of derivative liability is comprised of the change in fair values of the convertible loan derivative, warrant
derivative, and other derivatives. The
change in the fair value of the embedded convertible loan derivatives relates to the movement in the estimated fair value of the embedded
derivatives during the year ended December 31, 2023 (ie. as compared to December 31, 2022), which was calculated by using the Black Scholes
option pricing model. The convertible loan was fully settled (via either cash settlement or conversion) during the year ended December
31, 2023.
The
change in fair value of the warrant derivatives of £8.1 million for the year ended December 31, 2023 relates
to the movement in the estimated fair value of our issued detachable warrants. The warrants were issued at the time of the IPO and
at various times during each of the two years ended December 31, 2023. In addition, certain warrants were both modified and induced
over the course of our fiscal year ended December 31, 2023. All of our issued warrants are valued by using the Black Scholes option
pricing model.
Foreign
Currency Losses
The
decrease in foreign currency losses for the year ended December 31, 2023 compared to for the year ended December 31, 2022 was primarily
due to a lower foreign exchange rate during 2023.
Interest
Expense
Interest
expense was approximately £0.1 million for the year ended December 31, 2023 compared to £6.8 million for the year ended December
31, 2022. The decrease was due to the Convertible Loan Note being paid off during the year. Interest expense was partially offset by interest income earned on cash accounts.
Income
tax credit
The
research and development tax credit of £1.1 million was 37% lower for the year ended December 31, 2023 compared to £1.7 million
for the year ended December 31, 2022. This was due to lower levels of expenditure eligible for research and development tax credits.
After
accounting for tax credits receivable, there were accumulated tax losses for carry forward in the United Kingdom of £16.4 million
as of December 31, 2023. Unrecognized deferred tax assets totaling £4.2 million as of December 31, 2023 consist of temporary differences
on tax losses and share-based compensation arrangements. No deferred tax asset is recognized in respect of accumulated tax losses or
temporary differences on share-based compensation arrangements because future profits are not sufficiently certain.
Going
Concern
Our
existing cash of £2.5 million at December 31, 2023 will not be sufficient to enable us to conduct our business 12 months from the
issuance of these financial statements. We will need additional funding to complete the development and research of our products. If
we are unable to raise capital when needed or on acceptable terms, we would be forced to delay, reduce, or eliminate our research and
development efforts.
As
a result of the Company’s recurring losses from operations, and the need for additional financing to fund its operating and capital
requirements, there is uncertainty regarding the Company’s ability to maintain liquidity sufficient to operate its business effectively,
which raises substantial doubt as to the Company’s ability to continue as a going concern.
Liquidity
and Capital Resources
For
the years ended December 31, 2023 and 2022, we incurred net losses of £5.9 million and £1.3 million, respectively. We used
£10.5 million of cash in operating activities in the year ended December 31, 2023 and used £15.3 million of cash in operating
activities for the year ended December 31, 2022.
As
of December 31, 2023, and December 31, 2022, we had cash and cash equivalents of £2.5 million and £4.8 million, respectively.
From incorporation through December 31, 2023, we have financed our operations primarily through our IPO, private placements of equity
securities, convertible loans, government grants, research and development tax credits, and receipts from partner for collaborative research
and development services totaling £82.1 million.
While we
work towards obtaining regulatory approval to advance any of our GD-T cell therapeutic candidates into pivotal clinical trials or to
commercialization, we will incur significant research and development expenses, and also commercialization expenses related to product
sales, marketing, manufacturing and distribution and additional funding would be required. Where appropriate, we will seek to fund our
operations through milestone payments under our agreements with collaboration partners and additional equity financings.
Cash
Flows
The
following tables summarize the results of our cash flows for the below respective periods:
| | |
Year
Ended December 31, | |
| | |
2023 | | |
2022 | |
| Net cash provided by (used in): | |
| | | |
| | |
| Operating activities | |
£ | (10,544,870 | ) | |
£ | (15,292,297 | ) |
| Investing activities | |
£ | (208,789 | ) | |
£ | (310,197 | ) |
| Financing activities | |
£ | 8,520,376 | | |
£ | 18,653,214 | |
| Change in cash | |
£ | (2,345,451 | ) | |
£ | (3,241,372 | ) |
Operating
Activities
Net
cash used in operating activities was £10.5 million for the year ended December 31, 2023. The net loss for the year ended December
31, 2023 was £5.9 million, which was offset by noncash items of £6.2 million, consisting of £0.6 million in depreciation
and amortization, £0.4 million in share-based compensation expense, a £0.6 million loss on modification of the Convertible
Loan Note, an £8.1 million change in the fair value of the derivative liability, a £0.1 million net foreign exchange loss, and £0.1 million in noncash interest expense. Changes
in working capital amounted to £1.5 million, which consisted of an increase in the corporation tax receivable, operating lease
right of use assets, and accounts payable and accrued expenses. Prepaid expenses and other current assets and lease liabilities decreased
working capital.
Net
cash used in operating activities was £15.3 million for the year ended December 31, 2022. The net loss for the year ended December
31, 2022 was £1.3 million, which was offset by noncash items of £7.3 million, consisting of £0.8 million in depreciation
and amortization, £1.1 million in share-based compensation expense, a £0.1 million loss on modification of the Convertible
Loan Note, a £16.1 million change in the fair value of the derivative liability, a £0.1 million net foreign exchange
loss, and £6.6 million in noncash interest expense. Changes in working capital used £6.7 million in cash.
Investing
Activities
Net
cash used in investing activities was £0.2 million and £0.3 million for the years ended December 31, 2023 and 2022, respectively.
These amounts relate primarily to purchases of property, plant and equipment related to our facility and patent filing costs.
Financing
Activities
Net
cash from financing activities was £8.5 million and £18.7 million for the years ended December 31, 2023 and 2022, respectively.
For the year ended December 31, 2023, these amounts consisted of net proceeds from sale of own shares and warrants for £9.3 million
offset by issuance costs of £0.8 million. For the year ended December 31, 2022, these amounts consisted of net proceeds from sale
of own shares and warrants for £22.2 million offset by the repayment of convertible loan notes of £2.6 million.
Funding
Requirements
We
expect our expenses to increase substantially in connection with our ongoing activities, particularly as we advance the preclinical activities
and clinical trials of our product candidates. Our expenses will increase as we (i) advance our product candidates through phases of
clinical development and, potentially, registration, (ii) fund our research and development activities to further expand our GD-T cell
technologies and develop future product candidates and follow-on versions of our more advanced product candidates, (iii) fund our manufacturing
activities and the expansion of our plant to support our ongoing and future clinical trials and potential commercial launch; and (iv)
fund our general operations.
Since
February 10, 2022, we have been a publicly traded company and incur significant legal, accounting and other expenses that we were not
required to incur as a private company. In addition, the Sarbanes-Oxley Act of 2002, as well as rules adopted by the SEC and The Nasdaq
Stock Market, requires public companies to implement specified corporate governance practices. We expect to continue to incur substantial
legal and financial compliance costs, which may make some activities more time-consuming and costly.
We
will require additional capital to continue to conduct our business and implement our business plans.
Because
of the numerous risks and uncertainties associated with research, development and commercialization of pharmaceutical product candidates,
we are unable to estimate the amount of our future working capital requirements, which will depend on and are likely to increase significantly
as a result of many uncertain factors, including:
| |
● |
the
scope, progress, outcome and costs of our clinical trials and other research and development activities; |
| |
|
|
| |
● |
the
costs, timing, receipt and terms of any marketing approvals from applicable regulatory authorities; |
| |
|
|
| |
● |
the
costs of future sales and marketing activities, including cost of product sales, medical regulatory affairs, marketing, manufacturing
and distribution, for any of our product candidates for which we receive marketing approval; |
| |
|
|
| |
● |
the
amount and timing of the receipt of any future revenue from commercial sale of our products, should any of our product candidates
receive marketing approval and become successful in the market; |
| |
|
|
| |
● |
the
impact of the COVID-19 pandemic on our ability to progress research and development and clinical trials; |
| |
|
|
| |
● |
the
costs and timing of hiring new employees to support our future growth; |
| |
|
|
| |
● |
the
costs of preparing, filing and prosecuting patent applications, maintaining and enforcing our intellectual property rights and defending
intellectual property-related claims; and |
| |
|
|
| |
● |
the
cost of and extent to which we in-license or acquire additional product candidates or technologies. |
Until
such time, if ever, that we can generate product revenue sufficient to achieve profitability, we expect to finance our future cash needs
through equity offerings and debt and a combination thereof, including securities convertible into ordinary shares and through development
collaborations with partners. To the extent that we raise additional capital through the sale of equity, our shareholders’ ownership
interest will be diluted.
If
we raise additional funds through other third-party funding, collaborations agreements, strategic alliances, licensing arrangements or
marketing and distribution arrangements, we may have to relinquish valuable rights to our technologies, future revenue streams, research
programs or product candidates or grant licenses on terms that may not be favorable to us.
If
we are unable to raise additional funds when needed, we may be required to delay, limit, reduce or terminate our product development
or future commercialization efforts or grant rights to develop and market products or product candidates that we would otherwise prefer
to develop and market ourselves. If we raise funding through borrowings, we may have to enter into onerous covenants which may adversely
impact our operations and our ability to obtain further funding.
There
is no assurance that we will be able to raise any further funding, or if further funding is offered, it will be on terms that are acceptable
to us and may bring dilution which is unacceptable to our shareholders.
Contractual
Obligations
The
following table summarizes our contractual obligations as of December 31, 2023 and the effects that such obligations are expected to
have on our liquidity and cash flows in future periods:
| | |
Payments
due by period | |
| | |
Total | | |
Less
Than 1 Year | | |
1
- 3 Years | | |
4
- 5 Years | | |
More
Than 5 Years | |
| | |
| | | |
| | | |
| | | |
| | | |
| | |
| Trade payables | |
£ | 1,847,279 | | |
£ | 1,847,279 | | |
£ | – | | |
£ | – | | |
£ | – | |
| Lease liabilities | |
| 2,235,075 | | |
| 447,015 | | |
| 894,030 | | |
| 894,030 | | |
| – | |
| Payables related to clinical trial testing | |
| 1,177,500 | | |
| 1,177,500 | | |
| – | | |
| – | | |
| – | |
| Other payables | |
| 1,407,061 | | |
| 1,407,061 | | |
| – | | |
| – | | |
| – | |
| Total commitments | |
£ | 6,666,915 | | |
£ | 4,878,855 | | |
£ | 894,030 | | |
£ | 894,030 | | |
£ | - | |
Lease
liabilities
Amounts
shown as lease liabilities and similar reflect minimum payments due for our leases of office, laboratory and manufacturing space. We
entered into a lease for our corporate headquarters in April 2014 and, as part of this agreement, exercised an option to lease additional
space in January 2017 and March 2019. The overall lease expires in February 2029.
Other
commitments
We
enter into contracts in the normal course of business with third parties who support us in the conduct of certain specialist aspects
of clinical trials and preclinical research studies and testing. These contracts are generally cancellable by us upon prior notice. Payments
due upon cancellation consist only of payments for services provided or expenses incurred, including noncancelable obligations of our
service providers, up to the date of cancellation. These payments are not included in the preceding table, as the amount and timing of
such payments are not known.
We
have not included any contingent payment obligations that we may incur upon achievement of clinical, regulatory and commercial milestones,
as applicable, or royalty payments that we may be required to make under in-licensing agreements which we have or may enter into which
could be payable if any of our products generate future sales or license revenue as the amount, timing and likelihood of such payments
are not known and are not anticipated in the near term or before we generate significant revenues.
Off-balance
sheet arrangements
We
did not have during the periods presented, and we do not currently have, any off-balance sheet arrangements, as defined in the rules
and regulations of the SEC.
Critical
Accounting Policies and Estimates
Our
financial statements and accompanying notes have been prepared in accordance with U.S. GAAP. The preparation of these financial statements
requires us to make estimates, judgments and assumptions that affect the reported amounts of assets, liabilities, revenues, costs and
expenses, and related disclosures. On an ongoing basis, we continually evaluate our estimates and assumptions believed to be reasonable
under current facts and circumstances. Actual amounts and results may materially differ from these estimates made by management under
different assumptions and conditions.
Certain
accounting policies that require significant management estimates and are deemed critical to our results of operations or financial position,
are described below. Accordingly, these are the policies we believe are the most critical to aid in fully understanding and evaluating
our financial condition and results of operations.
Going
Concern
Our
evaluation of our ability to continue as a going concern requires us to evaluate our future sources and uses of cash sufficient to fund
our currently expected operations in conducting research and development activities one year from the date our consolidated financial
statements are issued. We evaluate the probability associated with each source and use of cash resources in making our going concern
determination. The research and development of cell therapies is inherently subject to uncertainty.
Management
believes that its existing cash balances will only be able to fund current operations to May 2024. Should the additional
planned financings not occur as expected, management will implement alternative arrangements and such arrangements could have a potentially
significant negative impact on the current net asset value of the Company. These alternatives include: (1) raising additional capital
by means other than those planned through equity and/or debt financings; (2) entering into new commercial relationships to help fund
future clinical trial costs (i.e. licensing and partnerships); (3) reducing and/or deferring discretionary spending on general corporate
overheads and one or more of our research and development and / or clinical programs; and/or (4) restructuring operations to change our
overhead structure and make use of our manufacturing facilities to generate revenues from through third party manufacturing contracts.
In the medium term, the Company’s future liquidity needs, and ability to address those needs, will largely be determined by the
success of its product candidates and key development and regulatory events and its decisions in the future.
As
a result of the Company’s recurring losses from operations, and the need for additional financing to fund its operating and capital
requirements, there is uncertainty regarding the Company’s ability to maintain liquidity sufficient to operate its business effectively,
which raises substantial doubt as to the Company’s ability to continue as a going concern.
Revenue
from contracts with customers
Identification
of contracts with pharma partners
The
Company entered into collaboration agreements with a number of parties. Application of Accounting Standard Codification (“ASC”)
606, Revenue from Contracts with Customers, on collaboration agreements requires judgement around whether these contracts fall
within the scope of ASC 606. The Company’s core business is around researching and developing immunotherapies and the contracts
entered into with pharma partners were consistent with those objectives and the outputs are in line with the Company’s ordinary
activities. The contracts with pharma partners did not involve sharing the risks and benefits of a joint arrangement in the sense of Topic
808 “Collaborative arrangements”. In light of work undertaken with pharma partners, and the fact that these agreements
had commercial substance with clearly defined milestones and rights and obligations for each party, management concluded that these
collaboration agreements met the definition of a contract with a customer and fall within the scope of ASC 606.
Identification
of performance obligations in contracts
The
collaboration agreements entered into by the Company included obligations to fulfil the research and development programs. The Company
identified, from reviews of the relevant agreements, that there are no specific obligations but an implied performance obligation to
deliver each overall contracted research and development program. Due to the broad nature of these obligations, spanning the full duration
of the contract, the obligations were satisfied over the expected duration of the relevant contract.
Determination
and allocation of the transaction price
The
collaboration agreements included a number of elements of consideration and were allocated to the satisfaction of the relevant obligation.
The Company can receive upfront payments as part of the consideration. The Company has determined that upfront payments are in connection
with the performance of the research and development program and are satisfied during the duration of the contract.
The
business is entitled to receive contractual milestone payments on achievement of certain performance obligations, with revenue being
recognized in the same way. The relevant transaction price is allocated to the related milestone.
Key
Sources of Estimation Uncertainty
The
key assumptions concerning the future, and other key sources of estimation uncertainty at the balance sheet date, that have a significant
risk of causing a material adjustment to the carrying amounts of assets and liabilities within the next year are discussed below.
Convertible
loan
The
Company established a $20.0 million convertible loan note instrument in April 2021. During the year ended December 31, 2023, the Company
converted loan notes totaling $809,692 (£619,315) into ordinary shares and warrants over ordinary shares such that no loan notes
were outstanding as at December 31, 2023. During the year to December 31, 2022, the Group converted loan notes totaling $14,228,245 (£10,506,174)
into ordinary shares and warrants over ordinary shares and repaid loan notes totaling $3,195,765 (£2,632,324). The Company analyzed
the conversion feature of the convertible loan note under ASC 815-Derivatives and Hedging. Where there are outstanding amounts under
the convertible loan note, these are recorded as a liability on the consolidated balance sheet at the reporting date. The conversion
feature for any such outstanding amounts requires liability treatment on the consolidated balance sheet and was recorded at fair value
with changes to the fair value being recorded through the consolidated statement of operations.
Except
for the loan notes described below, all other loan notes were repaid or converted into ordinary shares and warrants over ordinary shares
180 days after the listing date.
On
August 9, 2022, the Company agreed with one of the loan note holders not to exercise the right to require the loan notes to be repaid
in cash in accordance with the terms of the loan notes and to amend certain other aspects of the loan notes (“amended loan notes”).
As additional consideration, the Company has issued warrants to subscribe for 233,560 ordinary shares in the share capital of the Company.
Except for the amended loan notes, all other loan notes were repaid or converted into ordinary shares and warrants over ordinary shares
180 days after the listing date.
The
changes note above represent a substantial amendment as the modifications are related to:
| |
(i) |
Removing
the exercise of the right to require the loan in cash as of August 9, 2022. |
| |
(ii) |
Extending
the repayment date to January 31, 2023 and modifying the structure to be repaid in shares if not redeemed before in cash. |
| |
(iii) |
Revising
the conversion price for the conversion of the loan notes in shares. The revised conversion price would be $0.50 and, if the 5-day
trailing VWAP of the Company’s ADS is above that and $0.20 as a floor. |
| |
(iv) |
Giving
the option to the holder for redemption in cash, which will occur no later than February 10, 2023 and to the Company for an early
redemption at any moment but having the Holder an option to convert into shares using the revised conversion price at that moment. |
On
April 3, 2023, the Company agreed with the loan note holder not to exercise the right to require the loan notes to be repaid in cash
in accordance with the terms of the loan notes and to amend certain other aspects of the loan notes (“2023 amended loan notes”).
As additional consideration, the Company issued warrants to subscribe for 4,000,000 ordinary shares of the Company. These warrants
contained a condition whereby if a registration statement, to be filed by the Company, registering all of the securities underlying the
note holder’s amended convertible loan note, was not declared effective by July 31, 2023, the note holder will be entitled to receive 0.30 Ordinary
Shares for each share it was originally entitled to purchase under these warrants without the payment of any additional consideration.
No such registration statement was filed. The related fair value of the issue of any additional securities is approximately $37,000 and
is not considered material to the financial statements.
The
modifications to the 2023 amended loan notes represent as substantial amendment as the modifications are related to:
(i)
A waiver to any defaults arising in connection with the 2022 amended loan notes.
(ii)
Extending the repayment date to January 15, 2024; and
(iii)
Amend the Conversion Price (as defined in the Loan Note) of the outstanding loan notes to be the lesser of $1.00 or the lowest closing
price of the Ordinary Shares during the ten (10) day period prior to the date the Noteholder delivers a notice of conversion to the Company,
not to be lower than $0.20.
In
accordance with ASC 470, an exchange between an existing borrower and lender of debt instruments with substantially different terms shall
be accounted for as an extinguishment of the original financial liability (with the associated gain or loss presented in the statement
of operations) and the recognition of a new financial liability. In addition, as consideration for these modifications, the Company has
issued additional warrants to subscribe for 4,000,000 ordinary shares of the Company.
The
original financial instrument was derecognized, including any unamortized transaction costs, and the new instrument was initially recognized
at fair value and subsequently measured at amortized cost at each reporting date.
The
conversion option is a single embedded derivative that is separately recognized as a liability and accounted for at fair value through
the statement of operations. The conversion options are financial liabilities in accordance with ASC 815 because the Company issues shares
such that the fair value of the shares delivered is always equal to the amount of the contractual obligation (i.e. a variable number
of shares depending on the share price of the stock). As a result, the conversion options are part of the financial liability debt instrument
and should be evaluated under the embedded derivatives guidance. Because the conversion options are indexed to the equity of the issuer,
these are not closely related to the host contract. This instrument is considered a new freestanding financial instrument and constitutes
an embedded derivative liability that is separately recognized as a liability and subsequently adjusted to its fair value at each reporting
period, which is presented in the statement of operations.
As
noted earlier, the Company converted loan notes totaling $809,692 (£619,315) into ordinary shares and warrants over ordinary shares
such that no loan notes were outstanding as at December 31, 2023.
Derivative
liability
We
evaluate all of our financial instruments to determine if such instruments are derivatives or contain features that qualify as embedded
derivatives, pursuant to ASC 480 and ASC 815. Warrants that do not meet the criteria for equity treatment in accordance with the guidance
contained in ASC 815-40 have been recorded as liabilities. Accordingly, we classify these warrants as liabilities at their fair value
and adjust the warrants their fair value at each reporting period. This liability is subject to re-measurement at each balance sheet
date until exercised, and any change in fair value is recognized in our statement of operations.
Pre-funded
warrants
The
Pre-Funded Warrants are classified as a component of equity because they are freestanding financial instruments that are legally detachable
and separately exercisable from the shares of common stock with which they were issued, are immediately exercisable, do not embody an
obligation for the Company to repurchase its shares, and permit the holders to receive a fixed number of ordinary shares upon exercise
(foreign exchange on nominal value of the shares is not considered relevant for the analysis because not more than an insignificant amount
related to the value of the share remains outstanding which is the $0.0001 nominal amount that remains open to be paid upon exercising
it). In addition, Pre-Funded Warrants do not provide any guarantee of value or return.
| Item
7A. |
Quantitative
and Qualitative Disclosures about Market Risk |
We
are exposed to a variety of risks in the ordinary course of our business, including, but not limited to, currency risk, liquidity risk
and credit risk, as discussed below. We regularly assess each of these risks to minimize any adverse effects on our business as a result
of those factors.
Credit
risk
Credit
risk is the risk of financial loss to the Company if a customer or counterparty to a financial instrument fails to meet its contractual
obligations and arises principally from the Company’s receivables from customers and from its financing activities, including deposits
with banks and financial institutions, foreign exchange transactions and other financial instruments. The Company only engages with banks
and financial institutions with a Standard and Poor credit rating of BBB or greater. The Company has a small number of customers as part
of its collaboration agreements. To manage the credit risks around collaboration agreements, the Company will assess the creditworthiness
of partners as part of the engagement process. The Company has monitoring procedures in place to identify and follow up on any overdue
debts. Credit risk from balances with banks and financial institutions is managed by the Company’s finance department in accordance
with the Company’s policy to only place funds with approved counterparties with the appropriate credit rating. The Company is exposed
to no material credit risk.
Liquidity
risk
Liquidity
risk is the risk that necessary sources of funding for the Company’s business activities may not be available. The Company manages
liquidity risk by maintaining adequate reserves, banking facilities and reserve borrowing facilities, by continuously monitoring forecast
and actual cash flows, and by matching the maturity profiles of financial assets and liabilities. The Company is utilizing shareholder
funds, collaboration agreements, grant funding and asset finance to support its working capital requirements. All cash funds are held
with a maturity of three months or less.
Market
risk
Market
risk is the risk that the fair value or future cash flows of a financial instrument will fluctuate because of changes in market prices.
Market risk comprises three types of risk: interest rate risk, currency risk and other price risk, such as equity price risk and commodity
risk.
Interest
rate risk
Interest
rate risk is the risk that the fair value or future cash flows of a financial instrument will fluctuate because of changes in market
interest rates. The Company is not exposed to any material interest rate risk.
Currency
risk
The
Company has transactions denominated in various currencies, with the principal currency exposure being fluctuations in U.S. Dollars and
Euros against pound sterling. The Company’s exposure to the risk of changes in foreign exchange rates relates primarily to the
Company’s Convertible Loan Notes that are denominated in US Dollars and a limited number of supplier agreements denominated in
currencies other than pound sterling.
Equity
price risk
The
Convertible Loan Notes issued by the Group contained an embedded derivative component that was accounted for at fair value at each
period end. As such, a change in the estimated underlying price per share impacted the valuation of the embedded
derivative. The convertible debt was fully settled as of December 31, 2023.
| Item
8. |
Financial
Statements and Supplementary Data |
INDEX
TO FINANCIAL STATEMENTS
Report
of Independent Registered Public Accounting Firm
To
the Shareholders and Board of Directors of
TC
BioPharm (Holdings) plc
Opinion
on the Financial Statements
We
have audited the accompanying consolidated balance sheets of TC BioPharm (Holdings) plc (the “Company”) as of December 31,
2023 and 2022, the related consolidated statements of operations, shareholders’ equity and cash flows for each of the two years
in the period ended December 31, 2023, and the related notes (collectively referred to as the “financial statements”). In
our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of December
31, 2023 and 2022 , and the results of its operations and its cash flows for each of the two years in the period ended December 31 2023,
in conformity with accounting principles generally accepted in the United States of America.
Explanatory
Paragraph – Change in Reporting Framework
As
discussed in Note 3 to the financial statements, the Company has changed its reporting framework from International Financial Reporting
Standards as issued by the International Accounting Standards Board to accounting principles generally accepted in the United States
of America.
Explanatory
Paragraph – Going Concern
The
accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As more
fully described in Note 1, the Company, has incurred significant losses and needs to raise additional funds to meet its obligations and
sustain its operations. These conditions raise substantial doubt about the Company’s ability to continue as a going concern. Management’s
plans in regard to these matters are also described in Note 1. The consolidated financial statements do not include any adjustments that
might result from the outcome of this uncertainty.
Basis
for Opinion
These
financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s
financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight
Board (United States) (“PCAOB”) and are required to be independent with respect to the Company in accordance with the U.S.
federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We
conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audits
to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or
fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting.
As part of our audits we are required to obtain an understanding of internal control over financial reporting but not for the
purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly,
we express no such opinion.
Our
audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to
error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence
regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles
used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe
that our audits provide a reasonable basis for our opinion.
/s/
Marcum llp
Marcum LLP
We
have served as the Company’s auditor since 2022.
New
York, NY
April 1, 2024
TC
BIOPHARM (HOLDINGS) PLC
CONSOLIDATED
BALANCE SHEETS
| | |
December
31, 2023 | | |
December
31, 2022 | |
| | |
| | |
| |
| ASSETS | |
| | | |
| | |
| Current assets: | |
| | | |
| | |
| Cash and cash
equivalents | |
£ | 2,462,609 | | |
£ | 4,808,060 | |
| Corporation tax receivable | |
| 1,043,593 | | |
| 1,720,000 | |
| Prepaid
expenses and other current assets | |
| 2,194,725 | | |
| 919,456 | |
| Total current assets | |
| 5,700,927 | | |
| 7,447,516 | |
| | |
| | | |
| | |
| Non-current assets: | |
| | | |
| | |
| Property, plant, and equipment,
net | |
| 1,274,798 | | |
| 1,761,171 | |
| Operating lease right of
use assets | |
| 1,340,769 | | |
| 1,530,274 | |
| Intangible
assets, net | |
| 615,170 | | |
| 553,016 | |
| Total assets | |
| 8,931,664 | | |
| 11,291,977 | |
| | |
| | | |
| | |
| LIABILITIES AND SHAREHOLDERS’
EQUITY | |
| | | |
| | |
| Current liabilities: | |
| | | |
| | |
| Accounts payable and accrued
liabilities | |
| 4,431,840 | | |
| 2,159,058 | |
| Convertible loan | |
| - | | |
| 653,484 | |
| Derivative liability | |
| 13,437 | | |
| 6,023,302 | |
| Current
portion of operating lease liability | |
| 305,324 | | |
| 328,033 | |
| Total current liabilities | |
| 4,750,601 | | |
| 9,163,877 | |
| Non-current operating
lease liability | |
| 1,495,833 | | |
| 1,796,835 | |
| Total liabilities | |
| 6,246,434 | | |
| 10,960,712 | |
| | |
| | | |
| | |
| Shareholders’ Equity: | |
| | | |
| | |
| Ordinary shares, £0.0001 par value, 20,570,088 and 949,958 authorized,
issued, and outstanding as of December 31, 2023 and
2022, respectively | |
| 2,057 | | |
| 95 | |
| Deferred shares, £0.4999 par value, 794,955 and 794,955 authorized, issued, and outstanding as of December
31, 2023 and 2022, respectively | |
| 397,398 | | |
| 397,398 | |
| Additional paid-in capital | |
| 41,123,065 | | |
| 33,308,568 | |
| Accumulated deficit | |
| (38,837,290 | ) | |
| (33,374,796 | ) |
| Total shareholders’
equity | |
| 2,685,230 | | |
| 331,265 | |
| Total liabilities and
shareholders’ equity | |
£ | 8,931,664 | | |
£ | 11,291,977 | |
The
accompanying footnotes are an integral part of these consolidated financial statements.
TC
BIOPHARM (HOLDINGS) PLC
CONSOLIDATED
STATEMENTS OF OPERATIONS
| | |
For the Year
Ended | | |
|
| | |
December
31, 2023 | | |
December
31, 2022 | |
| | |
| | |
| |
| Revenue | |
£ | - | | |
£ | 3,844,532 | |
| | |
| | | |
| | |
| Operating expenses: | |
| | | |
| | |
| Research and development
expenses | |
| 7,771,391 | | |
| 7,592,470 | |
| Administrative expenses | |
| 6,467,932 | | |
| 7,030,972 | |
| Administrative
expenses - costs related to preparing for listing | |
| - | | |
| 1,305,087 | |
| Total
operating expenses | |
| 14,239,323 | | |
| 15,928,529 | |
| Loss from operations | |
| (14,239,323 | ) | |
| (12,083,997 | ) |
| | |
| | | |
| | |
| Other income (expense): | |
| | | |
| | |
| Loss on modification of
convertible loan | |
| (645,845 | ) | |
| (140,344 | ) |
| Change in fair value of derivative liability | |
| 8,052,581 | | |
| 16,064,945 | |
| Foreign currency losses | |
| (80,070 | ) | |
| (120,974 | ) |
| Interest
expense | |
| (83,025 | ) | |
| (6,753,231 | ) |
| Total
other income (expense), net | |
| 7,243,641 | | |
| 9,050,396 | |
| Income tax credit | |
| 1,088,729 | | |
| 1,720,000 | |
| | |
| | | |
| | |
| Net
loss | |
£ | (5,906,953 | ) | |
£ | (1,313,601 | ) |
| | |
| | | |
| | |
| Weighted-average ordinary shares outstanding,
basic and diluted | |
| 6,178,423 | | |
| 687,199 | |
| Basic and diluted net loss per share | |
£ | (0.96 | ) | |
£ | (1.91 | ) |
The
accompanying footnotes are an integral part of these consolidated financial statements.
TC
BIOPHARM (HOLDINGS) PLC
CONSOLIDATED
STATEMENTS OF CHANGES IN SHAREHOLDERS’ EQUITY (DEFICIT)
FOR THE YEARS ENDED DECEMBER 31, 2023 AND 2022
| | |
| | |
| | |
|
|
|
|
|
|
|
| | |
| | |
Total | |
| | |
Ordinary
shares | | |
Deferred shares |
|
Additional
Paid-in | | |
Accumulated | | |
Shareholders’ Equity | |
| | |
Shares | | |
Amount | | |
Shares |
|
|
Amount |
|
Capital | | |
Deficit | | |
(Deficit) | |
| | |
| | |
| | |
|
|
|
|
|
|
|
| | |
| | |
| |
| Balance, January 1, 2022 | |
| 390,952 | | |
£ | 195,476 | | |
|
- |
|
|
£ |
- |
|
£ | 16,710,757 | | |
£ | (33,465,282 | ) | |
£ | (16,559,049 | ) |
| Share-based compensation
expense | |
| - | | |
| - | | |
|
- |
|
|
|
- |
|
| - | | |
| 1,123,250 | | |
| 1,123,250 | |
| Issuance of ordinary shares,
net of issuance costs | |
| 559,006 | | |
| 202,017 | | |
|
- |
|
|
|
- |
|
| 16,597,811 | | |
| - | | |
| 16,799,828 | |
| Redesignation of nominal value to deferred
shares | |
| - | | |
| (397,398 | ) | |
|
794,955 |
|
|
|
397,398 |
|
| - | | |
| - | | |
| - | |
| Adjustments to convert from
IFRS to GAAP | |
| - | | |
| - | | |
|
- |
|
|
|
- |
|
| - | | |
| 280,837 | | |
| 280,837 | |
| Net
loss | |
| - | | |
| - | | |
|
- |
|
|
|
- |
|
| - | | |
| (1,313,601 | ) | |
| (1,313,601 | ) |
| Balance, December 31,
2022 | |
| 949,958 | | |
£ | 95 | | |
|
794,955 |
|
|
£ |
397,398 |
|
£ | 33,308,568 | | |
£ | (33,374,796 | ) | |
£ | 331,265 | |
| | |
| | |
| | |
|
|
|
|
|
|
|
Additional | | |
| | |
Total | |
| | |
Ordinary
shares | | |
Deferred shares |
|
Paid-in | | |
Accumulated | | |
Shareholders’ | |
| | |
Shares | | |
Amount | | |
Shares |
|
|
Amount |
|
Capital | | |
Deficit | | |
Equity | |
| | |
| | |
| | |
|
|
|
|
|
|
|
| | |
| | |
| |
| Balance, January 1, 2023 | |
| 949,958 | | |
£ | 95 | | |
|
794,955 |
|
|
£ |
397,398 |
|
£ | 33,308,568 | | |
£ | (33,374,796 | ) | |
£ |
331,265 | |
| Balance | |
| 949,958 | | |
£ | 95 | | |
|
794,955 |
|
|
£ |
397,398 |
|
£ | 33,308,568 | | |
£ | (33,374,796 | ) | |
£ |
331,265 | |
| Share-based compensation
expense | |
| - | | |
| - | | |
|
- |
|
|
|
- |
|
| - | | |
| 444,459 | | |
| 444,459 | |
| Issuance of ordinary shares,
net of issuance costs | |
| 19,620,130 | | |
| 1,962 | | |
|
- |
|
|
|
- |
|
| 5,231,151 | | |
| - | | |
| 5,233,113 | |
| Reclassification of warrants to equity | |
| - | | |
| - | | |
|
- |
|
|
|
- |
|
| 1,550,367 | | |
| - | | |
| 1,550,367 | |
| Issuance of warrants | |
| - | | |
| - | | |
|
- |
|
|
|
- |
|
| 1,032,979 | | |
| - | | |
| 1,032,979 | |
| Net
loss | |
| - | | |
| - | | |
|
- |
|
|
|
- |
|
| - | | |
| (5,906,953 | ) | |
| (5,906,953 | ) |
| Balance, December 31,
2023 | |
| 20,570,088 | | |
£ | 2,057 | | |
|
794,955 |
|
|
£ |
397,398 |
|
£ | 41,123,065 | | |
£ | (38,837,290 | ) | |
£ | 2,685,230 | |
| Balance | |
| 20,570,088 | | |
£ | 2,057 | | |
|
794,955 |
|
|
£ |
397,398 |
|
£ | 41,123,065 | | |
£ | (38,837,290 | ) | |
£ | 2,685,230 | |
The
accompanying footnotes are an integral part of these consolidated financial statements.
TC
BIOPHARM (HOLDINGS) PLC
CONSOLIDATED
STATEMENTS OF CASH FLOWS
| | |
For the Year
Ended | | |
For the Year
Ended | |
| | |
December
31, 2023 | | |
December
31, 2022 | |
| | |
| | |
| |
| CASH FLOWS FROM OPERATING
ACTIVITIES: | |
| | | |
| | |
| Net loss | |
£ | (5,906,953 | ) | |
£ | (1,313,601 | ) |
| Adjustments to reconcile net loss to net cash
used in operating activities: | |
| | | |
| | |
| Depreciation | |
| 596,726 | | |
| 714,550 | |
| Amortization of intangible
assets | |
| 36,282 | | |
| 55,046 | |
| Share-based compensation
expense | |
| 444,459 | | |
| 1,123,250 | |
| Loss on modification of
convertible loan | |
| 645,845 | | |
| 140,344 | |
| Change in fair value of derivative liability | |
| (8,052,581 | ) | |
| (16,064,945 | ) |
| Net foreign exchange loss | |
| 80,070 | | |
| 120,974 | |
| Noncash interest expense | |
| 71,568 | | |
| 6,628,406 | |
| Loss on disposal of property,
plant, and equipment, net | |
| - | | |
| 8,646 | |
| Changes in operating assets
and liabilities: | |
| | | |
| | |
| Change
in corporation tax receivable | |
| 676,407 | | |
| (312,801 | ) |
| Change
in prepaid expenses and other current assets | |
| (1,275,269 | ) | |
| (37,503 | ) |
| Change
in operating lease right of use assets | |
| 189,505 | | |
| 196,577 | |
| Change
in accounts payable and accrued liabilities | |
| 2,272,782 | | |
| (1,944,557 | ) |
| Change
in lease liabilities | |
| (323,711 | ) | |
| (762,150 | ) |
| Change
in deferred income | |
| - | | |
| (3,844,533 | ) |
| Net
cash used in operating activities | |
| (10,544,870 | ) | |
| (15,292,297 | ) |
| | |
| | | |
| | |
| CASH FLOWS FROM INVESTING
ACTIVITIES: | |
| | | |
| | |
| Purchase of property, plant,
and equipment | |
| (110,353 | ) | |
| (240,712 | ) |
| Disposal of property, plant,
and equipment | |
| - | | |
| 55,000 | |
| Purchase
of intangible assets | |
| (98,436 | ) | |
| (124,485 | ) |
| Net
cash used in investing activities | |
| (208,789 | ) | |
| (310,197 | ) |
| | |
| | | |
| | |
| CASH FLOWS FROM FINANCING
ACTIVITIES: | |
| | | |
| | |
| Proceeds from issuance
of convertible loan, net of issuance costs | |
| - | | |
| 18,110 | |
| Repayment of convertible
loan | |
| - | | |
| (2,632,324 | ) |
| Proceeds from sale of warrants | |
| 5,127,260 | | |
| 18,806,153 | |
| Issuance of ordinary shares | |
| 4,186,862 | | |
| 3,369,877 | |
| Ordinary
shares and warrants issuance costs | |
| (793,746 | ) | |
| (908,602 | ) |
| Net
cash provided by financing activities | |
| 8,520,376 | | |
| 18,653,214 | |
| | |
| | | |
| | |
| Foreign exchange (loss)
gain on cash and cash equivalents | |
| (112,168 | ) | |
| 190,652 | |
| | |
| | | |
| | |
| NET CHANGE IN CASH | |
| (2,345,451 | ) | |
| 3,241,372 | |
| Cash - Beginning of
period | |
| 4,808,060 | | |
| 1,566,688 | |
| Cash - End of period | |
£ | 2,462,609 | | |
£ | 4,808,060 | |
The
accompanying footnotes are an integral part of these consolidated financial statements.
TC
BIOPHARM (HOLDINGS) PLC
NOTES
TO CONSOLIDATED FINANCIAL STATEMENTS
NOTE
1 – NATURE AND DESCRIPTION OF BUSINESS
TC
BioPharm (Holdings) plc (“TC BioPharm” or the “Company”) was incorporated on October 25, 2021 as a Public limited
company, limited by shares, in Scotland and domiciled in the United Kingdom and has the following wholly owned subsidiaries: TC BioPharm
Limited, TC BioPharm (North America) Inc. and TC BioPharm BV (together the “Group” and “Company”).
The
principal activity of the Company is as a clinical stage immuno-therapy company pioneering commercialization of allogeneic, ‘off-the-shelf’
gamma-delta T cell (‘GD-T’) therapies, ranging from unmodified GD-T therapies to treat haematological cancers and viral infections,
to sophisticated proprietary GD-T CAR-T products designed to reach and treat solid tumors.
The
Company has historically been classified as a foreign private issuer (“FPI”). However, as of June 30, 2023, the Company determined
that, pursuant to the definition provided in Rule 405 of the Securities Act of 1933, it no longer satisfied the criteria to be considered
an FPI. As such, beginning on January 1, 2024, the Company was required to begin utilizing the SEC’s domestic reporting forms.
A reconciliation from International Financial Reporting Standards (“IFRS”) to accounting principles generally accepted in
the United States (“U.S. GAAP”) has been presented (see Note 3).
On December 15, 2023, the Company changed its ratio of American Depositary Shares (“ADSs”) ordinary shares from one ADS representing
one ordinary share to one ADS representing 20 ordinary shares (the “ADS Ratio Change”). As a result of the ratio change, all
references in these consolidated financial statements and accompanying notes to units of ordinary shares underlying ADSs are reflective
of the ratio change for all periods presented. In addition, the exercise prices and the numbers of ordinary shares issuable upon the exercise
of any outstanding options to purchase ordinary shares were proportionally adjusted pursuant to the respective anti-dilution terms of
the share-based payment plans.
The
Company’s ADSs began trading on the Nasdaq Capital Market (“Nasdaq”) under
the ticker symbol “TCBP” on February 10, 2022, following its initial public offering (“IPO”). As part of the
IPO, the Company, issued 4,117 American Depositary Shares (“ADSs”) representing 82,353 ordinary shares with nominal value
of £41,176 and warrants to buy 9,470 ADSs for proceeds before expenses of $17.5 million. Funding costs of $3.0 million including
underwriter fees were incurred. On February 10, 2022, the Company issued 3,164 ADSs representing 63,280 ordinary shares with nominal
value of £31,640 and warrants to buy 6,278 ADSs on conversion of loan notes totaling $13.4 million. Between June 7, 2022 and
June 8, 2022, the Company issued and sold 11,500 ADSs representing 230,000 ordinary shares generating proceeds of $4.6 million before
deductions for offering expenses of approximately $0.8 million (£0.6 million).
On
November 18, 2022 the Company undertook a reverse share split such that fifty issued ordinary shares were exchanged for one new ordinary
share. As a result of the share split, all references in these consolidated financial statements and accompanying notes to units of ordinary
shares or per share amounts are reflective of the reverse share split for all periods presented. In addition, the exercise prices and
the numbers of ordinary shares issuable upon the exercise of any outstanding options to purchase ordinary shares were proportionally
adjusted pursuant to the respective anti-dilution terms of the share-based payment plans.
On
November 27, 2022, the Company entered into a Securities Purchase Agreement (the “First Purchase Agreement”) with
certain accredited investors (the “Investors”) as purchasers. Pursuant to the First Purchase Agreement, the Company
sold, and the Investors purchased in a private placement an aggregate of 7,750
ADSs, pre-funded warrants to purchase up to 65,750
ADS (the “Pre-Funded Warrants”), Series A purchase warrants to purchase up to 73,500
ADSs (the “Series A Ordinary Warrants”) and Series B purchase warrants to purchase up to 73,500
ADSs (the “Series B Ordinary Warrants” and together with the Series A Ordinary Warrants, the “Ordinary
Warrants”) for aggregate gross proceeds of $7,350,000
(£6,073,376),
excluding any proceeds that may be received upon exercise of the Ordinary Warrants. The purchase price for each ADS and associated
Ordinary Warrants was $100
(on a post-split basis) and the purchase price per each Pre-Funded Warrant and associated Ordinary Warrants was $99.98
(on a post-split basis).
On
March 27, 2023, the Company, entered into a Second Securities Purchase Agreement (the “Second Purchase Agreement”) with Investors,
pursuant to which the Company agreed to issue and sell an aggregate of 10,750
ADSs, pre-funded warrants to purchase up to 161,125
ADS (the “Pre-Funded Warrants”),
and Series C purchase warrants to purchase up to 171,875
ADSs (the “Ordinary Warrants” and
together with the Pre-Funded Warrants and the ADSs, the “Securities”). In addition, the Company also issued placement agent
warrants to purchase 12,891 ADSs. The purchase price for each ADS and associated Ordinary Warrants was $32
(on a post-split basis) and the purchase price
per each Pre-Funded Warrant and associated Ordinary Warrants was $31.98.
The Ordinary Warrants were immediately exercisable, expire five (5) years from the date of issuance and the Pre-Funded Warrants may be
exercised at any time until all of the Pre-Funded Warrants are exercised in full. The total net proceeds from this offering were approximately
$4.9
million, after deducting estimated offering expenses
of approximately $0.6
million.
On
March 27, 2023 the Company also agreed that certain existing warrants to purchase up to an aggregate of 140,000
ADSs of the Company that were previously issued on November 30, 2022, at an exercise price of $100
(on a post-split basis) per ADS and expiration dates of May 30, 2025 and May 30, 2028, were amended so that the
amended warrants had a reduced exercise price of $35
(on a post-split basis) per ADS.
On August 30, 2023, the Company entered into an agreement with its Series A and B warrant holders whereby it induced
70,000 and 70,000 of the outstanding warrants, respectively. In addition, the Company also entered into an agreement with its Series C
warrant holders to induce all of the outstanding warrants (171,875). The inducement resulted in gross proceeds to the Company of approximately
$2.8 million. In order to incentivize the inducement, the Company issued 623,750 Series D warrants to the Series, A, B and C warrant holders.
In addition, the Company also issued placement agent warrants to purchase 23,391 ADSs. The
Company received aggregate gross proceeds of approximately £2.2
million (approximately $2.8
million) from the exercise of the Existing Warrants
by the Holders, before deducting placement agent fees payable by the Company.
On
December 18, 2023, the Company entered into a Third Securities Purchase Agreement (the “Third Purchase Agreement”) with a
certain institutional investor (the “Investor”) pursuant to which the Company agreed to issue and sell to the Investor in
a best-efforts public offering 75,000 ADSs representing 1,500,000 ordinary shares, pre-funded warrants to purchase up to 1,675,000 ADS
representing 33,500,000 Ordinary Shares (the “Pre-Funded Warrants”), and series E purchase warrants to purchase up to 1,750,000
ADSs representing 35,000,000 Ordinary Shares (the “Warrants” and together with the Pre-Funded Warrants and the ADSs, the
“Securities”). The purchase price for each ADS and associated Warrant was $2.00 and the purchase price per each Pre-Funded
Warrant and associated Warrant was $1.999. The Warrants are immediately exercisable, will expire five years from the date of issuance
and have an exercise price of £1.5814. The Pre-Funded Warrants may be exercised at any time until all of the Pre-Funded Warrants
are exercised in full at an exercise price of $0.001 per ADS. Additionally, the Company agreed that a certain number of existing warrants
to purchase up to an aggregate of 623,750 ADSs of the Company that was previously issued on September 5, 2023, at an exercise price of
£7.00 per ADS and an expiration date of March 5, 2029, were amended effective upon the closing of the offering so that the amended
warrant will have a reduced exercise price of £1.5814 (or $2.00, as translated for illustration to U.S. dollars at the rate of
£1.00 to $1.264 as of December 18, 2023) per ADS. The Securities Purchase Agreements resulted in gross proceeds of $3.5 million.
Risks
and Uncertainties
The
Company is exposed to a variety of risks in the ordinary course of business, including, but not limited to, currency risk, liquidity
risk, equity price risk and credit risk. The Company regularly assesses each of these risks to minimize any adverse effects on the business
as a result of those factors.
Going
Concern
The
Company has been focused on the development of therapeutic products based around its gamma delta T cell platform technology, with the
objective of conducting clinical trials to demonstrate safety and efficacy and eventually being granted regulatory approval to market
and sell its products since its incorporation. This activity was expected to be in development for several years and has incurred considerable
expenditures to date in research and development expenses and in conducting clinical trials. Similar to most development and/or clinical
stage biotechnology companies, the Company has not yet generated any revenues from sales of products, but has obtained cash to finance
its research, development and clinical trial activities from equity, debt and grant financings and from receipts from partners under
collaborative co-development agreements. The Company is expected to continue in this clinical development phase for a number of years
before any product becomes marketable. The Company therefore expects to continue to incur significant losses in the foreseeable future.
As
of December 31, 2023, the Company’s cash and cash equivalents amounted to £2.5 million. As of December 31, 2023, the Company
had working capital of £1.0 million. Cash used in operating activities for the year ended December 31, 2023 was £10.5 million,
and the Company expects to incur continued outflow of cash for the foreseeable future. Net loss for the year ended December 31, 2023
was £5.9 million.
On March 22,
2024, the Group had cash on hand of $1.7 million (£1.4 million), which will not be sufficient to enable the Group to meet the cash
requirements required to enable it to conduct its business plan through the going concern period (being to April 1, 2025)
(“Going Concern Period”). With existing resources, we expect to be able to fund current operations to May 2024.
Similar
to many clinical development stage biotechnology companies, the Company’s future liquidity needs, and ability to address them,
will largely be determined by the availability of capital, both generally and in particular to fund product candidates and key development
and regulatory projects. As a pre-revenue biotechnology Company, operations have been financed though continuously raising capital, and
management expects to continue to raise capital routinely. The Company is currently and continuously progressing various funding options
to fill the projected working capital gap, which could be in the form of an equity raise or other forms of financings such as debt funding,
collaborations or licensing arrangements. Management believes that the ongoing financing initiatives should provide sufficient capital
to finance planned operations through 2024, and thereafter we would expect to be in a position to raise significantly greater capital
as the clinical program progresses. However, there can be no certainty that these initiatives will be successful and, if they are not,
management will seek to deploy alternative plans, which could have a potentially significant negative impact on stockholder and asset
value. Such plans could include all or any of the following: raising additional capital through low priced and/or complex equity and/or
debt financings, entering transactions involving sales, joint venturing or licensing of intellectual property, reducing and/or deferring
discretionary spending on research and development or clinical programs, restructuring our operating model to take advantage of our manufacturing
capability to generate short term revenues or reducing our cash burn rate through reduction in planned operating costs.
The
accompanying financial statements have been prepared on the basis that the Company will continue as a going concern, which assumes the
realization of assets and the satisfaction of liabilities in the normal course of business. The Company has not established a source
of revenues sufficient to cover its operating costs, and as such, has been dependent on ongoing funding operations primarily through
ongoing initiatives to raise capital. The Company expects to require substantially more capital to fund its clinical, development and
operational requirements, and therefore incur further losses over the next several years as it develops its clinical products. The Company
has utilized, and expects to continue to utilize, substantial amounts of funding to implement its business strategy. If the Company is
unable to maintain adequate liquidity, future operations will need to be scaled back or discontinued. Based on these circumstances, management
has determined that there is substantial doubt about the Company’s ability to continue as a going concern. These consolidated financial
statements do not include any adjustments that might result from the outcome of this uncertainty.
NOTE
2 – SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES AND NEW ACCOUNTING STANDARDS
Basis
of Presentation
The
accompanying consolidated financial statements have been prepared and presented in accordance with U.S. GAAP and pursuant to the rules
and regulations of the Securities and Exchange Commission (“SEC”). In the opinion of management, these consolidated financial
statements include all adjustments necessary for a fair statement of the financial position, results of operations and cash flows of
the Company, and the adjustments are of a normal and recurring nature.
Principals
of Consolidation
The
consolidated financial statements include the accounts of TC BioPharm and its 100% controlled subsidiaries, TC BioPharm Limited, TC BioPharm
Inc. and TC BioPharm BV. All significant intercompany balances and transactions have been eliminated. “TC BioPharm”, the
“Company”, “we”, “our” or “us” is intended to mean TC BioPharm (Holdings) plc, including
the subsidiaries indicated above, unless otherwise indicated.
Emerging
Growth Company
The
Company is an “emerging growth company,” as defined in Section 2(a) of the Securities Act of 1933, as amended (the “Securities
Act”), as modified by the Jumpstart Our Business Startups Act of 2012 (the “JOBS Act”), and it may take advantage of
certain exemptions from various reporting requirements that are applicable to other public companies that are not emerging growth companies
including, but not limited to, not being required to comply with the auditor attestation requirements of Section 404(b) of the Sarbanes-Oxley
Act of 2002, reduced disclosure obligations regarding executive compensation in its periodic reports and proxy statements, and exemptions
from the requirements of holding a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute
payments not previously approved. Further, Section 102(b)(1) of the JOBS Act exempts emerging growth companies from being required to
comply with new or revised financial accounting standards until private companies (that is, those that have not had a Securities Act
registration statement declared effective or do not have a class of securities registered under the Securities Exchange Act of 1934,
as amended (the “Exchange Act”)) are required to comply with the new or revised financial accounting standards. The JOBS
Act provides that a company can elect to opt out of the extended transition period and comply with the requirements that apply to non-emerging
growth companies but any such election to opt out is irrevocable. The Company has elected not to opt out of such extended transition
period which means that when a standard is issued or revised and it has different application dates for public or private companies,
the Company, as an emerging growth company, can adopt the new or revised standard at the time private companies adopt the new or revised
standard. This may make comparison of the Company’s consolidated financial statements with another public company which is neither
an emerging growth company nor an emerging growth company which has opted out of using the extended transition period difficult or impossible
because of the potential differences in accounting standards used.
Use
of Estimates
The
preparation of these consolidated financial statements in conformity with U.S. GAAP requires the Company’s management to make estimates
and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the
date of the financial statements and the reported amounts of revenues and expenses during the reporting period.
Making
estimates requires management to exercise significant judgment. It is at least reasonably possible that the estimate of the effect of
a condition, situation or set of circumstances that existed at the date of the financial statements, which management considered in formulating
its estimate, could change in the near term due to one or more future confirming events. Accordingly, the actual results could differ
significantly from those estimates.
Segment
Reporting
The
Company operates in one operating segment. Operating segments are reported in a manner consistent with the internal reporting provided
to the Company’s chief operating decision maker (“the CODM”). The Company’s CODM, its Chief Executive Officer,
views the Company’s operations and manages its business as a single operating segment, which is the business of a clinical stage
immune-therapy Group pioneering commercialization of allogeneic, ‘off-the-shelf’ gamma-delta T cell (‘GD-T’)
therapies.
Income
and Other Taxes
Income
taxes are accounted for using the asset and liability method in accordance with Financial Accounting Standards Board (“FASB”)
Accounting Standard Codification (“ASC”) 740, Income Taxes (“ASC 740”), which requires the recognition
of deferred tax assets and liabilities for the expected future tax consequences of events that have been included in the financial statements.
Under this method, deferred tax assets and liabilities are determined based on the differences between the financial statements and tax
basis of assets and liabilities using enacted tax rates in effect for the year in which the differences are expected to reverse. The
effect of a change in tax rates on deferred tax assets and liabilities is recognized in income in the period that includes the enactment
date.
The
Company records net deferred tax assets to the extent they believe these assets will more-likely-than-not be realized. In making such
determination, the Company considers all available positive and negative evidence, including future reversals of existing taxable temporary
differences, projected future taxable income, tax planning strategies and recent financial operations. In the event the Company was to
determine that it would be able to realize its deferred income tax assets in the future in excess of its net recorded amount, the Company
would make an adjustment to the valuation allowance which would reduce the provision for income taxes.
Income
Tax Credit
The
Company carries out extensive research and development activities, where it benefits from the United Kingdom’s research and development
tax relief and expenditure credit regimes. The Company is able to surrender some of its income tax losses for a cash rebate of up to
33.35% of expenditures related to eligible research and development projects. Such credits are accounted for, depending on the appropriate
tax relief, either within the tax provision or other income, in the year in which the expenditures were incurred.
Cash
and cash equivalents
The
Company defines cash and cash equivalents as cash on hand, deposits held on call with banks and other short-term liquid investments with
maturities of three months or less. As of December 31, 2023 and 2022, cash and cash equivalents was £2.5 million and £4.8
million, respectively.
Concentration
of Risk
Financial
instruments that subject the Company to significant concentrations of credit risk primarily consist of cash and cash equivalents. The
Company maintains substantially all of its cash and cash equivalents with financial institutions, which, at times, may exceed federally
insured limits. The Company has not incurred any losses associated with this concentration of deposits.
The
Company currently has bank deposits with financial institutions in the U.S. of approximately £0.2 million as of December 31, 2023
which are below the FDIC insurance limits. FDIC insurance provides protection for bank deposits up to $250,000.
The Company had approximately £2.3
million
in uninsured bank deposits with financial institutions outside the U.S. All uninsured bank deposits are held at high quality credit institutions.
Foreign
currency translation and transactions
The
Company uses the British pound sterling as the reporting currency for its financial statements. Functional currency is the currency
of the primary economic environment in which an entity operates. The functional currency of the Company’s subsidiaries are the
local currencies. The Company has transactions denominated in various currencies, with the principal currency exposure being fluctuations
in U.S. Dollars and Euros against pound sterling. The Company’s exposure to the risk of changes in foreign exchange rates relates
primarily to the a limited number of supplier agreements denominated in currencies other than pound sterling.
Property,
Plant and Equipment
Property
and equipment consist of computer equipment, facility, and scientific equipment and office equipment, which are stated at cost, net of
accumulated depreciation and amortization, and depreciated over their estimated lives using the straight-line method.
Depreciation
is provided for by the straight-line method over the estimated useful lives as follows:
SCHEDULE
OF ESTIMATED USEFUL LIVES
| Property
and Equipment | |
Estimated
Useful Life |
| Facility and scientific equipment | |
4-10 years |
| Computer equipment | |
3 years |
| Office equipment | |
5 years |
Expenditures
for repairs and maintenance are expensed as incurred. When assets have been retired or sold, the cost and related accumulated depreciation
are removed from the accounts and any resulting gain or loss is recognized in the results of operations.
Intangible
assets
Intangible
assets consist of software, patents and licenses. Intangible assets are recognized where it is probable that there will be a future economic
benefit and that this can be reliably measured. Software represents the historical cost of installation of third-party software used
within the Company to maintain and control the Company’s quality system. The software is hosted and controlled on the Company’s
servers and can be used independently of the related hardware. Software is amortized, on a straight-line basis, over the life of the
relevant license of three to four years. Patent costs represent the costs of securing patents in relation to the Company’s intellectual
property. Patent costs are amortized, on a straight-line basis, over the remaining legal life of the relevant patents, which has an average
estimated patent life of 16 years. License costs represent costs incurred for securing use of third-party technology. License costs are
amortized, on a straight-line basis, over the life of the relevant license of three years. Amortization methods and useful lives are
reviewed at each reporting date and adjusted as appropriate.
The
Company reviews the carrying amounts of its tangible and intangible assets where there is any indication that those assets have suffered
an impairment loss. If any such indication exists, the recoverable amount of the asset is estimated in order to determine the extent
of the impairment loss (if any). The recoverable amount is determined for an individual asset, unless the asset does not generate cash
inflows that are largely independent of those from other assets or groups of assets in which case the Company estimates the recoverable
amount of the cash-generating unit to which the asset belongs. Recoverable amount is the higher of fair value less costs to sell and
value-in-use. In assessing value-in-use, the estimated future cash flows are discounted to their present value using a pre-tax discount
rate that reflects current market assessments of the time value of money and the risks specific to the asset for which the estimates
of future cash flows have not been adjusted. If the recoverable amount of an asset (or cash-generating unit) is estimated to be less
than its carrying amount, the carrying amount of the asset (or cash-generating unit) is reduced to its recoverable amount. An impairment
loss is recognized immediately in the statement of operations. There was no impairment of tangible or intangible assets during the years
ended December 31, 2023 and 2022.
Fair
Value Measurements
The
fair value of the Company’s assets and liabilities which qualify as financial instruments under ASC 820, Fair Value Measurement,
approximates the carrying amounts represented in the accompanying consolidated balance sheets.
Net
Loss per Share
Basic
net loss per share ordinary share is calculated based on the weighted-average number of ordinary shares outstanding in accordance with
ASC Topic 260, Earnings per Share. Diluted net loss per share is calculated based on the weighted-average number of ordinary shares
outstanding plus the effect of dilutive potential ordinary shares. When the Company reports a net loss, the calculation of diluted net
loss per share excludes potential ordinary shares as the effect would be anti-dilutive. Potential ordinary shares are composed of ordinary
shares issuable upon the exercise of options and warrants. The following table shows the basic and diluted loss per share for the years
ended December 31, 2023 and 2022:
SCHEDULE
OF BASIC AND DILUTED LOSS PER SHARE
| | |
Year
Ended December
31,
2023 | | |
Year
Ended December
31,
2022 | |
| Net loss | |
| (5,906,953 | ) | |
| (1,313,601 | ) |
| Basic and diluted weighted average number of shares
outstanding(1) | |
| 6,178,423 | | |
| 687,199 | |
| | |
| | | |
| | |
| Basic
and diluted loss per share | |
| (0.96 | ) | |
| (1.91 | ) |
Share-Based
Compensation
The
Company accounts for share-based compensation arrangements with employees, directors, and consultants and recognizes the compensation
expense for share-based awards based on the estimated fair value of the awards on the date of grant. Compensation expense for all share-based
awards is based on the estimated grant-date fair value and recognized in earnings over the requisite service period (generally the vesting
period).
Revenue
Recognition
Revenue
is recognized when a customer obtains control of promised goods or services, in an amount that reflects the consideration which the entity
expects to receive in exchange for those goods or services. The Company accounts for revenue contracts with customers by applying the
requirements of ASC 606, Revenue from Contracts with Customers, which includes the following five steps:
| |
i. |
Identification
of the contract with a customer. |
| |
ii. |
Identification
of the performance obligations in the contract. |
| |
iii. |
Determination
of the transaction price. |
| |
iv. |
Allocation
of the transaction price to the performance obligations in the contract. |
| |
v. |
Recognition
of revenue as the entity satisfies a performance obligation. |
The
Company earns revenue from collaboration agreements and contracts with customers (see Note 4). Revenue is recognized on upfront collaboration
payments on a straight-line basis over the estimated term over which the services promised will be provided. The Company is entitled
to receive contractual milestone payments on achievement of certain performance obligations and these are recognized when the milestones
are certain to occur.
Research
& Development Expenses
Research
expenditure is expensed in the year in which it is incurred. Identifiable development expenditure is capitalized to the extent that the
technical, commercial and financial feasibility can be demonstrated. The Company has not capitalized any development expenditures since
inception.
Commitments
and Contingencies
The
Company accounts for contingencies in accordance with ASC 450-20, Contingencies. Certain conditions may exist as of the date the
financial statements are issued, which may result in a loss to the Company, but which will only be resolved when one or more future events
occur or fail to occur. The Company assesses such contingent liabilities, and such assessment inherently involves an exercise of judgment.
In assessing loss contingencies related to legal proceedings that are pending against the Company or un-asserted claims that may result
in such proceedings, the Company evaluates the perceived merits of any legal proceedings or un-asserted claims as well as the perceived
merits of the amount of relief sought or expected to be sought therein.
If
the assessment of a contingency indicates that it is probable that a material loss has been incurred and the amount of the liability
can be estimated, then the estimated liability would be accrued in the Company’s consolidated financial statements. If the assessment
indicates that a potentially material loss contingency is not probable but is reasonably possible, or is probable but cannot be estimated,
then the nature of the contingent liability, and an estimate of the range of possible losses, if determinable and material, would be
disclosed.
Loss
contingencies considered remote are generally not disclosed unless they involve guarantees, in which case the guarantees would be disclosed.
Further details are included within Note 9 to the financial statements.
Recent
Accounting Pronouncements
The
Company has implemented all new accounting pronouncements that are in effect and that may impact its consolidated financial statements.
Further, during December 2023, the FASB issued Accounting Standards Update (“ASU”) 2023-09-Income Taxes (Topic 740)-Improvements to Income Tax Disclosures, which requires
entities to provide additional information in the rate reconciliation and additional disclosures about income taxes paid. The guidance
should be applied prospectively and is effective for annual periods beginning after December 15, 2024. The Company does not expect the
issued standard to have a material impact on its financial statements or results of operations.
NOTE
3. CONVERSION FROM IFRS TO GAAP
The
Company has retrospectively converted its consolidated financial statements from IFRS to GAAP. Refer to Note
1 for additional details.
The
significant differences between IFRS and GAAP as they relate to these financial statements are as follows:
(a)
Leases
Under
IFRS 16, the Company, as lessee, applied the single lease model that is similar to the accounting for a finance lease under GAAP. The
expense recognition presented a higher portion of the total expense earlier in the term as a combination of straight-line depreciation
of the right-of-use asset and the effective interest rate method applied to the lease liability which resulted in a decreasing rate of
interest expense recognition throughout the lease term.
Under
GAAP, there is dual classification lease accounting model for lessees: finance leases and operating leases. Specifically, in accordance
with ASC 842, the Company, as lessee, must determine whether a lease is a finance or operating lease at the inception of the contract.
Further, operating leases are recognized utilizing a single lease expense model, which takes the expected payments to be made over the
life of the lease and recognizes those payments on a straight-line basis throughout the lease term.
As
a result of the cutover from IFRS to GAAP, the Company began accounting for its office lease space as an operating lease (in lieu of
the “finance lease” model under IFRS 16). This resulted in the corresponding lease expense being reclassified from
interest expense and ROU amortization into operating lease expense, which decreased interest expense by
£238,985
for the year ended December 31, 2022. Additionally due to the change in the expense recognition method, the total resulting decrease
in lease related expense from the transition from IFRS to GAAP was £76,105
for the year ended December 31, 2022. The expense for 2023 was recognized under GAAP and did not require any adjustments.
The
following table summarizes the changes for the year ended December 31, 2022:
SCHEDULE
OF LEASE RELATED EXPENSE FROM THE TRANSITION FROM IFRS TO GAAP
| | |
IFRS | | |
Adjustments | | |
GAAP | |
| Research and development expenses | |
£ | 7,447,506 | | |
£ | 144,964 | | |
£ | 7,592,470 | |
| Administrative expenses | |
| 7,013,056 | | |
| 17,916 | | |
| 7,030,972 | |
| Interest expense | |
| (6,994,423 | ) | |
| (238,985 | ) | |
| (6,755,438 | ) |
| Total Conversion Adjustments | |
| | | |
| (76,105 | ) | |
| | |
| | |
| | | |
| | | |
| | |
| Net loss | |
£ | (1,389,706 | ) | |
£ | (76,105 | ) | |
£ | (1,313,601 | ) |
| Weighted-average common shares outstanding,
basic and diluted | |
| 687,199 | | |
| - | | |
| 687,199 | |
| Basic and diluted net loss per share | |
£ | (2.02 | ) | |
£ | 0.08 | | |
£ | (1.91 | ) |
The
following table summarizes the changes as of December 31, 2022:
| | |
IFRS | | |
Adjustments | | |
GAAP | |
| ASSETS | |
| | | |
| | | |
| | |
| Current assets: | |
| | | |
| | | |
| | |
| Operating
lease right of use assets | |
£ | 1,188,947 | | |
£ | 341,327 | | |
£ | 1,530,274 | |
| Total Conversion Adjustments | |
| | | |
£ | 341,327 | | |
| | |
| | |
| | | |
| | | |
| | |
| LIABILITIES AND STOCKHOLDERS’
(DEFICIT) | |
| | | |
| | | |
| | |
| Non-current operating lease liability | |
£ | 1,812,450 | | |
£ | (15,615 | ) | |
£ | 1,796,835 | |
| Accumulated deficit | |
| (33,731,738 | ) | |
| 356,942 | | |
| (33,374,796 | ) |
| Total Conversion Adjustments | |
| | | |
£ | 341,327 | | |
| | |
NOTE
4. REVENUE
The
Company earned revenue from collaboration agreements and contracts with customers. Revenue is recognized on upfront collaboration payments
on a straight-line basis over the estimated term over which the services promised will be provided. Collaboration agreement revenue for
the years ended December 31, 2023 and 2022 is as follows:
SCHEDULE
OF REVENUE FROM COLLABORATION AGREEMENT AND CONTRACT WITH CUSTOMERS
| |
|
Year
Ended
December
31, 2023 |
|
|
Year
Ended
December
31, 2022 |
|
| Revenue
from collaboration agreements | |
£ | — | | |
£ | 3,844,532 | |
The
terms of business for payment on satisfaction of a performance obligation are typically 30-60 days. Collaboration agreements entered
into by the Company provide for the entity to work with a partner to carry out collaborative research and development work. Performance
obligations around upfront payments are deemed to be satisfied over the estimated life of the services promised to be provided. This
estimated life of the services was estimated by management at the inception of each contract and evaluated at each reporting date. During
2022, the contract with the collaboration partner was terminated. As such, the Company re-classed any remaining deferred revenue (from
the upfront payment) to revenue as 1) no additional goods or services were required to be provided to the collaboration partner and 2)
the upfront payment was non-refundable.
Revenue
from reimbursement of research and development costs by collaboration partners is recognized as the costs are incurred.
NOTE
5. PROPERTY, PLANT AND EQUIPMENT
The
Company’s property, plant and equipment balances consist of the following:
SCHEDULE OF PROPERTY PLANT AND EQUIPMENT BALANCES
| | |
Facilities
& Scientific Equipment | | |
Computer
Equipment | | |
Office
Equipment | | |
Total | |
| Cost | |
| | | |
| | | |
| | | |
| | |
| At January 1, 2022 | |
£ | 4,889,662 | | |
£ | 333,148 | | |
£ | 86,331 | | | £ | 5,309,141 | |
| Additions | |
| 225,666 | | |
| 15,046 | | |
| - | | |
| 240,712 | |
| Disposals | |
| (228,026 | ) | |
| - | | |
| - | | |
| (228,026 | ) |
| At December 31, 2022 | |
| 4,887,302 | | |
| 348,194 | | |
| 86,331 | | |
| 5,321,827 | |
| Additions | |
| 91,724 | | |
| 18,629 | | |
| - | | |
| 110,353 | |
| Disposals | |
| - | | |
| - | | |
| - | | |
| - | |
| At December 31, 2023 | |
£ | 4,979,026 | | |
£ | 366,823 | | |
£ | 86,331 | | |
£ | 5,432,180 | |
| | |
| | | |
| | | |
| | | |
| | |
| Depreciation | |
| | | |
| | | |
| | | |
| | |
| At January 1, 2022 | |
£ | 2,636,772 | | |
£ | 312,718 | | |
£ | 60,996 | | |
£ | 3,010,486 | |
| Disposals | |
| (164,380 | ) | |
| - | | |
| - | | |
| (164,380 | ) |
| Depreciation expense | |
| 683,592 | | |
| 15,098 | | |
| 15,860 | | |
| 714,550 | |
| At December 31, 2022 | |
| 3,155,984 | | |
| 327,816 | | |
| 76,856 | | |
| 3,560,656 | |
| Depreciation expense | |
| 575,233 | | |
| 13,947 | | |
| 7,546 | | |
| 596,726 | |
| At December 31, 2023 | |
£ | 3,731,217 | | |
£ | 341,763 | | |
£ | 84,402 | | |
£ | 4,157,382 | |
| | |
| | | |
| | | |
| | | |
| | |
| Net book value | |
| | | |
| | | |
| | | |
| | |
| At December 31, 2023 | |
£ | 1,247,809 | | |
£ | 25,060 | | |
£ | 1,929 | | |
£ | 1,274,798 | |
| | |
| | | |
| | | |
| | | |
| | |
| At December 31, 2022 | |
£ | 1,731,318 | | |
£ | 20,378 | | |
£ | 9,475 | | |
£ | 1,761,171 | |
Depreciation
expense on these assets for the years ended December 31, 2023 and 2022, was £596,726 and £714,550, respectively, and is included
in research and development and administrative expenses in the accompanying consolidated statements of operations. The Company incurred
a loss on disposal of equipment totaling £8,646 for the year ended December 31, 2022.
NOTE
6. INTANGIBLE ASSETS
The
Company’s intangible assets consist of the following:
SCHEDULE
OF INTANGIBLE ASSETS
| | |
Software | | |
Patents
and Licenses | | |
Total | |
| Cost | |
| | | |
| | | |
| | |
| At January 1, 2022 | |
£ | 49,613 | | |
£ | 588,072 | | |
£ | 637,685 | |
| Additions | |
| - | | |
| 124,485 | | |
| 124,485 | |
| At December 31, 2022 | |
| 49,613 | | |
| 712,557 | | |
| 762,170 | |
| Intangible assets, Beginning | |
| 49,613 | | |
| 712,557 | | |
| 762,170 | |
| Additions | |
| - | | |
| 98,436 | | |
| 98,436 | |
| At December 31, 2023 | |
| 49,613 | | |
| 810,993 | | |
| 860,606 | |
| Intangible assets, Ending | |
| 49,613 | | |
| 810,993 | | |
| 860,606 | |
| | |
| | | |
| | | |
| | |
| Amortization | |
| | | |
| | | |
| | |
| At January 1, 2022 | |
| 39,536 | | |
| 114,572 | | |
| 154,108 | |
| Amortization expense | |
| 7,125 | | |
| 47,921 | | |
| 55,046 | |
| At December 31, 2022 | |
| 46,661 | | |
| 162,493 | | |
| 209,154 | |
| Amortization expense | |
| 2,952 | | |
| 33,330 | | |
| 36,282 | |
| At December 31, 2023 | |
| 49,613 | | |
| 195,823 | | |
| 245,436 | |
| | |
| | | |
| | | |
| | |
| Net book value | |
| | | |
| | | |
| | |
| At December 31, 2023 | |
£ | - | | |
£ | 615,170 | | |
£ | 615,170 | |
| | |
| | | |
| | | |
| | |
| At December 31, 2022 | |
£ | 2,952 | | |
£ | 550,064 | | |
£ | 553,016 | |
Amortization
expense on these assets for the years ended December 31, 2023 and 2022, was £36,282 and £55,046, respectively, and is included
in research and development expenses in the accompanying consolidated statements of operations.
NOTE
7. PREPAID EXPENSES AND OTHER CURRENT ASSETS
The
Company’s prepaid expenses and other current assets consist of the following:
SCHEDULE
OF PREPAID EXPENSES AND OTHER CURRENT ASSETS
| | |
December
31, 2023 | | |
December
31, 2022 | |
| Other receivables | |
£ | 51,584 | | |
£ | 56,264 | |
| VAT owed to the Company | |
| 135,642 | | |
| 27,055 | |
| Prepaid clinical trial costs | |
| 307,519 | | |
| 307,519 | |
| Deferred clinical trial testing costs | |
| 1,177,500 | | |
| - | |
| Other prepayments | |
| 522,480 | | |
| 528,618 | |
| Total | |
£ | 2,194,725 | | |
£ | 919,456 | |
The
fair value of trade and other receivables are not materially different to the book value.
NOTE
8. ACCOUNTS PAYABLE AND ACCRUED LIABILITIES
The
Company’s accounts payable and accrued liabilities consist of the following:
SCHEDULE
OF ACCOUNTS PAYABLE AND ACCRUED LIABILITIES
| | |
December
31, 2023 | | |
December
31, 2022 | |
| Trade payables | |
£ | 1,847,279 | | |
£ | 882,364 | |
| Other tax and social security | |
| 139,029 | | |
| 293,467 | |
| Accruals | |
| 1,229,419 | | |
| 944,904 | |
| Amounts accrued in respect to clinical trial testing | |
| 1,177,500 | | |
| - | |
| Other payables | |
| 38,613 | | |
| 38,323 | |
| Total | |
£ | 4,431,840 | | |
£ | 2,159,058 | |
The
fair value of accounts payable and accrued expenses are not materially different to the book value.
NOTE
9. COMMITMENTS AND CONTINGENCIES
From
time to time, the Company may become involved in various lawsuits and legal proceedings, which arise in the ordinary course of business.
Litigation is subject to inherent uncertainties, and an adverse result in these or other matters may arise from time to time that may
harm business.
In
accordance with the terms of a Convertible Loan Note (‘Note’) on August 9, 2022 (the Conversion Date) the Company issued
183,820 Ordinary Shares and 367,640 listed warrants to the Note holder in full satisfaction of the Note in the aggregate amount of $781,233.
The holder filed a claim in the English courts on June 19, 2023 asserting that notice was provided such that the Company should have
paid it the value of the Note in cash, rather than by settling it through the issuance of Ordinary Shares and listed warrants. The holder
is demanding payment of the face value of the Note, together with interest, (approximately $860,000). The litigation process is in its
early stages and is not expected to conclude until late 2024 or later. The Company has retained English solicitors and is contesting the claim
in its entirety. The Company believes that it acted correctly under the terms of the Note and has accounted for the transaction on that
basis, and that no further amounts are payable to the holder.
NOTE
10. LEASES
The
Company leases certain office space under operating leases for use in operations. The Company recognizes operating lease expense on
a straight-line basis over the lease term. Management determines if an arrangement is a lease at contract inception. Lease and
non-lease components are accounted for as a single component for all leases. Operating lease right to use (ROU) assets and
liabilities are recognized at the lease commencement date based on the present value of the future lease payments over the expected
lease term, which includes optional renewal periods if we determine it is reasonably certain that the option will be exercised. As
our leases do not provide an implicit rate, the discount rate used in the present value calculation represents our incremental
borrowing rate determined using information available at the commencement date. Operating lease expense is included as a component
of research and development and administrative expenses in the consolidated statements of operations. For the years ended December
31, 2023 and 2022, the Company recorded operating lease expense of £402,314
and £402,314,
respectively. Cash payments on lease
liabilities during the years ended December 31, 2023 and 2022 totaled £447,015
and £763,487,
respectively. At December 31, 2023 and 2022, weighted-average remaining lease term and discount rate were as follows:
SCHEDULE
OF WEIGHTED AVERAGE REMAINING LEASE TERM AND DISCOUNT RATE
| | |
December
31, | |
| | |
2023 | | |
2022 | |
| Weighted-average remaining lease term | |
| 5.11
years | | |
| 8.2
years | |
| Weighted-average discount rate | |
| 8.6 | % | |
| 8.6 | % |
The
following is a maturity analysis of the annual undiscounted cash flows reconciled to the carrying value of the operating lease liabilities
as of December 31, 2023:
SCHEDULE
OF OPERATING LEASE LIABILITIES
| Years Ended December 31, | |
| |
| 2024 | |
£ | 447,015 | |
| 2025 | |
| 447,015 | |
| 2026 | |
| 447,015 | |
| 2027 | |
| 447,015 | |
| 2028 | |
| 447,015 | |
| Less imputed interest | |
| (433,919 | ) |
| Total | |
£ | 1,801,156 | |
NOTE
11. CONVERTIBLE LOAN
The
Company entered into a $20 million
convertible loan note instrument in April 2021. The note has a 5% annual interest rate. During the years ended December 31, 2023 and
2022, the Company converted loan notes totaling $809,692 and
$14,228,245,
respectively, into ordinary shares and warrants and repaid $0 and
$3,195,765,
respectively, of the convertible loan note. The convertible loan was recognized as a hybrid financial instrument and accounted for
as two separate components: (i) a loan and (ii) an embedded conversion option derivative. As of December 31 2023, the convertible loan had either been fully paid down or converted. As such, the balance of
both the convertible loan and corresponding embedded derivative was $0 as of December 31, 2023.
(i)
The convertible loan’s initial fair value was the residual amount of the consideration received, net of attributable costs, after
separating out the fair value of the embedded conversion option derivative. The loan is subsequently measured at its amortized cost.
(ii)
The embedded conversion option derivative was initially measured at fair value and is subsequently remeasured to fair value at each reporting
date. The embedded derivative could have been classified as a component of equity only if in all cases the contract would be settled
by the Company delivering a fixed number of its own equity instruments in exchange for a fixed amount of cash or debt redemption. That
is, had the embedded instrument satisfied the “fixed-for-fixed” criteria outlined in ASC 815-40. However, the convertible
instrument included a conversion feature resulting in settlement in a variable number of shares and consequently, was not considered
indexed to the company’s shares (i.e. it did not qualify for the scope exception to derivative accounting outlined in ASC 815-40).
As a result, the derivative is presented in the consolidated balance sheet as a liability in accordance with ASC 815-15, Derivatives
and Hedging-Embedded Derivatives. Changes in the fair value (gains or losses) of the derivative at the end of each period are recorded
in the consolidated statements of operations.
On
August 9, 2022, the Company agreed with one of the loan note holders not to exercise the right to require the loan notes to be repaid
in cash in accordance with the terms of the loan notes and to amend certain other aspects of the loan notes (“2022 amended loan
note”). As additional consideration, the Company has issued warrants to subscribe for 11,678 ordinary shares in the share capital
of the Company.
In
accordance with ASC 470-50, the modifications to the 2022 amended loan notes represent a substantial modification as the amended debt instrument was considered substantially different (as compared to the original instrument) due to the following changes:
(i)
Removing the exercise of the right to require the loan in cash as of August 9, 2022.
(ii)
Extending the repayment date to January 31, 2023 and modifying the structure to be repaid in shares if not redeemed before in cash.
(iii)
Revising the conversion price for the conversion of the loan notes in shares. The revised conversion price would be $0.50
and would be based on the the 5-day trailing VWAP of the Company’s ADS if it is trading above that amount. Further, the conversion option would have a $0.20
as a floor.
(iv)
Giving the option to the holder for redemption in cash, which will occur no later than February 10, 2023 and to the Company for an early
redemption at any moment but having the Holder an option to convert into shares using the revised conversion price at that moment.
On
April 3, 2023, the Company agreed with the loan note holder not to exercise the right to require the loan notes to be repaid in cash
in accordance with the terms of the loan notes and to amend certain other aspects of the loan notes (“2023 amended loan note”).
As additional consideration, the Company has issued warrants to subscribe for 200,000 ordinary shares in the share capital of the Company.
This warrant contained a condition whereby if a registration statement, to be filed by the Company, registering all of the securities
underlying the note holder’s amended convertible loan note, was not declared effective by July 31, 2023, the note holder will be
entitled to receive 0.30 Ordinary Shares for each share it was originally entitled to purchase under these warrants without the payment
of any additional consideration. No such registration statement was filed. The related fair value of the issue of any additional securities
is approximately £29,000 ($37,000) and is not considered material to the financial statements.
The
modifications to the 2023 amended loan notes represent as substantial amendment as the modifications are related to:
(i)
A waiver to any defaults arising in connection with the 2022 amended loan notes.
(ii)
Extending the repayment date to January 15, 2024; and
(iii)
Amend the Conversion Price (as defined in the Loan Note) of the outstanding loan notes to be the lesser of $1.00 or the lowest closing
price of the Ordinary Shares during the ten (10) day period prior to the date the Noteholder delivers a notice of conversion to the Company,
not to be lower than $0.20
The
following table summarizes the changes in the convertible debt instrument during the year ended December 31, 2023:
SCHEDULE
OF CHANGES IN CONVERTIBLE DEBT INSTRUMENT
| | |
Residual
loan | | |
Embedded
Derivative | | |
Total | |
| Balance at December 31, 2022 | |
£ | 653,484 | | |
£ | 2,439 | | |
£ | 655,923 | |
| Accrued interest | |
| 71,568 | | |
| - | | |
| 71,568 | |
| Repayment | |
| (639,336 | ) | |
| (2,439 | ) | |
| (641,775 | ) |
| Modification of loan notes | |
| (53,619 | ) | |
| - | | |
| (53,619 | ) |
| Currency adjustment | |
| (32,097 | ) | |
| - | | |
| (32,097 | ) |
| Balance at December 31, 2023 | |
£ | - | | |
£ | - | | |
£ | - | |
The
value of the embedded derivative was remeasured to fair value at each reporting date, based on the Black-Scholes valuation model,
with recognition of the changes in fair value in the consolidated statements of operations. The inputs associated with calculating
the fair value of the embedded derivative are considered to be Level 2. The following inputs were used to determine the conversion
option:
SCHEDULE
OF BLACK SCHOLES VALUATION MODEL OF MEASUREMENT INPUT
| | |
December 31, 2022 | |
| Exercise price in USD | |
$ | 100.00 | |
| Share price in USD | |
$ | 77.00 | |
| Time to maturity | |
| 0.8
years | |
| Expected volatility | |
| 90 | % |
| Risk free interest rate (US treasury bond) | |
| 4.1 | % |
| Dividend yield | |
| - | |
The
following inputs were used in the Black-Scholes valuation model to determine the related share purchase warrants:
BLACK-SCHOLES VALUATION MODEL TO DETERMINE THE RELATED SHARE PURCHASE WARRANTS
| |
|
December
31, 2023 | |
| Exercise price in USD |
|
$ | 100.00 | |
| Share price in USD |
|
$ | 3.17 | |
| Time to maturity |
|
| 4.3
years | |
| Expected volatility |
|
| 90 | % |
| Risk free interest rate (US treasury bond) |
|
| 4.5 | % |
| Dividend yield |
|
| - | |
NOTE
12. SHAREHOLDERS’ EQUITY
Ordinary
shares
The
Ordinary shares have no specific rights, preferences or restrictions attached to them.
Deferred
shares
Deferred
shares have the following properties:
| |
a. |
do
not entitle their holders to receive any dividend or other distribution; |
| |
b. |
do
not entitle their holders to receive a share certificate in respect of the relevant shareholding; |
| |
c. |
do
not entitle their holders to receive notice of, nor to attend, speak or vote at, any general meeting of the Company; |
| |
d. |
entitles
their holders on a return of capital on a winding up of the Company (but not otherwise) only to the repayment of the amount paid
up on that share after payment of the capital paid up on each Ordinary Share in the share capital of the Company and the further
payment of £100,000,000 on each ordinary share; |
| |
e. |
do
not entitle their holders to any further participation in the capital, profits or assets of the Company. The Deferred Shares shall
not be capable of transfer at any time other than with the prior written consent of the directors of the Company. |
A
Ordinary shares
The
A Ordinary shares ranked equally with all other shares in issue in that on a vote every member has one vote for each share held. The
A ordinary shares contain preferential economic rights such that, in the event of a share or asset sale (as defined in the Articles of
Association), they provide a return to the holders of the A Ordinary Shares of an amount greater than or equal to 1.5x the price paid
by the investors for A Ordinary Shares. The A Ordinary shares have an anti-dilution provision where shares are subsequently issued at
a price below £215.00 per share, whereby the existing A Ordinary shareholders receive additional compensation shares in line with
the formula set out in the Articles of Association. The A Ordinary shares rank equally with all other shares in issue with respect to
dividends.
Immediately
prior to the completion of the IPO, 493,860 ordinary shares were issued, under the terms of the Articles of Association to certain shareholders
who, prior to the IPO, owned A Ordinary shares which carried the right, to subscribe at nominal value for a certain number of additional
shares, calculated by reference to the pre-money valuation of the IPO. As part of the IPO share issue, the Company re-organized its share
capital whereby all of the outstanding series A ordinary shares were re-designated as ordinary shares of the Company on a one for one
basis and as such no anti-dilution provisions are included within the issued shares.
Reorganization
and IPO
Immediately
prior to completion of the IPO, the Company re-organized its share capital whereby all of the outstanding series A ordinary shares were
re-designated as ordinary shares of TC BioPharm (Holdings) plc on a one for one basis. Immediately prior to the completion of the offering,
a further 493,860 ordinary shares were issued, under the terms of the Articles of Association to certain shareholders who, prior to the
IPO, owned A ordinary shares which carried the right, to subscribe at nominal value for a certain number of additional shares, calculated
by reference to the pre-money valuation of the IPO. The fair value of the shares issued was £3.8 million.
On
February 10, 2022, the Company issued 3,164 ADSs representing 63,280 ordinary shares with nominal value of £31,640 and warrants
to buy 6,278 ADSs on conversion of loan notes totaling $13.4 million (£9.9 million).
On
February 10, 2022, the Company completed the IPO, listing on Nasdaq, issuing 4,117 ADSs representing 82,353 ordinary shares with
nominal value of £41,176 and warrants to buy 9,470 ADSs for proceeds before expenses of $17.5 million (£12.8 million).
Funding costs of $3.0 million (£2.2 million) including underwriter fees were incurred.
Share
Issuances
Between
June 7, 2022 and June 8, 2022, the Company issued and sold 11,500 ADSs representing 230,000 ordinary shares generating proceeds of
$4.6 million (£3.7 million) before deductions for offering expenses of approximately $0.8 million (£0.6 million).
On
August 9, 2022, Convertible Loan Noteholders with loan notes with a face value of $0.8 million (£0.6 million) agreed to not exercise
their right to be repaid and in consideration for this agreement received warrants over 233,560 ordinary shares. In addition, the conversion
price of the loan notes was amended to be the lower of (i) the 5-day trailing VWAP of the Company’s ADS calculated as at 31 January
2023 and (ii) $25.00, subject to not being below $10.00.
On
August 9, 2022, the Company issued 183 ADSs representing 3,676 ordinary shares and warrants to buy 366 ADSs on conversion of loan
notes totaling $0.8 million (£0.7 million).
On
November 15, 2022, the Company issued 21 Ordinary shares (number stated prior to the reverse split) for a consideration of $7.565 (£6.362)
per share.
On
November 24, 2022, the Company issued 3 Ordinary shares for a consideration of $6.51 (£5.41) per share.
On
November 27, 2022, the Company entered into a Securities Purchase Agreement (the “Purchase Agreement”) with certain accredited
investors (the “Investors”) as purchasers. Pursuant to the Purchase Agreement, the Company sold, and the Investors purchased
in a private placement an aggregate of 7,750 American Depositary Shares (the “ADSs”), pre-funded warrants to purchase up
to 65,750 ADS (the “Pre-Funded Warrants”), Series A purchase warrants to purchase up to 73,500 ADSs (the “Series
A Ordinary Warrants”) and Series B purchase warrants to purchase up to 73,500 ADSs (the “Series B Ordinary Warrants”
and together with the Series A Ordinary Warrants, the “Ordinary Warrants”) for aggregate gross proceeds of $7.4 million (£6.1
million), excluding any proceeds that may be received upon exercise of the Ordinary Warrants. The purchase price for each ADS and associated
Ordinary Warrants is $100 and the purchase price per each Pre-Funded Warrant and associated Ordinary Warrants is $99.98.
On
March 27, 2023, the Company, entered into a Second Securities Purchase Agreement with Investors, pursuant to which the Company agreed
to issue and sell an aggregate of 10,750
ADSs, pre-funded warrants to purchase up to 161,125
ADSs, and Series C purchase warrants to purchase
up to 171,875
ADSs Securities. In addition, the Company also
issued placement agent warrants to purchase 12,891 ADSs. The purchase price for each ADS and associated Ordinary Warrants was $32
(on a post-split basis) and the purchase price
per each Pre-Funded Warrant and associated Ordinary Warrants was $31.98
(on a post-split basis). The Ordinary Warrants
were immediately exercisable, expire five (5) years from the date of issuance and the Pre-Funded Warrants may be exercised at any time
until all of the Pre-Funded Warrants are exercised in full. The total net proceeds from this offering were approximately $4.6
million, after deducting estimated offering expenses
of approximately $0.9
million.
On
March 27, 2023, the Company also agreed that certain existing warrants to purchase up to an aggregate of 140,000
ADSs of the Company that were previously issued on November 30, 2022, at an exercise price of $100
(on a post-split basis) per ADS and expiration dates of May 30, 2025 and May 30, 2028, were amended effective upon the closing of
the Offering so that the amended warrants will have a reduced exercise price of $35
(on a post-split basis) per ADS. To account for the modification, the Company recognized the increase in fair value of the
modified warrants (measured as the difference between the fair value immediately before and after the modification) a as a charge
against the gross proceeds of the offering.
On
August 30, 2023, the Company entered into an agreement with its Series A and B warrant holders whereby it induced 70,000 and 70,000 of
the outstanding warrants, respectively. In addition, the Company also entered into an agreement with its Series C warrant holders to
induce all of the outstanding warrants (171,875). The inducement resulted in gross proceeds to the Company of approximately $2.8 million.
In order to incentivize the inducement, the Company issued 623,750 Series D warrants to the Series, A, B and C warrant holders. In addition,
the Company also issued placement agent warrants to purchase 23,391 ADSs. The Ordinary Warrants were immediately exercisable and expire
five 5.5 years from the date of issuance. The Company received aggregate gross proceeds of approximately £2.2
million (approximately $2.8
million) from the exercise of the Existing Warrants
by the Holders, before deducting placement agent fees payable by the Company. The Company accounted for the inducement in accordance
with modification and exchange guidance in ASC 815-40 and recognized the fair value of the issued Series D warrants as an equity issuance
cost.
On
December 18, 2023, the Company entered into a Third Securities Purchase Agreement with a certain institutional Investor pursuant to
which the Company agreed to issue and sell to the Investor in a best-efforts public offering 75,000
ADSs representing 1,500,000
ordinary shares, pre-funded warrants to purchase up to 1,675,000
ADS representing 33,500,000
Ordinary Shares (the “Pre-Funded Warrants”), and Series E purchase warrants to purchase up to 1,750,000
ADSs representing 35,000,000
Ordinary Shares (the “Warrants” and together with the Pre-Funded Warrants and the ADSs, the “Securities”).
In addition, the Company issued placement agent warrants to purchase 131,250 ADSs representing 2,625,000 ordinary shares. The
purchase price for each ADS and associated Warrant was $2.00
and the purchase price per each Pre-Funded Warrant and associated Warrant was $1.999.
The Warrants are immediately exercisable, will expire five
years from the date of issuance and have an exercise price of £1.5814.
Further, pursuant to ASC 815-40, we concluded that the warrants were indexed to the company’s stock and should therefore be
equity classified. The relative fair value of the warrants upon issuance were approximately $1.5
million. The Pre-Funded Warrants may be exercised at any time until all of the Pre-Funded Warrants are exercised in full at an
exercise price of $0.001
per ADS. Additionally, the Company agreed that the Series E warrants to purchase up to an aggregate of 623,750
ADSs of the Company that were previously issued on September 5, 2023, at an exercise price of £7.00
per ADS and an expiration date of March 5, 2029, were amended effective upon the closing of the offering so that the amended warrant
will have a reduced exercise price of £1.5814
(or $2.00,
as translated for illustration to U.S. dollars at the rate of £1.00
to $1.264
as of December 18, 2023) per ADS. To account for the modification, the Company recognized the increase in fair value of the modified
warrants (measured as the difference between the fair value immediately before and after the modification) as a charge against the
gross proceeds of the offering.
Reverse
share split
On
November 18, 2022, the Company completed a reverse stock split of one (1) new share for every fifty (50) existing shares effective November
21, 2022. As a result, the depositary bank, BNY Mellon effected a reverse stock split on the Company’s American Depositary Receipt
(“ADR”) program. Following the reverse split, a sub-division of every Ordinary share into one new Ordinary Share with a nominal
value of £0.0001 and one deferred share with a nominal value of £0.4999 was enacted.
As
a result of the share split, all references in these consolidated financial statements and accompanying notes to units of ordinary shares
or per share amounts are reflective of the reverse share split for all periods presented. In addition, the exercise prices and the numbers
of ordinary shares issuable upon the exercise of any outstanding options to purchase ordinary shares were proportionally adjusted pursuant
to the respective anti-dilution terms of the share-based payment plans.
ADS
Ratio Change
On
December 15, 2023, the Company changed its ratio of ADSs ordinary shares from one ADS representing one ordinary share to one ADS representing
20 ordinary shares. As a result of the ratio change, all references in these consolidated financial statements and accompanying notes
to units of ordinary shares underlying ADSs are reflective of the ratio change for all periods presented. In addition, the exercise prices
and the numbers of ordinary shares issuable upon the exercise of any outstanding options to purchase ordinary shares were proportionally
adjusted pursuant to the respective anti-dilution terms of the share-based payment plans.
NOTE
13. SHARE-BASED COMPENSATION
Enterprise
Management Incentive (EMI) share option scheme
The
Company operates an HMRC Approved Enterprise Management Incentive (“EMI”) share option scheme for employees. Effective December
16, 2014, the Company approved a share option scheme under which the Board of Directors of the Company can award options to directors,
officers, employees and consulting personnel of the Company. The Board of Directors will determine the terms, limitations, restrictions
and conditions of the options granted under the plan.
The
Company has granted options over ADSs to certain employees.
SCHEDULE
OF GRANTED OPTIONS
| | |
Number
of share options | | |
Weighted average
exercise price per ADS £ | |
| | |
| | |
| |
| Outstanding at December 31, 2022 | |
| 5,329 | | |
£ | 460 | |
| Granted during the period | |
| - | | |
| - | |
| Exercised during the period | |
| - | | |
| - | |
| Forfeited during the
period | |
| - | | |
| - | |
| Outstanding at December 31, 2023 | |
| 5,329 | | |
£ | 460 | |
| | |
| | | |
| | |
| Exercisable at December 31, 2023 | |
| 5,329 | | |
£ | 460 | |
| Unexercisable at December 31, 2023 | |
| - | | |
£ | - | |
The
estimated fair value of the options outstanding in the period was calculated by applying a Monte Carlo Simulation for those options issued
in 2020 and 2019 and a Black Scholes Model for those options issued in prior periods. The most appropriate approach is selected with
reference to the share capital structure at the time of grant. The expense recognized for share-based payments in respect of employee
services received during the year ended December 31, 2023 was £0 as all options were fully vested as of December 31, 2023.
2021
Share Option Scheme
Effective
immediately prior to completion of the IPO on February 10, 2022, the Company adopted a new share option scheme, or the 2021 Share Option
Scheme, for the purpose granting share options to incentivize the Company’s directors, employees and consultants. The 2021 Share
Option Scheme incorporates a sub-plan for option holders subject to taxation in the United States, or the 2021 U.S. Sub-Plan, to provide
for the grant of U.S. qualified incentive options.
The
Company has granted options over ADSs to certain employees and directors.
| | |
Number
of share options | | |
Weighted average
exercise price per ADS $ | |
| | |
| | |
| |
| Outstanding at December 31, 2022 | |
| 2,615 | | |
$ | 4,240 | |
| Granted during the period | |
| 35,125 | | |
| 8 | |
| Exercised during the period | |
| - | | |
| - | |
| Forfeited during the
period | |
| (674 | ) | |
| 4,240 | |
| Outstanding at December 31, 2023 | |
| 37,066 | | |
$ | 230 | |
| | |
| | | |
| | |
| Exercisable at December 31, 2023 | |
| 36,782 | | |
$ | 8 | |
| Unexercisable at December 31, 2023 | |
| 284 | | |
$ | 4,240 | |
The
totals of options and related exercise price are for options over ADSs and reflect the ratio change on December 15, 2023.
The
estimated fair value of the options outstanding in the period was calculated by applying a Black Scholes Model. The most appropriate
approach is selected with reference to the share capital structure at the time of grant. The weighted average fair value of the
options as of December 31, 2023 and 2022 was $62.12 and $1,068, respectively.
The expense recognized for share-based payments in respect of employee services received during the years ended December 31, 2023
and 2022 was £444,459
and £1,123,250,
respectively.
The
options granted under the 2021 share option scheme will typically vest over three years after the date of grant. In some cases, options
granted to senior management vested immediately. As of December 31, 2023 the unvested options would, under the agreed terms, vest evenly
over the remaining period in either six month or annual installments.
Additional
Right to Subscribe for Shares
On
August 25, 2020, the Company issued Ordinary shares, which included an additional right to subscribe for a fixed number (15,891) of
shares at £215.00 per share at a future date based on certain clinical and commercial milestones. The estimated fair value of the
right to subscribe was calculated by applying a Black Scholes Model. This was deemed the most appropriate approach due to the future
liquidity event being date-uncertain and could take one of many forms.
NOTE
14. FAIR VALUE MEASUREMENTS
The
fair value of the Company’s financial assets and liabilities reflects management’s estimate of amounts that the Company would
have received in connection with the sale of the assets or paid in connection with the transfer of the liabilities in an orderly transaction
between market participants at the measurement date. In connection with measuring the fair value of its assets and liabilities, the Company
seeks to maximize the use of observable inputs (market data obtained from independent sources) and to minimize the use of unobservable
inputs (internal assumptions about how market participants would price assets and liabilities). The following fair value hierarchy is
used to classify assets and liabilities based on the observable inputs and unobservable inputs used in order to value the assets and
liabilities:
| |
Level
1: |
Quoted
prices in active markets for identical assets or liabilities. An active market for an asset or liability is a market in which transactions
for the asset or liability occur with sufficient frequency and volume to provide pricing information on an ongoing basis. |
| |
|
|
| |
Level
2: |
Observable
inputs other than Level 1 inputs. Examples of Level 2 inputs include quoted prices in active markets for similar assets or liabilities
and quoted prices for identical assets or liabilities in markets that are not active. |
| |
|
|
| |
Level
3: |
Unobservable
inputs based on the Company’s assessment of the assumptions that market participants would use in pricing the asset or liability. |
The
Company had cash and cash equivalents of £2.5m as of December 31, 2023. The cash and cash equivalents are carried at fair value
due to the liquid nature of the instruments and are measured in Level 1.
In
addition, the Company also had numerous outstanding warrants that were classified in Level 2 due to our use of implied volatility in
determining the expected volatility input for purposes of determining the instruments fair value via the Black-Scholes valuation model.
The details of the issued warrants were as follows:
On
February 10, 2022, the Company completed its IPO and issued ADSs and listed warrants to buy ADSs. The ADSs and warrants are
considered two freestanding financial instruments because each can be traded separately. The exercise price of the Warrants is
$4,250 per
ADS (on a post-split basis) and will expire on the sixth anniversary of the date of issuance. The exercise price is subject to
standard anti-dilutive adjustments in the event of certain stock splits, stock combinations, stock dividends or recapitalizations,
and it is also subject to adjustment in certain events specified in the warrant agreement.
On
November 27, 2022, the Company entered into the First Purchase Agreement with Investors as purchasers. Pursuant to the First
Purchase Agreement, the Company sold, and the Investors purchased in a private placement an aggregate of 7,750
ADSs, pre-funded warrants to purchase up to 65,750
ADS (the “Pre-Funded Warrants”), Series A purchase warrants to purchase up to 73,500
ADSs (the “Series A Ordinary Warrants”) and Series B purchase warrants to purchase up to 73,500
ADSs (the “Series B Ordinary Warrants” and together with the Series A Ordinary Warrants, the “Ordinary
Warrants”) for aggregate gross proceeds of $7,350,000,
excluding any proceeds that may be received upon exercise of the Ordinary Warrants. In addition, the Company also issued placement
agent warrants to purchase 5,513 ADSs. The
purchase price for each ADS and associated Ordinary Warrants is $100
(on a post-split basis) and the purchase price per each Pre-Funded Warrant and associated Ordinary Warrants is $99.98
(on a post-split basis).
On
March 27, 2023, the Company, entered into a Second Securities Purchase Agreement with Investors, pursuant to which the Company
agreed to issue and sell an aggregate of 10,750 ADSs,
pre-funded warrants to purchase up to 161,125 Pre-Funded
Warrants, and Series C purchase warrants to purchase up to 171,875 ADSs
Securities. In addition, the Company also issued placement agent warrants to purchase 12,891 ADSs. The purchase price for each ADS
and associated Ordinary Warrants was $32 (on
a post-split basis) and the purchase price per each Pre-Funded Warrant and associated Ordinary Warrants was $31.98 (on
a post-split basis). The Ordinary Warrants were immediately exercisable, expire five (5) years from the date of issuance and the
Pre-Funded Warrants may be exercised at any time until all of the Pre-Funded Warrants are exercised in full.
On July 10, 2023, the Company entered into a warrant amendment with an existing investor pursuant to which the Company and the investor
agreed that certain Series A and Series B warrants to purchase 140,000 ADSs of the Company that were previously issued on November 30, 2022 (the
“November 2022 Warrants”) and certain Series C warrants to purchase 171,875 ADSs of the Company that were previously issued
on March 30, 2023 (the “March 2023 Warrants,” and together with the November 2022 Warrants, the “Existing Warrants”)
would be amended as follows: (i) amend the current exercise price on all Existing Warrants so that it is now equal to £7.00, (ii)
extend the termination date on 50% of the November 2022 Warrants and all of the March 2023 Warrants until May 30, 2028 and (iii) amend
to the definition of “Black Scholes Value” included in Section 3(e) of the Existing Warrants. As a result of the amendments to
the related warrants (Series A, Series B and Series C) they were reclassified to equity as they no longer contained a strike price denominated in a foreign currency.
Further, the Series A, B and C warrants were also re-measured to a fair value of $6.40 per warrant as of the modification date using the
Black-Scholes model with the following inputs:
SCHEDULE
OF WARRANT USING BLACK-SCHOLES MODEL INPUTS
| | |
July 10, 2023 | |
| Exercise price | |
$ | 9.00 | |
| Share price | |
$ | 9.00 | |
| Time to maturity | |
| 4.8 years | |
| Expected volatility | |
| 90 | % |
| Risk free interest rate (US treasury bond) | |
| 4.0 | % |
| Dividend yield | |
| - | |
On August
30, 2023, the Company entered into an agreement with its Series A and B warrant holders whereby it induced 70,000 and 70,000 of the outstanding
warrants, respectively. In addition, the Company also entered into an agreement with its Series C warrant holders to induce all of the
outstanding warrants (171,875). The inducement resulted in gross proceeds to the Company of approximately $2.8 million. In order to incentivize
the inducement, the Company issued 623,750 Series D warrants to the Series, A, B and C warrant holders. In addition, the Company also
issued placement agent warrants to purchase 23,391 ADSs. The Ordinary Warrants were immediately exercisable and expire five 5.5 years
from the date of issuance.
On December
18, 2023, the Company, entered into a Securities Purchase Agreement with Investors, pursuant to which the Company agreed to issue and
sell an aggregate of 75,000 ADSs, pre-funded warrants to purchase up to 1,675,000 Pre-Funded Warrants, and Series C purchase warrants
to purchase up to 1,750,000 ADSs Securities. The offering resulted in gross proceeds of $3.5 million. In addition, the Company also issued
placement agent warrants to purchase 131,250 ADSs. The purchase price for each ADS and associated Ordinary Warrants was $2.00 and the
purchase price per each Pre-Funded Warrant and associated Ordinary Warrants was $1.99. The Ordinary Warrants were immediately exercisable,
expire five (5) years from the date of issuance and the Pre-Funded Warrants may be exercised at any time until all of the Pre-Funded Warrants
are exercised in full.
While
the Series D and Series E warrants are equity classified, there are remaining IPO, Series A and Series B warrants that are still
liability classified. However, as a result of being significantly out of the money, the fair value of the liability classified
warrants was not material as of December 31, 2023.
Unlisted
warrants in issue
Series
A warrants
The
fair value of each of the warrants was approximately $0.37 and $51.60 as
of December 31, 2023 and 2022, respectively.
The
inputs associated with calculating the fair value of the warrants are considered to be Level 2 and were valued using a Black-Scholes
valuation model. The inputs were as follows:
SCHEDULE OF FAIR VALUE ASSETS AND
LIABILITIES MEASURED ON RECURRING AND NON RECURRING BASIS
| | |
December
31, 2023 | | |
December
31, 2022 | |
| Exercise price | |
$ | 100.00 | | |
$ | 100.00 | |
| Share price | |
$ | 3.17 | | |
$ | 77.00 | |
| Time to maturity | |
| 4.4
years | | |
| 5.4
years | |
| Expected volatility | |
| 90 | % | |
| 85 | % |
| Risk free interest rate (US treasury bond) | |
| 4.0 | % | |
| 3.9 | % |
| Dividend yield | |
| - | | |
| - | |
Series
B warrants
The
fair value of each of the warrants was approximately $0.01
and $36.80
as of December 31, 2023 and 2022, respectively.
The inputs associated with calculating the fair value of the warrants are considered to be Level 2 and were valued using a Black-Scholes
valuation model. The inputs were as follows:
| | |
December
31, 2023 | | |
December
31, 2022 | |
| Exercise price | |
$ | 100.00 | | |
$ | 100.00 | |
| Share price | |
$ | 3.17 | | |
$ | 77.00 | |
| Time to maturity | |
| 1.4
years | | |
| 2.4
years | |
| Expected volatility | |
| 90 | % | |
| 90 | % |
| Risk free interest rate (US treasury bond) | |
| 4.0 | % | |
| 4.3 | % |
| Dividend yield | |
| - | | |
| - | |
Series
A-B placement agent warrants
The
fair value of each of the warrants was $0.31
and $51.20
as of December 31, 2023 and 2022, respectively.
The inputs associated with calculating the fair value of the warrants are considered to be Level 2 and were valued using a Black-Scholes
valuation model. The inputs were as follows:
| | |
December
31, 2023 | | |
December
31, 2022 | |
| Exercise price | |
$ | 125.00 | | |
$ | 125.00 | |
| Share price | |
$ | 3.17 | | |
$ | 77.00 | |
| Time to maturity | |
| 4.4
years | | |
| 5.4
years | |
| Expected volatility | |
| 90 | % | |
| 90 | % |
| Risk free interest rate (US treasury bond) | |
| 4.0 | % | |
| 4.0 | % |
| | |
| - | | |
| - | |
Series
C warrants
The
fair value of each of the warrants was $21.63 as of March 30, 2023 (issuance
date). The warrants were induced during August of 2023 (further information is included in Note 12).
The inputs associated with calculating the fair value of the warrants are considered to be Level 2 and were valued using a Black-Scholes
valuation model. The inputs were as follows:
| | |
March 30, 2023 | |
| Exercise price | |
$ | 35.00 | |
| Share price | |
$ | 31.00 | |
| Time to maturity | |
| 5.0 years | |
| Expected volatility | |
| 90 | % |
| Risk free interest rate (US treasury bond) | |
| 4.0 | % |
| Dividend yield | |
| - | |
Series
C placement agent warrants
The
fair value of each of the warrants was $0.69
as of December 31, 2023 and $21.09
as of March 30, 2023 (issuance date), respectively.
The inputs associated with calculating the fair value of the warrants are considered to be Level 2 and were valued using a Black-Scholes
valuation model. The inputs were as follows:
| | |
December
31, 2023 | |
|
March 30, 2023 |
|
| Exercise price | |
$ | 40.00 | |
|
$ |
40.00 |
|
| Share price | |
$ | 3.17 | |
|
$ |
31.00 |
|
| Time to maturity | |
| 4.2
years | |
|
|
5.0 years |
|
| Expected volatility | |
| 90 | % |
|
|
90 |
% |
| Risk free interest rate (US treasury bond) | |
| 4.0 | % |
|
|
4.49 |
% |
| Dividend yield | |
| - | |
|
|
- |
|
Series
D warrants
The
fair value of each of the warrants was $8.46 as of the August 30, 2023 issuance date. As the warrants were equity classified, they were
not re-measured to fair value as of December 31, 2023.
The inputs associated with calculating the fair value of the warrants are considered to be Level 2 and were valued using a Black-Scholes
valuation model. The inputs were as follows:
| | |
August 30, 2023 | |
| Exercise price | |
$ | 8.80 | |
| Share price | |
$ | 11.00 | |
| Time to maturity | |
| 5.5 years | |
| Expected volatility | |
| 90 | % |
| Risk free interest rate (US treasury bond) | |
| 4.0 | % |
Series
D placement agent warrants
The
fair value of each of the warrants was $8.20 as of the August 30, 2023 issuance date. As the warrants were equity classified, they were
not re-measured to fair value as of December 31, 2023.
The inputs associated with calculating the fair value of the warrants are considered to be Level 2 and were valued using a Black-Scholes
valuation model. The inputs were as follows:
| | |
August 30, 2023 | |
| Exercise price | |
$ | 11.00 | |
| Share price | |
$ | 11.00 | |
| Time to maturity | |
| 5.5 years | |
| Expected volatility | |
| 90 | % |
| Risk free interest rate (US treasury bond) | |
| 4.0 | % |
| Dividend yield | |
| - | |
Series
E warrants
The
fair value of each of the warrants was $1.97
as of the December 18, 2023 issuance date. As
the warrants were equity classified, they were not re-measured to fair value as of December 31, 2023.
The inputs associated with calculating the fair value of the warrants are considered to be Level 2 and were valued using a Black-Scholes
valuation model. The inputs were as follows:
| |
|
December
18, 2023 |
|
| Exercise
price |
|
$ |
2.00 |
|
| Share
price |
|
$ |
2.61 |
|
| Time
to maturity |
|
|
5.0
years |
|
| Expected
volatility |
|
|
90 |
% |
| Risk
free interest rate (US treasury bond) |
|
|
4.0 |
% |
Series
E placement agent warrants
The
fair value of each of the warrants was $1.89
as of the December 18, 2023 issuance
date. As the warrants were equity classified, they were not re-measured to fair value as of December 31, 2023.
The inputs associated with calculating the fair value of the warrants are considered to be Level 2 and were valued using a Black-Scholes
valuation model. The inputs were as follows:
| | |
December 18, 2023 | |
| Exercise price | |
$ | 2.50 | |
| Share price | |
$ | 2.61 | |
| Time to maturity | |
| 5.0 years | |
| Expected volatility | |
| 90 | % |
| Risk free interest rate (US treasury bond) | |
| 4.0 | % |
| Dividend yield | |
| - | |
NOTE
15. INCOME TAXES
SCHEDULE
OF INCOME TAXES
| | |
Year
Ended | | |
Year
Ended | |
| | |
December
31, 2023 | | |
December
31, 2022 | |
| | |
£ | | |
£ | |
| Current tax | |
| | | |
| | |
| Corporation
tax credit | |
| 1,088,729 | | |
| 1,720,000 | |
| Total current tax credit | |
| 1,088,729 | | |
| 1,720,000 | |
| | |
| | | |
| | |
| Reconciliation of loss before
tax to the tax credit for the year | |
| | | |
| | |
| | |
| | | |
| | |
| Loss on ordinary activities multiplied by the
standard rate of tax of 23.5% (2022: 19%) | |
| 1,643,985 | | |
| 576,384 | |
| | |
| | | |
| | |
| Adjustments in respect of prior years | |
| 45,136 | | |
| 154,422 | |
| Non-deductible expenses | |
| (1,497,764 | ) | |
| (1,834,581 | ) |
| Super deductions | |
| - | | |
| 13,178 | |
| Change in deferred tax asset | |
| - | | |
| (186,879 | ) |
Change in valuation allowance | |
| 357,897 | | |
| (671,566
| ) |
| Foreign rate differential | |
| (29,318 | ) | |
| - | |
| Other | |
| (233,096 | ) | |
| - | |
| Additional allowance in respect of enhanced
R&D relief | |
| - | | |
| 4,198,115 | |
| Surrender of tax losses for R&D tax credit
refund | |
| - | | |
| (2,109,111 | ) |
| Adjustments relating to GAAP | |
| (241,704 | ) | |
| 14,460 | |
| R&D tax credits
generated | |
| 1,043,593 | | |
| 1,565,578 | |
| Current
tax credit | |
| 1,088,729 | | |
| 1,720,000 | |
As
of December 31, 2023, the Company’s net operating loss carryforwards in the United Kingdom totaled £16.4
million. U.K. tax credit carryforwards can be
carried forward indefinitely to be offset against future tax liabilities of the Company. As of December 31, 2023, the Company’s
net operating loss carryforwards in the United States of America totaled £1.2 million. U.S. tax credit carryforwards can be
carried forward indefinitely to be offset against future tax liabilities of the TC BioPharm (North America) Inc.
Significant
components of the Company’s deferred tax assets as of December 31, 2023 and 2022 are summarized below.
SCHEDULE
OF COMPONENTS OF DEFERRED TAX ASSETS
| | |
2023 | | |
2022 | |
| Deferred tax assets: | |
| | | |
| | |
| Net operation losses | |
£ | 4,357,668 | | |
£ | 4,433,305 | |
| Fixed
asset |
|
|
79,338 |
|
|
|
71,426 |
|
| Other |
|
|
17,946 |
|
|
|
308,118 |
|
| Total
deferred tax assets | |
| 4,454,952 | | |
| 4,812,849 | |
| | |
| | | |
| | |
| Valuation
allowance | |
| (4,454,952 | ) | |
| (4,812,849 | ) |
| | |
| | | |
| | |
| Net
deferred tax asset, net of valuation allowance | |
£ | – | | |
£ | – | |
After
weighing all available positive and negative evidence for the periods ended December 31, 2023 and 2022, the Company has recorded a valuation
allowance of £4,454,952 and £4,812,849, respectively.
The
Company continuously monitors its current and prior filing positions in order to determine if any unrecognized tax positions should be
recorded. The analysis involves considerable judgement and is based on the best information available. For the periods ended December
31, 2023 and 2022, the Company is not aware of any positions which require an uncertain tax position liability.
NOTE
16. SUBSEQUENT EVENTS
Management
evaluated subsequent events and transactions that occurred after the balance sheet date, up to the date that the financial statements
were issued. Based upon this review, other than as set forth below, management did not identify any subsequent events that would have
required adjustment or disclosure in the financial statements.
Exercise of Pre-funded Warrants
During
January 2024, pre-funded warrants representing 1,398,000 ADSs were exercised in
three separate tranches.
Issuances of ADSs and
grants of options to purchase ADSs
On
February 29, 2024, the Renumeration Committee of the Board of Directors approved a grant of options to purchase ADSs to the Company’s
executive officers and non-employee directors under the Company’s 2021 Shares Option Scheme (the “Plan”) and agreed
to cancel all shares options previously issued to such persons. Each non-employee director received an option to purchase 41,760 ADSs,
or ADSs representing 835,200 Ordinary Shares at an exercise price of $1.09 per ADS, which is equal to the closing price of the Company’s
ADSs on the Nasdaq Capital Market on January 31, 2024. Martin Thorp, the Company’s Chief Financial Officer, received an option
to purchase 177,122 ADSs, or ADSs representing 3,542,440 Ordinary Shares at an exercise price of $1.09 per ADS, which is equal to the
closing price of the Company’s ADSs on the Nasdaq Capital Market on January 31, 2024. Bryan Kobel, the Company’s Chief Executive
Officer received an option to purchase 381,606 ADSs, or ADSs representing 7,632,120 Ordinary Shares at an exercise price of $1.09 per
ADS, which is equal to the closing price of the Company’s ADSs on the Nasdaq Capital Market on January 31, 2024. All share options
that were issued vest immediately upon issuance.
On
March 8, 2024, Bryan Kobel, the Chief Executive Officer of the Company agreed to (a) defer the payment of accrued but
unpaid contractual pension benefits owed to him in the amount of $66,000 for a period of 9 months and (b) convert an aggregate amount
of $24,760 of accrued but unpaid contractual pension benefits owed to him into 476,153 ordinary shares, par value £0.0001 per share
of Company, based on a price per share equal to the closing price of the Company’s ADSs
on the Nasdaq Capital Market on March 7, 2024 divided by twenty (20), or the ADS ratio.
In
addition, the board of directors approved a grant of options to Mr. Kobel purchase 153,000 ADSs, or ADSs representing 3,060,000 Ordinary
Shares at an exercise price of $2.00 per ADS, which is a premium to the closing price of the Company’s ADSs on the Nasdaq Capital
Market on March 7, 2024. The options granted to Mr. Kobel were issued under the Company’s 2021 Plan.
All share options that were issued vested immediately.
Exercise
of Series D Warrants
On
March 12, 2024, the Company issued 623,750
ADS representing 12,475,000
ordinary shares of the Company upon exercise of outstanding Series D warrants resulting in gross cash proceeds to the Company of
£986,398
(approximately $1,263,000).
| Item
9. |
Changes
and Disagreements with Accountants on Accounting and Financial Disclosure |
None.
| Item
9A. |
Controls
and Procedures |
Evaluation
of Disclosure Controls and Procedures
Our
management, with the participation of our Chief Executive Officer and Chief Financial Officer, evaluated the effectiveness of our disclosure
controls and procedures (as defined in Rule 13a-15(e) of the Exchange Act) as of the end of the period covered by this report. Based
on that evaluation, our Chief Executive Officer and Chief Financial Officer concluded that our disclosure controls and procedures as
of the end of the period covered by this report were not effective at a reasonable assurance level due to the material weaknesses in
internal control over financial reporting described below. The Company’s disclosure controls and procedures are designed to provide
reasonable assurance that information required to be disclosed by us in reports that we file or submit under the Exchange Act (i) is
recorded, processed, summarized and reported within the time periods specified in the SEC’s rules and forms; and (ii) accumulated
and communicated to management, including our Chief Executive Officer and Chief Financial Officer, as appropriate, to allow timely discussions
regarding required disclosure. We believe that a control system, no matter how well designed and operated, cannot provide absolute assurance
that the objectives of the control system are met, and no evaluation of controls can provide absolute assurance that all control issues
and instances of fraud, if any, within a company have been detected.
Management’s
Annual Report on Internal Control Over Financial Reporting
Management
is responsible for establishing and maintaining adequate internal control over financial reporting. Our internal control over financial
reporting is a process designed under the supervision of our principal executive and principal financial officer and effected by our
Board of Directors, management and other personnel, to provide reasonable assurance regarding the reliability of financial reporting
and the preparation of our consolidated financial statements for external reporting purposes in accordance with GAAP.
Because
of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. In addition, projections
of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in
conditions or that the degree of compliance with the policies or procedures may deteriorate.
We
are a “smaller reporting company” as defined in Item 10(f)(1) of Regulation S-K under the Securities Act. For as long as
we continue to be a smaller reporting company, we may take advantage of exemptions from various reporting requirements that are applicable
to other public companies that are not smaller reporting companies. Additionally, this Report does not contain an attestation report
of our registered public accounting firm regarding internal control over financial reporting since the Company, as a smaller reporting
company and non-accelerated filer, is not required to provide such report.
Material
Weaknesses in Internal Control over Financial Reporting
Management
assessed the effectiveness of our internal control over financial reporting as of December 31, 2023 based on the framework established
in Internal Control—Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of the Treadway Commission.
Based on this assessment, management has determined that our internal control over financial reporting as of December 31, 2023 was not
effective.
We identified a material weakness in our internal control over accounting
for complex financial instruments (including in determining the appropriate valuation basis in areas requiring significant judgement)
during the years ended December 31, 2022 and December 31, 2023, and in the accounting for our property leases on conversion from IFRS
to GAAP, which remained unremediated as of December 31, 2023, prior to processing corrective adjustments to the financial information
which is included in the financial statements that are included in this Form 10K. After identifying the material weaknesses, we implemented
measures designed to improve our financial control in the aforementioned areas through early engagement with third party technical accounting
experts and those measures are now in operation.
Changes
in Internal Control Over Financial Reporting
There
have been no changes in our internal control over financial reporting (as defined in Rules 13a-15(f) and 15d-15(f) of the Exchange Act)
during the fiscal year ended December 31, 2023, that have materially affected, or are reasonably likely to materially affect, our internal
control over financial reporting.
| Item
9B. |
Other
Information |
None.
| Item
9C. |
Disclosure
Regarding Foreign Jurisdictions that Prevent Inspections |
Not
applicable.
PART
III
| Item
10. |
Directors,
Executive Officers, and Corporate Governance |
The
information required by this item will be filed by amendment to the 10-K not later than 30 days after the applicable due date or be incorporated by reference to our Proxy Statement for our 2024 Annual General Meeting of Shareholders
to be filed with the SEC within 120 days after the end of the fiscal year ended December 31, 2023.
Code
of Business Conduct and Ethics
We
have adopted a Code of Business Conduct and Ethics, or Code of Ethics, applicable to our employees, senior management, and directors,
including our principal executive officer, principal financial officer, principal accounting officer or controller, or persons performing
similar functions. A current copy of the Code of Ethics is posted on our website, which is located at www.tcbiopharm.com. Information
contained on, or that can be accessed through, our website does not constitute a part of this Annual Report and is not incorporated by
reference herein.
| Item
11. |
Executive
Compensation |
The
information required by this item will be filed by amendment to the 10-K not later than 30 days after the applicable due date or be incorporated by reference to our Proxy Statement for our 2024 Annual General Meeting of Shareholders
to be filed with the SEC within 120 days after the end of the fiscal year ended December 31, 2023.
| Item
12. |
Security
Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters |
The
information required by this item will be filed by amendment to the 10-K not later than 30 days after the applicable due date or be incorporated by reference to our Proxy Statement for our 2024 Annual General Meeting of Shareholders
to be filed with the SEC within 120 days after the end of the fiscal year ended December 31, 2023.
| Item
13. |
Certain
Relationships and Related Party Transactions and Director Independence |
The information required by this item will be filed by amendment to the 10-K not later than 30 days after the applicable due date or be incorporated by reference to our
Proxy Statement for our 2024 Annual General Meeting of Shareholders to be filed with the SEC within 120 days after the end of the fiscal
year ended December 31, 2023.
| Item
14. |
Principal
Accountant Fees and Services |
The
information required by this item will be filed by amendment to the 10-K not later than 30 days after the applicable due date or be incorporated by reference to our Proxy Statement for our 2024 Annual General Meeting of Shareholders
to be filed with the SEC within 120 days after the end of the fiscal year ended December 31, 2023.
PART
IV
| Item
15. |
Exhibits
and Financial Statement Schedules |
(a)
1. Financial Statements
For
a list of the financial statements included herein, see Index to the Financial Statements on page 83 of this Annual Report, incorporated
into this Item by reference.
2.
Financial Statement Schedules
Financial
statement schedules have been omitted because they are either not required or not applicable or the information is included in the financial
statements or the notes thereto.
3.
Exhibits
The
exhibits required by Item 601 of Regulation S-K and Item 15(b) of this Annual Report are listed in the Exhibit Index below. The exhibits
listed in the Exhibit Index are incorporated by reference herein.
| Exhibit |
|
Description |
|
Schedule/
Form |
|
File
Number |
|
Exhibit |
|
File
Date |
| 3.1 |
|
Articles of Association of TC BioPharm (Holdings) plc |
|
F-1 |
|
333-260492 |
|
3.2 |
|
03/08/2022 |
| 3.2 |
|
Amendment to Articles of Association |
|
6-K |
|
001-41231 |
|
99.1 |
|
10/06/2022 |
| 4.1 |
|
Deposit Agreement – Bank of New York Mellon for American Depositary Shares |
|
F-1 |
|
333-260492 |
|
4.1 |
|
01/14/2022 |
| 4.2 |
|
Form of American Depositary Share (included in Exhibit 2.1) |
|
F-1 |
|
333-260492 |
|
4.2 |
|
01/14/2022 |
| 4.3 |
|
Warrant Agent Agreement with Computershare Inc. |
|
F-1 |
|
333-260492 |
|
4.4 |
|
01/14/2022 |
| 4.4 |
|
Form of Warrant Certificate (included in Exhibit 2.3) |
|
F-1 |
|
333-260492 |
|
4.5 |
|
01/14/2022 |
| 4.5 |
|
Form of Ordinary Share Certificate |
|
F-1 |
|
333-260492 |
|
4.6 |
|
01/14/2022 |
| 4.6 |
|
Form of Representative Warrant |
|
F-1 |
|
333-260492 |
|
4.3 |
|
01/14/2022 |
| 4.7 |
|
Description of Securities of Registrant |
|
20-F |
|
001-41231 |
|
4.11 |
|
05/13/2022 |
| 4.8 |
|
Form
of Pre-Funded Warrant |
|
6-K |
|
001-41231 |
|
10.1 |
|
11/30/2022 |
| 4.9 |
|
Form
of Series A and Series B Ordinary Warrant |
|
6-K |
|
001-41231 |
|
10.2 |
|
11/30/2022 |
| 4.10 |
|
Form
of Placement Agent Warrant |
|
6-K |
|
001-41231 |
|
10.3 |
|
11/30/2022 |
| 4.11 |
|
Form of Pre-Funded Warrant |
|
6-K |
|
001-41231 |
|
10.1 |
|
03/23/2023 |
| 4.12 |
|
Form of Placement Agent Warrant |
|
6-K |
|
001-41231 |
|
10.2 |
|
03/23/2023 |
| 4.13 |
|
Form of Series C Ordinary Warrant |
|
6-K |
|
001-41231 |
|
10.3 |
|
03/23/2023 |
| 4.14 |
|
Form of Series D Warrant |
|
6-K |
|
001-41231 |
|
4.1 |
|
08/31/2023 |
| 4.15 |
|
Form of Pre-Funded Warrant |
|
6-K |
|
001-41231 |
|
10.1 |
|
12/21/2023 |
| 4.16 |
|
Form of Placement Agent Warrant |
|
6-K |
|
001-41231 |
|
10.3 |
|
12/21/2023 |
| 4.17 |
|
Form of Series E Warrant |
|
6-K |
|
001-41231 |
|
10.2 |
|
12/21/2023 |
| 10.1 |
|
Form of 2014 Share Option Scheme of Registrant |
|
F-1 |
|
333-260492 |
|
10.1 |
|
01/14/2022 |
| 10.2 |
|
Form of 2021 Share Option Scheme (including sub-plan for U.S. based persons) of Registrant |
|
F-1 |
|
333-260492 |
|
10.2 |
|
01/14/2022 |
| 10.3 |
|
Form of 2021 Company Share Option Plan (CSOP) of Registrant |
|
F-1 |
|
333-260492 |
|
10.3 |
|
01/14/2022 |
| 10.4 |
|
Convertible Loan Note, up to $20,000,000 in principal amount |
|
F-1 |
|
333-260492 |
|
10.6 |
|
01/14/2022 |
| 10.5 |
|
Form of Lock Up Agreement of Pre-IPO Smaller Shareholders |
|
F-1 |
|
333-260492 |
|
10.8 |
|
01/14/2022 |
| 10.6 |
|
Form of Lock Up Agreement of Pre-IPO Management and Larger Shareholders |
|
F-1 |
|
333-260492 |
|
10.9 |
|
01/14/2022 |
| 10.7 |
|
Form of Lock Up Agreement of Holders of Convertible Loan Notes |
|
F-1 |
|
333-260492 |
|
10.10 |
|
01/14/2022 |
| 10.8 |
|
Form of Deed of Indemnity for directors and officer |
|
20-F |
|
001-41231 |
|
4.10 |
|
05/13/2022 |
| 10.9 |
|
Form of Securities Purchase Agreement for Nov 2022 Private Placement |
|
6-K |
|
001-41231 |
|
10.4 |
|
11/30/2022 |
| 10.10 |
|
Form of Registration Rights Agreement for Nov 2022 Private Placement |
|
6-K |
|
001-41231 |
|
10.5 |
|
11/30/2022 |
| 10.11 |
|
Form of Securities Purchase Agreement for March 2023 Offering |
|
6-K |
|
001-41231 |
|
10.4 |
|
03/30/2023 |
| 10.12 |
|
Warrant Amendment Agreement, dated March 27, 2023 |
|
6-K |
|
001-41231 |
|
10.5 |
|
03/30/2023 |
| 10.13 |
|
Form of Warrant Amendment Agreement, dated July 10, 2023 |
|
6-K |
|
001-41231 |
|
10.1 |
|
07/24/2023 |
| 10.14 |
|
Form of Inducement Letter, dated August 30, 2023 |
|
6-K |
|
001-41231 |
|
10.1 |
|
08/31/2023 |
| 10.15 |
|
Form of Securities Purchase Agreement for December 2023 Offering |
|
6-K |
|
001-41231 |
|
10.4 |
|
12/21/2023 |
| 10.16 |
|
Form of Warrant Amendment Agreement, dated December 19, 2023 |
|
6-K |
|
001-41231 |
|
10.5 |
|
12/21/2023 |
| 14.1 |
|
Code of Ethics of the Registrant |
|
F-1 |
|
333-260492 |
|
11.1 |
|
01/14/2022 |
| 21.1 |
|
List of Subsidiaries of Registrant |
|
F-1 |
|
333-260492 |
|
21.1 |
|
01/14/2022 |
| |
|
|
|
|
|
|
|
|
|
|
| 31.1* |
|
Certification by Principal Executive Officer Pursuant to Section 302 of the Sarbanes-Oxley Act of 2022 |
|
|
|
|
|
|
|
|
| 31.2* |
|
Certification by Principal Executive Officer Pursuant to Section 302 of the Sarbanes-Oxley Act of 2022 |
|
|
|
|
|
|
|
|
| 32.1* |
|
Certification by Principal Executive Officer and Principal Financial Officer Pursuant to Section 906 of the Sarbanes-Oxley Act of 2022 |
|
|
|
|
|
|
|
|
| 97.1* |
|
Clawback Policy |
|
|
|
|
|
|
|
|
| 101.INS |
|
Inline
XBRL Instance Document (the instance document does not appear in the Interactive Data File because its XBRL tags are embedded within
the Inline XBRL document) |
| 101.SCH |
|
Inline
XBRL Taxonomy Extension Schema Document |
| 101.CAL |
|
Inline
XBRL Taxonomy Extension Calculation Linkbase Document |
| 101.DEF |
|
Inline
XBRL Taxonomy Extension Definition Linkbase Document |
| 101.LAB |
|
Inline
XBRL Taxonomy Extension Label Linkbase Document |
| 101.PRE |
|
Inline
XBRL Taxonomy Extension Presentation Linkbase Document |
| 104 |
|
Cover
Page Interactive Data File (formatted in IXBRL, and included in exhibit 101). |
| Item
16. |
Form
10-K Summary |
The
Company has elected not to include summary information.
SIGNATURES
Pursuant
to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed
on its behalf by the undersigned, thereunto duly authorized.
| |
TC
BIOPHARM (HOLDINGS) PLC |
| |
|
| |
By: |
/s/
Bryan Kobel |
| |
|
Bryan
Kobel
Chief
Executive Officer |
| |
|
|
| |
By:
|
/s/
Martin Thorp |
| |
|
Martin
Thorp
Chief
Financial Officer |
Date:
April 1, 2024







